Abstract
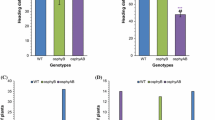
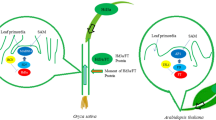
Photoperiod and temperature are two pivotal regulatory factors of plant flowering. The floral transition of plants depends on accurate measurement of changes in photoperiod and temperature. The flowering time of rice (Oryza sativa) as a facultative short-day (SD) plant is delayed under long-day (LD) and/or low temperature conditions. To elucidate the regulatory functions of photoperiod and temperature on flowering time in rice, we systematically analyzed the expression and regulation of several key genes (Hd3a, RFT1, Ehd1, Ghd7, RID1/Ehd2/OsId1, Se5) involved in the photoperiodic flowering regulatory pathway under different temperature and photoperiod treatments using a photoperiod-insensitive mutant and wild type plants. Our results indicate that the Ehd1-Hd3a/RFT1 pathway is common to and conserved in both the photoperiodic and temperature flowering regulatory pathways. Expression of Ehd1, Hd3a and RFT1 is dramatically reduced at low temperature (23°C), suggesting that suppression of Ehd1, Hd3a and RFT1 transcription is an essential cause of delayed flowering under low temperature condition. Under LD condition, Ghd7 mRNA levels are promoted at low temperature (23°C) compared with normal temperature condition (28°C), suggesting low temperature and LD treatment have a synergistic role in the expression of Ghd7. Therefore, upregulation of Ghd7 might be a crucial cause of delayed flowering under low temperature condition. We also analyzed Hd1 regulatory relationships in the photoperiodic flowering pathway, and found that Hd1 can negatively regulate Ehd1 transcription under LD condition. In addition, Hd1 can also positively regulate Ghd7 transcription under LD condition, suggesting that the heading-date of rice under LD condition is also regulated by the Hd1-Ghd7-Ehd1-RFT1 pathway.
Article PDF
Similar content being viewed by others
References
Izawa T. Daylength measurements by rice plants in photoperiodic short-day flowering. Int Rev Cytol, 2007, 256: 191–222
Itoh H, Nonoue Y, Yano M, et al. A pair of floral regulators sets critical day length for Hd3a florigen expression in rice. Nat Genet, 2010, 42: 635–638
Kojima S, Takahashi Y, Kobayashi Y, et al. Hd3a, a rice ortholog of the Arabidopsis FT gene, promotes transition to flowering downstream of Hd1 under short-day conditions. Plant Cell Physiol, 2002, 43: 1096–1105
Corbesier L, Vincent C, Jang S, et al. FT protein movement contributes to long-distance signaling in floral induction of Arabidopsis. Science, 2007, 316: 1030–1033
Tamaki S, Matsuo S, Wong H L, et al. Hd3a protein is a mobile flowering signal in rice. Science, 2007, 316: 1033–1036
Yano M, Katayose Y, Ashikari M, et al. Hd1, a major photoperiod sensitivity quantitative trait locus in rice, is closely related to the Arabidopsis flowering time gene CONSTANS. Plant Cell, 2000, 12: 2473–2483
Hayama R, Yokoi S, Tamaki S, et al. Adaptation of photoperiodic control pathways produces short-day flowering in rice. Nature, 2003, 422: 719–722
Putterill J, Robson F, Lee K, et al. The CONSTANS gene of Arabidopsis promotes flowering and encodes a protein showing similarities to zinc finger transcription factors. Cell, 1995, 80: 847–857
Suárez-López P, Wheatley K, Robson F, et al. CONSTANS mediates between the circadian clock and the control of flowering in Arabidopsis. Nature, 2001, 410: 1116–1120
Komiya R, Ikegami A, Tamaki S, et al. Hd3a and RFT1 are essential for flowering in rice. Development, 2008, 135: 767–774
Doi K, Izawa T, Fuse T, et al. Ehd1, a B-type response regulator in rice, confers short-day promotion of flowering and controls FT-like gene expression independently of Hd1. Genes Dev, 2004, 18: 926–936
Wu C, You C, Li C, et al. RID1, encoding a Cys2/His2-type zinc finger transcription factor, acts as a master switch from vegetative to floral development in rice. Proc Natl Acad Sci USA, 2008, 105: 12915–12920
Matsubara K, Yamanouchi U, Wang Z, et al. Ehd2, a rice ortholog of the maize INDETERMINATE1 gene, promotes flowering by up-regulating Ehd1. Plant Physiol, 2008, 148: 1425–1435
Park S J, Kim S L, Lee S, et al. Rice indeterminate 1 (OsId1) is necessary for the expression of Ehd1 (early heading date 1) regardless of photoperiod. Plant J, 2008, 56: 1018–1029
Kim S L, Lee S, Kim H J, et al. OsMADS51 is a short-day flowering promoter that functions upstream of Ehd1, OsMADS14, and Hd3a. Plant Physiol, 2007, 145: 1484–1494
Izawa T, Oikawa T, Sugiyama N, et al. Phytochrome mediates the external light signal to repress FT orthologs in photoperiodic flowering of rice. Genes Dev, 2002, 16: 2006–2020
Xue W, Xing Y, Weng X, et al. Natural variation in Ghd7 is an important regulator of heading date and yield potential in rice. Nat Genet, 2008, 40: 761–767
Izawa T, Oikawa T, Tokutomi S, et al. Phytochromes confer the photoperiodic control of flowering in rice (a short day plant). Plant J, 2000, 22: 391–399
Andre’s F, Galbraith D W, Talo’n M, et al. Analysis of PHOTOPERIOD SENSITIVITY 5 sheds light on the role of phytochromes in photoperiodic flowering in rice. Plant Physiol, 2009, 151: 681–690
Ryu C H, Lee S, Cho L H, et al. OsMADS50 and OsMADS56 function antagonistically in regulating long day (LD)-dependent flowering in rice. Plant Cell Environ, 2009, 32: 1412–1427
Matsubara K, Yamanouchi U, Nonoue Y, et al. Ehd3, encoding a plant homeodomain finger-containing protein, is a critical promoter of rice flowering. Plant J, 2011, 66: 603–612
Komiya R, Yokoi R, Shimamoto K. A gene network for long-day flowering activates RFT1 encoding a mobile flowering signal in rice. Development, 2009, 136: 3443–3450
Luan W, Chen H, Fu Y, et al. The effect of the crosstalk between photoperiod and temperature on the heading-date in rice. PLoS ONE, 2009, 4: e5891
Wei X, Xu J, Guo H, et al. DTH8 suppresses flowering in rice, influencing plant height and yield potential simultaneously. Plant Physiol, 2010, 153: 1747–1758
Yan W, Wang P, Chen H, et al. A major QTL, Ghd8, plays pleiotropic roles in regulating grain productivity, plant height, and heading date in rice. The Molecular Plant advance online publication December 10, 2010; doi:10.1093/mp/ssq070
Amasino R M. Vernalization and flowering time. Curr Opin Biotech, 2005, 16: 154–158
Searle I, He Y H, Turck F, et al. The transcription factor FLC confers a flowering response to verbalization by repressing meristem competence and systemic signaling in Arabidopsis. Genes Dev, 2006, 20: 898–912
Michaels S D, Amasino R M. FLOWERING LOCUS C encodes a novel MADS domain protein that acts as a repressor of flowering. Plant Cell, 1999, 11: 949–956
Helliwell C A, Wood C C, Robertson M, et al. The Arabidopsis FLC protein interacts directly in vivo with SOC1 and FT chromatin and is part of a high-molecular-weight protein complex. Plant J, 2006, 46: 183–192
Lee J H, Yoo S J, Park S H. Role of SVP in the control of flowering time by ambient temperature in Arabidopsis. Genes Dev, 2007, 21: 397–402
Kumar S V, Wigge P A. H2A.Z-Containing nucleosomes mediate the thermosensory response in Arabidopsis. Cell, 2010, 140: 136–147
Lee S, Jeong D, An G. A possible working mechanism for rice SVP-group MADS-box proteins as negative regulators of brassinosteroid responses. Plant Signal Behav, 2008, 3: 471–474
Xu W X. A novel rice cultivar Heigeng 5, can be planted in high latitude and cold regions. China Rice, 1999, 3: 16
Balasubramanian S, Sureshkumar S, Lempe J, et al. Potent induction of Arabidopsis thaliana flowering by elevated growth temperature. PLoS Genet, 2006, 2: e106
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Song, Y., Gao, Z. & Luan, W. Interaction between temperature and photoperiod in regulation of flowering time in rice. Sci. China Life Sci. 55, 241–249 (2012). https://doi.org/10.1007/s11427-012-4300-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-012-4300-4