Abstract
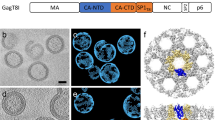
The three-dimensional structure of recombinant hepatitis B core antigen (HBcAg) particles truncated at residue 154 (HBcAg-154) was determined to 7.8 Å resolution by cryo-electron microscopy (cryoEM) and computer reconstruction. The capsid of HBcAg-154 is mainly constituted by α-helical folds, highly similar to that of HBcAg-149. The C-terminal region between residues 155 and 183 of the core protein is more crucial to the encapsidation of RNA, and the short C-terminal tail of HBcAg-154 results in a nearly empty capsid.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Chisari F V, Ferrari C. Hepatitis-B virus immunopathogenesis. Annu Rev Immunol, 1995, 13: 29–60, 1:CAS:528:DyaK2MXltFamtLY%3D, 10.1146/annurev.iy.13.040195.000333, 7612225
Beck J, Nassal M. Hepatitis B virus replication. World J Gastroenterol, 2007, 13: 48–64, 1:CAS:528:DC%2BD2sXitFSgt7w%3D, 17206754
Ganem D. Hepadnaviridae and their replication. In: Fields B N, Knipe D M, Howley P M, eds. Fields Virology. Philadelphia: Raven Press, 1996. 2703–2737
Wingfield P T, Stahl S J, Williams R W, et al. Hepatitis core antigen produced in Escherichia coli: Subunit composition, conformational analysis, and in vitro capsid assembly. Biochemistry, 1995, 34: 4949–4932, 10.1021/bi00015a003
Böttcher B, Wynne S A, Crowther R A. Determination of the fold of the core protein of hepatitis B virus by electron cryomicroscopy. Nature, 1997, 386: 88–91, 10.1038/386088a0, 9052786
Conway J F, Cheng N, Zlotnick A, et al. Visualization of a 4-helix bundle in the hepatitis B virus capsid by cryo-electron microscopy. Nature, 1997, 386: 91–94, 1:CAS:528:DyaK2sXhslCnsb8%3D, 10.1038/386091a0, 9052787
Wynne S A, Crowther R A, Leslie A G. The crystal structure of the human hepatitis B virus capsid. Mol Cell, 1999, 3: 771–780, 1:CAS:528:DyaK1MXktlOktbs%3D, 10.1016/S1097-2765(01)80009-5, 10394365
Dryden K A, Wieland S F, Whitten-Bauer C, et al. Native hepatitis B virions and capsids visualized by electron cryomicroscopy. Mol Cell, 2006, 22: 843–850, 1:CAS:528:DC%2BD28XmvVCitb8%3D, 10.1016/j.molcel.2006.04.025, 16793552
Roseman A M, Berriman J A, Wynne S A, et al. A structural model for maturation of the hepatitis B virus core. Proc Natl Acad Sci USA, 2005, 102: 15821–15826, 1:CAS:528:DC%2BD2MXht1Wru7nJ, 10.1073/pnas.0504874102, 16247012
Le Pogam S, Chua P K, Newman M, et al. Exposure of RNA templates and encapsidation of spliced viral RNA are influenced by the arginine-rich domain of human hepatitis B virus core antigen (HBcAg 165–173). J Virol, 2005, 79: 1871–1887, 10.1128/JVI.79.3.1871-1887.2005, 15650211
Yu X, Jin L, Zhou Z H. 3.88 Å structure of cytoplasmic polyhedrosis virus by cryo-electron microscopy. Nature, 2008, 453: 415–419, 1:CAS:528:DC%2BD1cXlvV2qtLs%3D, 10.1038/nature06893, 18449192
Ludtke S J, Baldwin P R, Chiu W. EMAN: Semi-automated software for high resolution single particle reconstructions. J Struct Biol, 1999, 128: 82–97, 1:CAS:528:DyaK1MXnvFejsrw%3D, 10.1006/jsbi.1999.4174, 10600563
Zhou Z H, Chiu W. Determination of icosahedral virus structures by electron cryomicroscopy at subnanometer resolution. Adv Protein Chem, 2003, 64: 93–124, 1:CAS:528:DC%2BD3sXovFKju7o%3D, 10.1016/S0065-3233(03)01003-9, 13677046
Liang Y, Ke E Y, Zhou Z H. IMIRS: A high-resolution 3D reconstruction package integrated with a relational image database. J Struct Biol, 2002, 137: 292–304, 1:CAS:528:DC%2BD38XltVKmurc%3D, 10.1016/S1047-8477(02)00014-X, 12096897
Crowther R A. Procedures for three-dimensional reconstruction of spherical viruses by Fourier synthesis from electron micrographs. Philos Trans R Soc Lond B Biol Sci, 1971, 261: 221–230, 1:STN:280:DyaE38%2FksVSksw%3D%3D, 10.1098/rstb.1971.0054, 4399207
Fuller S D. The T=4 envelope of Sindbis virus is organized by interactions with a complementary T=3 capsid. Cell, 1987, 48: 923–934, 1:CAS:528:DyaL1cXlt1GhsA%3D%3D, 10.1016/0092-8674(87)90701-X, 3829124
Baker T S, Cheng R H. A model-based approach for determining orientations of biological macromolecules imaged by cryoelectron microscopy. J Struct Biol, 1996, 116: 120–130, 1:CAS:528:DyaK28XitVKiur0%3D, 10.1006/jsbi.1996.0020, 8742733
Liu H, Cheng L, Zeng S, et al. Symmetry-adapted spherical harmonics method for high-resolution 3D single-particle reconstructions. J Struct Biol, 2008, 161: 64–73, 1:CAS:528:DC%2BD2sXhsVOrtLvL, 10.1016/j.jsb.2007.09.016, 17977017
Zhou Z H, Hardt S, Wang B, et al. CTF determination of images of ice-embedded single particles using a graphics interface. J Struct Biol, 1996, 116: 216–222, 1:STN:280:DyaK28zmtF2itg%3D%3D, 10.1006/jsbi.1996.0033, 8742746
Zhou Z H, Chen D H, Jakana J, et al. Visualization of tegumentcapsid interactions and DNA in intact herpes simplex virus type 1 virions. J Virol, 1999, 73: 3210–3218, 1:CAS:528:DyaK1MXhvFyntb4%3D, 10074174
Pettersen E F, Goddard T D, Huang C C, et al. UCSF Chimera—a visualization system for exploratory research and analysis. J Comput Chem, 2004, 25: 1605–1612, 1:CAS:528:DC%2BD2cXmvVOhsbs%3D, 10.1002/jcc.20084, 15264254
Crowther R A, Kiselev N A, Bottcher B, et al. Three-dimensional structure of hepatitis-B virus core particles determined by electron cryomicroscopy. Cell, 1994, 77: 943–950, 1:CAS:528:DyaK2cXkslyit7c%3D, 10.1016/0092-8674(94)90142-2, 8004680
Author information
Authors and Affiliations
Corresponding author
Additional information
Contributed equally to this work
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License ( https://creativecommons.org/licenses/by-nc/2.0 ), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Liu, S., He, J., Li, K. et al. Three-dimensional structure of the hepatitis B core antigen particle truncated at residue 154. Sci. China Life Sci. 54, 171–174 (2011). https://doi.org/10.1007/s11427-010-4098-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-010-4098-x