Abstract
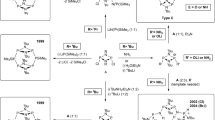
Previously we have shown that POCl3-mediated H-bonding-directed one-pot macrocyclization allows for the highly selective preparation of five-residue macrocycles as the predominant product with low yields of hexamers and an undetectable occurrence of both tetramers and heptamers. Replacing the interiorly arrayed methyl groups with ethyl groups in these 4–7 residue macrocycles alters the relative stability order among them. Specifically, ethoxy-substituted six-residue macrocycle, rather than pentamer, turns out to be computationally the most stable, suggesting that ethoxy-containing hexamer possibly can be formed as the major product under suitable conditions. We have investigated this possibility by varying reaction temperatures and concentrations, invariably affording pentamer as the major macrocycle with strained circular hexamers and highly strained circular heptamers produced in substantial amounts. This discrepancy can be reasonably explained on the basis of bimolecular reactions between two oligomers higher than monomers via kinetic simulations. In this scenario, the acyclic pentamer is kinetically “trapped” to undergo an intramolecular cyclization to yield circular pentamer, rather than to produce acyclic hexamer. As a result, acyclic hexamer precursor is generated largely from sterically demanding bimolecular reactions between a dimer and a tetramer, or between two trimers that are kinetically slower than the pentamer-producing chain-growth reactions. We additionally found that one-pot macrocyclization proceeds to the largest extent at 40 °C, an intriguing finding that highlights the low reactivities of acid chloride and amine groups in these H-bond-enforced acyclic oligomeric intermediates.
Similar content being viewed by others
References
Pedersen CJ. Cyclic polyethers and their complexes with metal salts. J Am Chem Soc, 1967, 89: 7017–7086
Dietrich B, Lehn JM, Sauvage J-P. Diazapolyoxamacrocycles and macrobicycles. Tetrahedron Lett, 1969, 34: 2885–2889
Kyba EP, Siegel MG, Sousa LR, Sogah GDY, Cram DJ. Chiral, hinged, and functionalized multiheteromacrocycles. J Am Chem Soc, 1973, 95: 2691–2692
Sanford AR, Yuan L, Feng W, Yamato K, Flowersb RA, Gong B. Cyclic aromatic oligoamides as highly selective receptors for the guanidinium ion. Chem Commun, 2005: 4720–4722
Shirude PS, Gillies ER, Ladame S, Godde F, Shin-Ya K, Huc I, Balasubramanian S. Macrocyclic and helical oligoamides as a new class of g-quadruplex ligands. J Am Chem Soc, 2007, 129: 11890–11891
Helsel AJ, Brown AL, Yamato K, Feng W, Yuan LH, Clements AJ, Harding SV, Szabo G, Shao ZF, Gong B. Highly conducting transmembrane pores formed by aromatic oligoamide macrocycles. J Am Chem Soc, 2008, 130: 15784–15785
Zhu YY, Li C, Li GY, Jiang XK, Li ZT. Hydrogen-bonded aryl amide macrocycles: Synthesis, single-crystal structures, and stacking interactions with fullerenes and coronene. J Org Chem, 2008, 73: 1745–1751
Qin B, Chen XY, Fang X, Shu YY, Yip YK, Yan Y, Pan SY, Ong WQ, Ren CL, Su HB, et al. Crystallographic evidence of an unusual, pentagon-shaped folding pattern in a circular aromatic pentamer. Org Lett, 2008, 10: 5127–5130
Qin B, Ren CL, Ye RJ, Sun C, Chiad K, Chen XY, Li Z, Xue F, Su HB, Chass GA, et al. Persistently folded circular aromatic amide pentamers containing modularly tunable cation-binding cavities with high ion selectivity. J Am Chem Soc, 2010, 132: 9564–9566
Qin B, Ong WQ, Ye RJ, Du ZY, Chen XY, Yan Y, Zhang K, Su HB, Zeng HQ. Highly selective one-pot synthesis of H-bonded pentagon-shaped circular aromatic pentamers. Chem Commun, 2011, 47: 5419–5421
Knops P, Sendhoff N, Mekelburger HB, Voegtle F. High dilution reactions — New synthetic applications. Top Curr Chem, 1992, 161: 1–36
Faust R. Explosions as a synthetic tool? Cycloalkynes as precursors to fullererenes, buckytubes and buckyonions. Angew Chem Int Ed, 1998, 37: 2825–2828
Höger S. Shape-persistent macrocycles: From molecules to materials. Chem Eur J, 2004, 10: 1320–1329
Zhang W, Moore JS. Shape-persistent macrocycles: Structures and synthetic approaches from arylene and ethynylene building blocks. Angew Chem Int Ed, 2006, 45: 4416–4439
Woodward RB, Au-Yeung BW, Balaram P, Browne LJ, Ward DE, Au-Yeung BW, Balaram P, Browne LJ, Card PJ, Chen CH. Asymmetric total synthesis of erythromycin. 2. Synthesis of an erythronolide A lactone system. J Am Chem Soc, 1981, 103: 3213–3215
Eschenmoser A. Vitamin B12: Experiments concerning the origin of its molecular structure. Angew Chem Int Ed, 1988, 27: 5–39
Xing LY, Ziener U, Sutherland TC, Cuccia LA. Hydrogen bond directed synthesis of pyridazine and naphthyridine containing macrocycles. Chem Commun, 2005: 5751–5753
Imabeppu F, Katagiri K, Masu H, Kato T, Tominaga M, Therrien B, Takayanagi H, Kaji E, Yamaguchi K, Kagechika H, et al. Calix[3]amides-bowlshaped cyclic trimers toward building block for molecular recognition: Self-complementary dimeric structure in the crystal. Tetrahedron Lett, 2006, 47: 413–416
Campbell F, Plante J, Carruthers C, Hardie MJ, Prior TJ, Wilson AJ. Macrocyclic scaffolds derived from p-aminobenzoic acid. Chem Commun, 2007: 2240–2242
Yokoyama A, Maruyama T, Tagami K, Masu H, Katagiri K, Azumaya I, Yokozawa T. One-pot synthesis of cyclic triamides with a triangular cavity from trans-stilbene and diphenylacetylene monomers. Org Lett, 2008, 10: 3207–3210
Katagiri K, Tohaya T, Masu H, Tominaga M, Azumaya I. Effect of aromatic-aromatic interactions on the conformational stabilities of macrocycle and preorganized structure during macrocyclization. J Org Chem, 2009, 74: 2804–2810
Campbell F, Wilson AJ. An ‘impossible’ macrocyclisation using conformation directing protecting groups. Tetrahedron Lett, 2009, 50: 2236–2238
He L, An Y, Yuan L, Yamato K, Feng W, Gerlitz O, Zheng C, Gong B. Macrocyclic aromatic tetrasulfonamides with a stable cone conformation. Chem Commun, 2005: 3788–3790
He L, An Y, Yuan LH, Feng W, Li MF, Zhang DC, Yamato K, Zheng C, Zeng XC, Gong B. Shape-persistent macrocyclic aromatic tetrasulfonamides: Molecules with nanosized cavities and their nanotubular assemblies in solid state. Proc Natl Acad Sci USA, 2006, 103: 10850–10855
Geng M, Zhang D, Wu X, He L, Gong B. One-pot formation and characterization of macrocyclic aromatic tetrasulfonates. Org Lett, 2009, 11: 923–926
Carver FJ, Hunter CA, Shannon RJ. Directed macrocyclization reactions. Chem Commun, 1994: 1277–1280
Yuan L, Feng W, Yamato K, Sanford AR, Xu D, Guo H, Gong B. Highly efficient, one-step macrocyclizations assisted by the folding and preorganization of precursor oligomers. J Am Chem Soc, 2004, 126:11120–11121
Zhang AM, Han YH, Yamato K, Zeng XC, Gong B. Aromatic oligoureas: Enforced folding and assisted cyclization. Org Lett, 2006, 8: 803–806
Ahn HC, Yun SM, Choi K. A proline-based macrocyclic amide with S-4 symmetry. Chem Lett, 2008, 37: 10–11
Feng W, Yamato K, Yang LQ, Ferguson JS, Zhong LJ, Zou SL, Yuan LH, Zeng XC, Gong B. Efficient kinetic macrocyclization. J Am Chem Soc, 2009, 131: 2629–2637
Jiang H, Leger JM, Guionneau P, Huc I. Strained aromatic oligoamide macrocycles as new molecular clips. Org Lett, 2004, 6: 2985–2988
Hui JKH, MacLachlan MJ. [6+6] Schiff-base macrocycles with 12 imines: Giant analogues of cyclohexane. Chem Commun, 2006: 2480–2482
Ferguson JS, Yamato K, Liu R, He L, Zeng XC, Gong B. One-pot formation of large macrocycles with modifiable peripheries and internal cavities. Angew Chem Int Ed, 2009, 48: 3150–3154
Filarowski A, Koll A, Sobczyk L. Intramolecular hydrogen bonding in o-hydroxy Aryl Schiff Bases. Curr Org Chem, 2009, 13: 172–193
Li F, Gan Q, Xue L, Wang ZM, Jiang H. H-bonding directed one-step synthesis of novel macrocyclic peptides from ɛ-aminoquinolinecarboxylic acid. Tetrahedron Lett, 2009, 50: 2367–2369
Yang LQ, Zhong LJ, Yamato K, Zhang XH, Feng W, Deng PC, Yuan LH, Zeng XC, Gong B. Aromatic oligoamide macrocycles from the bimolecular coupling of folded oligomeric precursors. New J Chem, 2009, 33: 729–733
Zoua S, Hea Y, Yanga Y, Zhaoa Y, Yuana L, Feng W, Yamato K, Gong B. Improving the efficiency of forming ‘unfavorable’ products: eight-residue macrocycles from folded aromatic oligoamide precursors. Synlett. 2009, 9
Hu JC, Feng W, Li XH, Yang LQ, Yuan LH, He L, Gong B. One-pot formation of aromatic oligoamide macrocycles and the corresponding mechanism. Acta Phys Chim Sin, 2010, 26: 1811–1822
Yang YA, Yuan LH, Hu JC, Zou SL, Feng W, Gong B. Spherical assemblies of cyclo[6]aramide with polar side chains. Acta Phys Chim Sin, 2010, 26: 1557–1564
Qin B, Sun C, Liu Y, Shen J, Ye RJ, Zhu J, Duan XF, Zeng HQ. One-pot synthesis of hybrid macrocyclic pentamers with variable functionalizations around the periphery. Org Lett, 2011, 13: 2270–2273
Qin B, Shen S, Sun C, Du ZY, Zhang K, Zeng HQ. One-pot multi-molecular macrocyclization for the expedient synthesis of macrocyclic aromatic pentamers by a chain growth mechanism. Chem Asian J, 2011: asia.201100409
For some recent reviews in foldamers, see: (a) Gellman SH. Foldamers: A manifesto. Acc Chem Res, 1998, 31: 173–180
Gong B. Crescent oligoamides: From acyclic “macrocycles” to folding nanotubes. Chem Eur J, 2001, 7: 4336–4342
Hill DJ, Mio MJ, Prince RB, Hughes TS, Moore JS. A field guide to foldamers. Chem Rev, 2001, 101: 3893–4011
Sanford AR, Gong B. Evolution of helical foldamers. Curr Org Chem, 2003, 7: 1649–1659
Sanford AR, Yamato K, Yang X, Yuan L, Han Y, Gong B. Well-defined secondary structures — Information-storing molecular duplexes and helical foldamers based on unnatural peptide backbones. Eur J Biochem, 2004, 271: 1416–1425
Huc I. Aromatic oligoamide foldamers. Eur J Org Chem, 2004: 17–29
Cheng RP. Beyond de novo protein design-de novo design of non-natural folded oligomers. Curr Opin Struc Biol, 2004, 14: 512–520
Li ZT, Hou JL, Li C, Yi HP. Shape-persistent aromatic amide oligomers: New tools for supramolecular chemistry. Chem Asian J, 2006, 1: 766–778
Gong B. Hollow crescents, helices, and macrocycles from enforced folding and folding-assisted macrocyclization. Acc Chem Res, 2008, 41: 1376–1386
Li ZT, Hou JL, Li C. Peptide mimics by linear arylamides: A structural and functional diversity test. Acc Chem Res, 2008, 41: 1343–1353
Horne WS, Gellman SH. Foldamers with heterogeneous backbones. Acc Chem Res, 2008, 41: 1399–1408
Haldar D, Schmuck C. Metal-free double helices from abiotic backbones. Chem Soc Rev, 2009, 38: 363–371
Hamuro Y, Geib SJ, Hamilton AD. Novel molecular scaffolds: Formation of helical secondary structure in a family of oligoanthranilamides. Angew Chem Int Ed, 1994, 33: 446–448
Hamuro Y, Geib SJ, Hamilton AD. Oligoanthranilamides. non-peptide subunits that show formation of specific secondary structure. J Am Chem Soc, 1996, 118: 7529–7541
Hamuro Y, Geib SJ, Hamilton AD. Novel folding patterns in a family of oligoanthranilamides: Non-peptide oligomers that form extended helical secondary structures. J Am Chem Soc, 1997, 119: 10587–10593
Berl V, Huc I, Khoury RG, Krische MJ, Lehn JM. Interconversion of single and double helices formed from synthetic molecular strands. Nature, 2000, 407: 720–723
Berl V, Huc I, Khoury R, Lehn JM. Helical molecular programming: supramolecular double helices by dimerization of helical oligopyridine-dicarboxamide strands. Chem Eur J, 2001, 7: 2810–2820
Kolomiets E, Berl V, Odriozola I, Stadler AM, Kyritsakas N, Lehn JM. Contraction/extension molecular motion by protonation/deprotonation induced structural switching of pyridine derived oligoamides. Chem Commun, 2003: 2868–2869
Zhu J, Parra RD, Zeng HQ, Skrzypczak-Jankun E, Zeng XC, Gong B. A new class of folding oligomers: Crescent oligoamides. J Am Chem Soc, 2000, 122: 4219–4220
Gong B, Zeng HQ, Zhu J, Yuan LH, Han YH, Cheng SZ, Furukawa M, Parra RD, Kovalevsky AY, Mills JL, et al. Creating nanocavities of tunable sizes: Hollow helices. Proc Natl Acad Sci USA, 2002, 99: 11583–11588
Yuan LH, Zeng HQ, Yamato K, Sanford AR, Feng W, Atreya HS, Sukumaran DK, Szyperski T, Gong B. Helical aromatic oligoamides: Reliable, readily predictable folding from the combination of rigidified structural motifs. J Am Chem Soc, 2004, 126: 16528–16537
Yang XW, Yuan LH, Yamamoto K, Brown AL, Feng W, Furukawa M, Zeng XC, Gong B. Backbone-rigidified oligo(m-phenylene ethynylenes). J Am Chem Soc, 2004, 126: 3148–3162
Corbin PS, Zimmerman SC, Thiessen PA, Hawryluk NA, Murray TJ. Complexation-induced unfolding of heterocyclic ureas. Simple foldamers equilibrate with multiply hydrogen-bonded sheetlike structures. J Am Chem Soc, 2001, 123: 10475–10488
Jiang H, Leger J-M, Huc I. Aromatic δ-peptides. J Am Chem Soc, 2003, 125: 3448–3449
Dolain C, Leger JM, Delsuc N, Gornitzka H, Huc I. Probing helix propensity of monomers within a helical oligomer. Proc Natl Acad Sci USA, 2005, 102: 16146–16151
Haldar D, Jiang H, Leger JM, Huc I. Interstrand interactions between side chains in a double-helical foldamer. Angew Chem Int Ed, 2006, 45: 5483–5486
Gan Q, Bao CY, Kauffmann B, Grelard A, Xiang JF, Liu SH, Huc I, Jiang H. Quadruple and double helices of 8-fluoroquinoline oligoamides. Angew Chem Int Ed, 2008, 47: 1715–1718
Hou JL, Shao XB, Chen GJ, Zhou YX, Jiang XK, Li ZT. Hydrogen bonded oligohydrazide foldamers and their recognition for saccharides. J Am Chem Soc, 2004, 126: 12386–12394
Li C, Ren SF, Hou JL, Yi HP, Zhu SZ, Jiang XK, Li ZT. F…H-N Hydrogen bonding driven foldamers: Efficient receptors for dialkylammonium Ions. Angew Chem Int Ed, 2005, 44: 5725–5729
Hou JL, Yi HP, Sha XB, Li C, Wu ZQ, Jian XK, Wu LZ, Tung CH, Li ZT. Helicity induction in hydrogen-bonding-driven zinc porphyrin foldamers by chiral C60-incorporating histidines. Angew Chem Int Ed, 2006, 45: 796–800
Cai W, Wang GT, Xu YX, Jiang XK, Li ZT. Vesicles and organogels from foldamers: A solvent-modulated self-assembling process. J Am Chem Soc, 2008, 130: 6936–6937
Rebek J. Molecular behavior in small spaces. Acc Chem Res, 2009, 42: 1660–1668
Kanamori D, Okamura TA, Yamamoto H, Ueyama N. Linear-to-turn conformational switching induced by deprotonation of unsymmetrically linked phenolic oligoamides. Angew Chem Int Ed, 2005, 44: 969–972
Li X, Zhan CL, Wang YB, Yao JN. Pyridine-imide oligomers. Chem Commun, 2008: 2444–2446
Yan Y, Qin B, Shu YY, Chen XY, Yip YK, Zhang DW, Su HB, Zeng HQ. Helical organization in foldable aromatic oligoamides by a continuous hydrogen-bonding network. Org Lett, 2009, 11: 1201–1204
Yan Y, Qin B, Ren CL, Chen XY, Yip YK, Ye RJ, Zhang DW, Su HB, Zeng HQ. Synthesis, structural investigations, hydrogen-deuterium exchange studies, and molecular modeling of conformationally stablilized aromatic oligoamides. J Am Chem Soc, 2010, 132: 5869–5879
Ong WQ, Zhao HQ, Du ZY, Yeh JZY, Ren CL, Tan LZW, Zhang K, Zeng HQ. Computational prediction and experimental verification of pyridine-based helical oligoamides containing four repeating units per turn. Chem Commun, 2011, 47: 6416–6418
Ong WQ, Zhao HQ, Fang X, Woen S, Zhou F, Yap WL, Su HB, Li SFY, Zeng HQ. Encapsulation of conventional and unconventional water dimers by water-ninding foldamers. Org Lett, 2011, 13: 3194–3197
Ren CL, Xu SY, Xu J, Chen HY, Zeng HQ. Planar macrocyclic fluoropentamers as supramolecular organogelators. Org Lett, 2011: 3840–3843
Ren CL, Maurizot V, Zhao HQ, Shen J, Zhou F, Ong WQ, Du ZY, Zhang K, Su HB, Zeng HQ. Fivefold-symmetric macrocyclic aromatic pentamers: High affinity cation recognition, ion-pair induced columnar stacking and nanofibrillation. J Am Chem Soc, 2011, 133: 13930–13933
Ren CL, Zhou F, Qin B, Ye RJ, Shen S, Su HB, Zeng HQ. Crystallographic realization of the mathematically predicted densest “All Pentamer” packing lattice by C5-symmetric “sticky” fluoropentamers. Angew Chem Int Ed, 2011: DOI: 10.1002/anie.201101553
Zhang Z, Xia B, Han C, Yu Y, Huang F. Syntheses of copillar[5]arenes by co-oligomerization of different monomers. Org Lett, 2010, 12: 3285–3287
Zhang Z, Luo Y, Xia B, Han C, Yu Y, Chen X, Huang F. Four constitutional isomers of BMpillar[5]arene: Synthesis, crystal structures and complexation with n-octyltrimethyl ammonium hexafluorophosphatew. Chem Commun, 2011, 47: 2417–2419
Guieu S, Crane AK, MacLachlan MJ. Campestarenes: Novel shape-persistent Schiff base macrocycles with 5-fold symmetry. Chem Commun, 2011, 47: 1169–1171
Du ZY, Ren CL, Ye RJ, Shen J, Lu YJ, Wang J, Zeng HQ. BOP-mediated one-pot synthesis of C5-symmetric macrocyclic pyridone pentamers. Chem Commun, 2011, 47: 12488–12490
Frisch MJ: In Gaussian 03, Gaussian, Inc. Edited by: Wallingford CT; 2004
Becke AD. Density functional thermochemistry. III. The role of exact exchange. J Chem Phys, 1993, 98: 5648–5652
Petersson GA, Bennett A, Tensfeldt TG, Al-Laham MA, Shirley WA, Mantzaris J. A complete basis set model chemistry. I. The total energies of closed shell atoms and hydrides of the first row elements. J Chem Phys, 1988, 89: 2193–2218
Petersson GA, Al-Laham MA. A complete basis set model chemistry. II. Open shell systems and the total energies of the first row atoms. J Chem Phys, 1991, 94: 6081–6090
Carried out by using mechanism-based kinetics simulator avaiable from http://www.stolaf.edu/depts/chemistry/courses/toolkits/126/js/kinetics/index.htm
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, Y., Qin, B. & Zeng, H. POCl3-mediated H-bonding-directed one-pot synthesis of macrocyclic pentamers, strained hexamers and highly strained heptamers. Sci. China Chem. 55, 55–63 (2012). https://doi.org/10.1007/s11426-011-4438-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-011-4438-0



