Abstract
Upper limb motor function is a potential new biomarker of cognitive impairment and may aid discrimination from healthy ageing. However, it remains unclear which assessments to use. This study aimed to explore what methods have been used and to describe associations between upper limb function and cognitive impairment. A scoping review was conducted using PubMed, CINAHL and Web of Science. A systematic search was undertaken, including synonyms for key concepts ‘upper limb’, ‘motor function’ and ‘cognitive impairment’. Selection criteria included tests of upper limb motor function and impaired cognition in adults. Analysis was by narrative synthesis. Sixty papers published between 1998 and 2022, comprising 41,800 participants, were included. The most common assessment tasks were finger tapping, Purdue Pegboard Test and functional tasks such as writing. Protocols were diverse in terms of equipment used and recording duration. Most participants were recruited from clinical settings. Alzheimer’s Disease was the most common cause of cognitive impairment. Results were mixed but, generally, slower speed, more errors, and greater variability in upper limb movement variables was associated with cognitive impairment. This review maps the upper limb motor function assessments used and summarises the available evidence on how these associate with cognitive impairment. It identifies research gaps and may help guide protocols for future research. There is potential for upper limb motor function to be used in assessments of cognitive impairment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The underlying brain pathology for most types of dementia develops over decades, prior to the cognitive symptoms emerging [1]. Motor changes related to this neuropathology have shown potential as non-invasive biomarkers [1,2,3]. In 2020, The 5th Canadian Consensus Conference on Diagnosis and Treatment of Dementia (CCCDTD) recommended that assessment of motor function should be included in dementia investigations as there is strong evidence that it can aid detection of cognitive impairment or dementia risk in older adults [2]. Motor biomarkers provide a low cost and accessible method for identifying early-stage cognitive impairment [4] and predict transition from mild cognitive impairment (MCI) to dementia [3, 4]. This may facilitate referral to specialised clinics, early risk modification and recruitment to intervention trials [5,6,7].
Gait has been the most studied motor biomarker with strong evidence showing cognitive impairment associates with impairments in gait [1, 5, 8, 9]. From gait studies, we know that the premotor cortex plays a key role in controlling and coordinating the neural activity in areas of the brain (such as the basal ganglia, brainstem and cerebellum) that are involved in planning and execution of movement [10]. The higher level control of the prefrontal cortex is further implicated when a cognitive task is performed while walking (dual-task). Damage to the prefrontal cortex caused by stroke or neurodegenerative disease is associated with gait impairment such as slowed walking speed and greater step time variability [2, 10,11,12]. Although the neurocognitive mechanisms underpinning the upper limb motor function (ULMF) changes seen with cognitive impairment are not fully understood yet, it would seem likely that they are comparable to those for gait.
Assessment of ULMF may provide additional benefits as many subtle measures of gait are undetectable by clinical observation and require electronic gait analysis systems which limits widespread access [13, 14]. In addition, gait analysis poses challenges for remote assessment and in people who have ambulatory difficulties. In contrast, analysis of ULMF is generally more accessible as it can be assessed using readily available mobile phones and computers and tests can be performed seated.
Emerging evidence shows that a range of ULMFs change in cognitive impairment and may aid discrimination from healthy ageing, but this has been less explored than gait [15,16,17,18,19,20]. It remains unclear what tasks of ULMF to use, how best to measure these and what movement variables associate with cognitive impairment. This hinders integration of ULMF assessments into investigations of cognitive impairment. This review thus aimed to address the question: ‘What methods of assessing ULMF have been used to investigate the association of ULMF with cognitive impairment in adults?’ via four sub questions:
-
1.
What tests (including the task, equipment, protocol, and movement variables) of ULMF have been used to investigate cognitive impairment in adults?
-
2.
What conditions/diseases with resultant cognitive impairment have been studied?
-
3.
What were the major participant recruitment settings?
-
4.
How does ULMF associate with cognitive impairment?
Methods
A scoping review was conducted using JBI methodology for Scoping Reviews and reported following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses—extension for scoping reviews (PRISMA-ScR) guidelines [21, 22]. A protocol was designed to define the questions and clarify methods and reporting (published in Figshare [23]).
We searched PubMed, CINAHL and Web of Science databases for studies published in English up to March 2022. Search terms included synonyms for the three main concepts: 1. Cognitive impairment, 2. Upper limb, 3. Motor function. We included terms describing specialised tasks of the hands and upper limbs such as writing, drawing and grasping. Appendix 1 shows the keywords and Medical Subject Headings (MeSH) terms used for the PubMed search.
Eligibility criteria
All human research studies and systematic reviews examining the association between ULMF and cognitive impairment (caused by any disease/condition) in adults (≥ 18 years) were included. Books, theses, research protocols, and blogs were excluded. Eligible studies required at least one test of cognition and inclusion of participants with cognitive impairment. All tests involving dynamic and volitional functions of the upper limb were eligible, except for grip strength. Evidence from many other studies shows grip strength is associated with cognitive impairment and its measurement is recommended by the fifth Canadian Consensus Conference on Diagnosis and Treatment of Dementia (CCCDTD5) on early non-cognitive markers of dementia [2]. Furthermore, it could be argued that grip strength is not kinematic/dynamic, but rather a kinetic/isometric contraction. Studies of movement analysis in sleep were also excluded as these movements are considered involuntary. All methods of assessing ULMF were eligible.
Data extraction process
Search results were exported to Covidence software, and duplicates removed. Titles and abstracts were screened independently by two reviewers (KR plus JA, MC or KL) against the eligibility criteria. Full texts of selected articles were retrieved and independently screened by two reviewers for inclusion; disagreements were resolved through consensus of the two reviewers and, when required, a third reviewer. As recommended by JBI manual for evidence synthesis [22], a draft data extraction table was developed, piloted, and revised by all authors before it was created in Covidence. This extraction table structured the researchers approach to ensure they extracted the same sets of data from each study and provided a logical summary of results based on the questions of the scoping review [22]. One author (KR) used this to extract data on each study’s design, recruitment setting and characteristics, disease or condition resulting in cognitive impairment, tests (including ULMF task, equipment, protocol, and movement variables) and key findings.
Results
Selection of sources of evidence
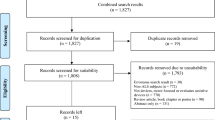
2,219 records were initially identified and, after removing duplicates, 2,169 sources remained. Sixty papers met all selection criteria. Figure 1 shows the flow of information through the steps of this review.
Characteristics of the evidence
Table 1 summarises the characteristics of 60 included articles. The papers were dated from 1995 to February 2022 and comprised 41,800 participants. Most studies (55%) were conducted in the United States, Germany, Japan, China, and the United Kingdom. There were 54 cross-sectional studies (90%), five longitudinal studies (8%) and one systematic review. Five of the cross-sectional studies were sub-studies of longitudinal cohorts.
-
Sub-question 1. What tests of ULMF have been used to investigate cognitive impairment in adults?
Table 2 and Fig. 2 outline the tests of ULMF in the included papers. The narrative synthesis considers the 4 main components of motor function tests: the task, equipment, protocol and movement variables. We recognise there are many ways to group upper limb assessments and that, some may argue that there are better ways to group the tests, especially for those that are new/experimental. In this review, and for ease of classification, we grouped the tasks based on the number of parts of the upper limb that are involved in completing the ULMF assessment.
Tasks involving a part of the upper limb
Finger tapping
Finger tapping (FT) was the most common task with twenty-seven (45%) studies using it as the main task, or one of the tasks. Nearly all studies analysed FT frequency or number of finger-taps. Protocols have evolved with advances in technology, allowing more precise recording with more recent papers including analysis of additional variables such as time between taps and rhythm fluctuations. FT was performed either by tapping a key/lever with the index finger (index-target tapping) or tapping the index finger to the thumb (index-thumb tapping):
Index-target tapping
Twenty-two (37%) studies used this task. The first, published in 1995 [24], used a computer keyboard to count the number of fast-paced finger-taps. Since then, eight studies used fast-paced tapping of a computer key or lever [6, 18, 27, 29, 55, 59, 63, 71]. Movement variables such as number of taps and tapping speed were extracted. Three studies measured self-paced and fast-paced FT for 15 s, using a force transducer [36, 77] or touchpad [50]. Three studies employed ‘cued’ FT protocols requiring tapping to defined frequencies paced by auditory cues [51] or visual cues [46].
More recent studies used infrared-light sensor technologies to measure FT: two used photoelectric sensors arranged around a frame to measure fast-paced FT over 8 s [51] and one required participants to tap their index finger through an infrared light beam for 10 s [51]. Another study [68], used wearable electromechanical sensors on the index finger during 15 s of comfortable pace tapping on the table.
Index-thumb tapping
In total, five (8%) studies used index-thumb tapping; the first, published in 1998, measured the number of taps in 10 s performed by people with Alzheimer’s Disease (AD) [28]. In four recent papers (since 2020) participants were asked to tap at a fast pace for 15 s while wearing reflective markers or magnetic sensors on their thumb and index finger [60, 61, 73, 74]. Extracted movement variables included speed, amplitude, and variabilities in time and speed of a finger-tap cycle [60, 61, 73, 74].
Elbow/forearm movements
The first study of forearm movement in cognitive impairment was in1998 [28] and researchers visually counted the number of correct supination/pronation cycles in 10 s. The next study was 12 years later [33] using an optical shaft encoder to measure the number of fast-paced supination/pronation cycles in 10 s. Since then, two studies used wearable 3-D gyroscopes to measure additional variables such as speed, rise time and speed variability of self-paced and fast-paced elbow flexion movements in 20 s [42, 58] and one used the same device to assess speed and variability of self-paced elbow flexions over 60 s [65].
Tasks involving multiple parts of the upper limb
Purdue Pegboard Test (PPT)
The PPT involves placing a series of pegs into holes on a board as fast as possible and has been utilised in four studies. Three used the 25-hole PPT [19, 32, 71] and one used a 9-hole pegboard [43]. Studies used various protocols: two measured the number of pegs inserted into holes of a 25-hole pegboard in 30 s [19, 71] and two timed participants inserting pegs and removing them from a 25-hole pegboard [32] and a 9-hole pegboard respectively [43].
Writing/drawing tasks
Three studies [30, 49, 76] used writing or drawing tasks to investigate whether kinematic measures (such as speed and smoothness) of digital pen movements on a digitising tablet or paper could differentiate between people with cognitive impairment—including MCI, AD and Vascular dementia (VaD)—and healthy controls (HC). In one study [76], participants drew a clockface on paper using a digitising pen. In another, participants drew concentric circles on a digitising tablet, at a fast pace [30]. In a study [49] which included sentence writing tasks too, participants drew circles on a digitising tablet at a self-selected pace.
Reach/Grasp/Lift tasks
Three studies used tasks involving reach/grasp/lift of an object [34, 55, 75]. In one study [34], participants with MCI and AD lifted objects with different weights and held them for 4 s. In another [55], participants with dementia (various types) lifted an object for 20 s. Both studies analysed steadiness and speed. One study [75] assessed reaching for an object at self-selected and fast paces, under various visual conditions in HC and Parkinson’s Disease (PD), some with cognitive impairment, measuring the time to complete the task.
Gesture imitation
Six studies analysed the ability of participants to imitate bimanual hand gestures after watching a demonstration by the examiner and recorded the number of correct performances and number of errors [35, 38, 40, 48, 54, 69]. In one study, the examiner demonstrated gestures sitting next to the participants to reduce perceptual complexities [48]. In the rest, examiners demonstrated gestures in front of participants [44, 78]. Two studies used the Interlocking Finger Test (ILFT) [44, 78] in which the examiner demonstrates specific shapes with their hands one at a time, and then asks the participants to imitate those gestures, as accurately as possible—for example, interlocking the fingers in a particular manner.
Dragging or tracking tasks
One study used a robotic haptic interface to measure reaction times and mean error of tracking movements in participants with cognitive impairment [47]. The device guided the hand to a target position and gave real-time visual feedback about the hand position as it tracked the target.
Another study used a custom-made electronic board to measure target-tracking reaction times in AD and HC [41]. An electric pen has been used to measure tracking movement variables (such as number of errors and total time) of various upper limb tasks such as hitting targets or guiding the pen through a narrow space [45]. In a recent study, participants used a computer tablet to drag virtual blocks to a target without dropping them in the wrong area [66]; the number of successful and failed attempts within 60 s, and time taken to move a block were analysed.
Tasks resembling day-to-day upper limb functions
One study explored ULMF in people with dementia by measuring the time taken to shelve groceries [67]. Another analysed “hand to mouth” movement variables such as time, speed, and smoothness, in dementia with Lewy bodies [57]. One study used a smartphone app custom-made keyboard to analyse characteristics of virtual key presses (such as keystroke timing) during typing [64].
Three studies analysed participants’ movements as they followed a specific protocol of various functional tasks, and scores were given by observation of their performance. One used parts of the Cambridge Cognitive Examination involving tasks such as putting paper into an envelope, waving goodbye, cutting paper with scissors and brushing teeth [39]. One study used part of the Functional Disability Evaluation Scale-Adult version (FUNDES-Adult), which includes pen-holding, buttoning, and knotting tasks [62]. While another chose the Fugl-Meyer assessment [70].
-
Sub-question 2. What conditions/diseases with resultant cognitive impairment have been studied?
Table 3 summarises the conditions or diseases leading to cognitive impairment included in this review. Of 54 cross-sectional studies, 37 included participants with dementia—26 of them with AD diagnosis—and 22 had a group with MCI. Eight studies investigated participants with PD and cognitive impairment, one recruited participants with Minimal Hepatic Encephalopathy [63]. Two studies recruited participants with cognitive decline related to excessive alcohol consumption [25, 52]. Participants with no known cognitive impairment were recruited in seven studies and after testing for cognitive impairment allocated to groups with and without cognitive impairment. Of five longitudinal studies, four recruited people with no known cognitive impairment at baseline [6, 19, 26, 71] and one recruited participants with MCI [31].
-
Sub-question 3. What were the major recruitment settings?
Forty-two studies recruited participants from clinical settings (e.g., neurological/ memory/cognition clinics, hospitals or rehabilitation centres), fourteen from community settings such as primary health services, day centres and exercise classes [6, 18, 19, 26, 33, 36, 53, 56, 60, 64, 65, 67, 71, 72], and four from research settings (e.g., from other research cohorts) [27, 66, 76, 77].
-
Sub-question 4. How does ULMF associate with cognitive impairment?
Most studies of index-target tapping found significant differences between HC and people with dementia [24, 25, 37, 51, 59, 71, 72] and MCI [6, 17, 26, 36, 46, 51, 56, 63, 77]. Generally, MCI and dementia were both associated with slower, less rhythmic and lower frequency finger-taps. However, two studies found no association between tapping frequency and cognitive impairment [27, 29].
Studies of index-thumb tapping had mixed results too. Three studies reported associations between cognitive impairment and lower frequency, and increased variability, of FT [26, 60, 73]. However, two studies using wearable sensors, found no differences between FT frequency and amplitude in MCI or AD compared to HC [61, 74] although one found FT in MCI was less rhythmic [74]. The systematic review [3] concluded that FT was not associated with incident dementia in people with MCI.
For studies of forearm supination/pronation, slower speed and increased variability were associated with cognitive impairment [28, 33, 53, 55]. The three studies of elbow flexion [42, 58, 65] found no differences under single-task condition between participants with cognitive impairment (MCI and AD) and HC but with dual-task conditions (elbow flexion and a cognitive task), there were significant associations.
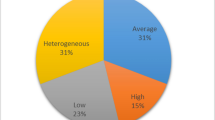
All studies using the PPT found dementia was associated with slower movements compared to MCI [18, 19, 32, 43]. All studies analysing writing/drawing kinematics found increased irregularity of movements, variability in speed and decreased accuracy differentiated HC participants from AD [30, 39, 49, 76] and from MCI [30, 39, 49]. One study [76] that compared measures of clock drawing in AD and VaD found that VaD drew more slowly (having slower speed and taking longer to draw).
Using reach/grasp/lift tasks, one study [34] found no differences between those with MCI or dementia, but another found dementia was associated with more variability than MCI [18]. A study of PD reported that those with dementia [75] had longer reaching reaction times. Studies employing gestures found significant imitation impairment in participants with dementia [35, 40, 48, 54, 69]. The inability in correct imitation of gestures in the ILFT was also correlated with cognitive impairment [44, 78]. Studies using functional tasks found variables of ULMF (such as increased time to complete the task, decreased smoothness, and less accuracy of movements) correlated with cognitive impairment [45, 57, 62, 67, 70]. Studies measuring tracking abilities found that participants with MCI and AD had more errors and slower reaction times than HC [41, 47]. Using digital tests, two studies [64, 66] showed differences between MCI and HC: one [66] reported reduced speed of dragging virtual blocks and another [64] identified more errors in virtual keyboard presses.
Among the longitudinal studies, two [6, 26] found that slower baseline FT in HC was associated with cognitive impairment at follow up, but another [31] found no such association. One study [19] found lower PPT scores were associated with higher risk of developing dementia at follow up. A study [71], using both PPT and index-target tapping concluded that lower performance scores of both tests were associated with risk of MCI and dementia at follow up.
Fifteen studies additionally investigated how ULMF associates with individual cognitive domains. Eight studies, using index-target tapping, found significant associations between FT variables (tapping cycle time, tapping rate and time variability) and memory (working and episodic), verbal fluency and executive function [18, 24, 27, 36, 37, 46, 59, 63]. One study found slowed index-thumb tapping speed associated with verbal fluency and executive function but not with delayed memory [25].
One study of elbow flexion found associations between speed and rhythm fluctuations with executive function [42]. Lower PPT scores correlated with impaired attention, visuo-spatial and executive function [18]. Using gesture tasks, studies found imitation accuracy associated with verbal fluency, attention [44, 69, 78] and executive function [44] but another did not [78]. Tracking ability was correlated with memory and visuospatial domains [47] and functional tasks were associated with attention, visuospatial and executive function [70].
Discussion
Sixty studies published between 1995 and 2022, and comprising 41,800 participants, met the criteria to inform this review. To our knowledge, this is the first review investigating the association of ULMF with cognitive impairment. The studies used a diverse range of ULMF tasks from a simple movement, such as FT or elbow flexion, to more complex movements such as writing/drawing. Studies also used a range of protocols (self-paced, fast-paced, dual-task etc.), test durations (ranging from 8 to 60 s), and equipment. With technology advancements over time, the precision of data collection equipment has progressed, so analyses have evolved from counting the number of repetitions to detailed quantification of rhythm, amplitude and speed. The recruitment settings were mostly clinical, and the conditions included were predominantly AD, MCI and PD. Many studies found that, compared to age-matched older adults, people with cognitive impairment had slower speeds, longer reaction times and more errors and variability in their ULMF performance. However, these associations were not universal, especially among the FT studies. FT (index-target or index-thumb tapping) was the most common ULMF test, but protocols, durations and equipment varied significantly among these studies which may be the reason why FT studies had mixed results. Studies of elbow flexion, writing/drawing tasks and the PPT had no conflicting results, although there were fewer studies compared to the large number that assessed FT.
With no limitation in dates, this review provides a broad view of how ULMF assessments in the context of cognitive impairment have evolved since conception about 25 years ago. We systematically searched published literature using established guidelines and published our protocol in advance in an open access repository (Figshare). It is important to acknowledge that we excluded studies measuring hands/arms strength, such as grip strength, and excluded studies with only healthy participants which may have excluded some of the ULMF tests. We also acknowledge that it is possible relevant studies without linked keywords may have inadvertently been excluded.
This review highlights that ULMF assessments hold potential to be used in cognitive impairment investigations as many (but not all) of the studies found associations between ULMF and cognitive impairment. However, it also revealed a major gap in the current literature and that is the lack of consistency between the experimental methods used to assess ULMF. It remains unclear whether one specific type of test is superior to others, and it remains unclear how many repetitions of a task, or what test duration, should be used to balance sensitivity with potential effects of fatigue. The review demonstrated that, in a similar way to how gait analysis now has some recommended standard protocols [14], there remains a need to also standardise ULMF assessment methods—in terms of test durations and protocols (fast-paced vs. self-selected pace); this would substantially aid comparison of studies and clarify which tests are most discriminatory.
Most ULMF studies used 10 to 15 s as the test duration which seems to be a pragmatic balance between capturing enough data for robust analysis of movements whilst minimising the effects of fatigue. Several studies measured ULMF performance at various paces (self-selected pace vs fast pace) or under different conditions (single task and dual task). These approaches, as well as analysing multiple component measures of movements (such as frequency, speed, amplitude and rhythm) appeared to be more sensitive to cognitive impairment than testing just one movement under one condition and/or few movement components. Future research should consider analysing frequency, speed, and variability of ULMF as the core measures as these have repeatedly been shown to associate with cognitive impairment.
As most studies of ULMF have compared healthy controls to just one group who had clinically-manifested cognitive impairment, especially Alzheimer’s Disease (AD) dementia, it remains unclear how ULMF changes across the dementia continuum. Furthermore, there have been relatively few studies of other types of dementia. Future research should therefore aim to recruit participants with earlier stages of dementia pathology, such as subjective cognitive impairment, MCI, and early-stage dementia to provide richer insights into the changes related to disease progression. It was noteworthy that most studies classified participants according to screening tool cut-off scores rather than a more comprehensive cognitive assessment using established diagnosis criteria; we would recommend that future researchers aim to ascertain a more rigorous evaluation of the various domains of cognitive impairment as this would allow a more granular comparison with ULMF features and the opportunity to explore whether certain underlying pathologies have specific ULMF motor signatures.
For ULMF assessment to be included in CCCDTD as a recommended motor function assessment in dementia investigations, it is necessary to know how best to assess ULMF that is significantly associated with cognitive impairment and dementia. We are still learning about the association between ULMF and cognitive impairment and methods of testing ULMF are yet to be fully explored. This review shows that despite some inconclusive results, there is emerging evidence to support including ULMF in cognitive impairment investigations.
Conclusion
In this scoping review, we summarised the current available evidence on the association of ULMF and cognitive impairment and also the tests, protocols, recruitment settings and conditions used to assess this association. Of the identified methods of ULMF assessment, FT was the most commonly used test followed by functional tasks of upper limb, PPT and elbow/forearm movement. Despite some mixed results, the ULMF movement variables were generally associated with cognitive impairment and could aid in distinguishing cognitive impairment from healthy ageing.
References
Kivimaki Mika, Singh-Manoux A. Prevention of dementia by targeting risk factors. Lancet. 2018;391(10130):1574–5. https://doi.org/10.1016/S0140-6736(18)30578-6.
Montero-Odasso M, Pieruccini-Faria F, Ismail Z, et al. CCCDTD5 recommendations on early non cognitive markers of dementia: A Canadian consensus. Alzheimers Dement (N Y). 2020;6(1):e12068. https://doi.org/10.1002/trc2.12068.
Kueper JK, Speechley M, Lingum NR, et al. Motor function and incident dementia: a systematic review and meta-analysis. Age Ageing. 2017;46(5):729–38. https://doi.org/10.1093/ageing/afx084.
Muller K, Frohlich S, Germano AMC, et al. Sensor-based systems for early detection of dementia (SENDA): a study protocol for a prospective cohort sequential study. BMC Neurol. 2020;20(1):84. https://doi.org/10.1186/s12883-020-01666-8.
Mielke MM, Roberts RO, Savica R, et al. Assessing the temporal relationship between cognition and gait: slow gait predicts cognitive decline in the Mayo Clinic Study of Aging. J Gerontol A Biol Sci Med Sci. 2013;68(8):929–37. https://doi.org/10.1093/gerona/gls256.
Buracchio TDH, Diane Howieson D, Wasserman D, Kaye J. The trajectory of gait speed preceding mild cognitive impairment. Arch Neurol. 2010;67(8):980–6. https://doi.org/10.1001/archneurol.2010.159.
Ahmed RM, Paterson RW, Warren JD, et al. Biomarkers in dementia: clinical utility and new directions. J Neurol Neurosurg Psychiatry. 2014;85(12):1426–34. https://doi.org/10.1136/jnnp-2014-307662.
Arvanitakis Z, Shah RC, Bennett DA. Diagnosis and management of dementia: review. JAMA. 2019;322(16):1589–99. https://doi.org/10.1001/jama.2019.4782.
Belleville S, Fouquet C, Hudon C, et al. Neuropsychological measures that predict progression from mild cognitive impairment to Alzheimer’s type dementia in older adults: a systematic review and meta-analysis. Neuropsychol Rev. 2017;27(4):328–53. https://doi.org/10.1007/s11065-017-9361-5.
Cohen JA, Verghese J. Chapter 22 - Gait and dementia. In: Dekosky ST, Asthana SS, editors. Handbook of Clinical Neurology. Elsevier; 2019. p. 419–27.
Allali G, Annweiler C, Blumen HM, et al. Gait phenotype from mild cognitive impairment to moderate dementia: results from the GOOD initiative. Eur J Neurol. 2016;23(3):527–41. https://doi.org/10.1111/ene.12882.
Verghese J, Robbins M, Holtzer R, et al. Gait dysfunction in mild cognitive impairment syndromes. J Am Geriatr Soc. 2008;56(7):1244–51. https://doi.org/10.1111/j.1532-5415.2008.01758.x.
Bahureksa L, Najafi B, Saleh A, et al. The impact of mild cognitive impairment on gait and balance: a systematic review and meta-analysis of studies using instrumented assessment. Gerontology. 2017;63(1):67–83. https://doi.org/10.1159/000445831.
Beauchet O, Allali G, Sekhon H, et al. Guidelines for assessment of gait and reference values for spatiotemporal gait parameters in older adults: the Biomathics and Canadian gait consortiums initiative. Front Hum Neurosci. 2017;11:353. https://doi.org/10.3389/fnhum.2017.00353.
Robert Briggs SPK, O’Neill D. Drug treatments in Alzheimer’s disease. Clin Med. 2016;16(3):247–53. https://doi.org/10.7861/clinmedicine.16-3-247.
Scherder E, Dekker W, Eggermont L. Higher-level hand motor function in aging and (preclinical) dementia: its relationship with (instrumental) activities of daily life–a mini-review. Gerontology. 2008;54(6):333–41. https://doi.org/10.1159/000168203.
Carment L, Abdellatif A, Lafuente-Lafuente C, et al. Manual dexterity and aging: A pilot study disentangling sensorimotor from cognitive decline. Front Neurol. 2018;9:910. https://doi.org/10.3389/fneur.2018.00910.
Hesseberg K, Tangen GG, Pripp AH, et al. Associations between cognition and hand function in older people diagnosed with mild cognitive impairment or dementia. Dement Geriatr Cogn Dis Extra. 2020;10(3):195–204. https://doi.org/10.1159/000510382.
Darweesh SK, Wolters FJ, Hofman A, et al. Simple test of manual dexterity can help to identify persons at high risk for neurodegenerative diseases in the community. J Gerontol A Biol Sci Med Sci. 2017;72(1):75–81. https://doi.org/10.1093/gerona/glw122.
Williams S, Zhao Z, Hafeez A, et al. The discerning eye of computer vision: Can it measure Parkinson’s finger tap bradykinesia? J Neurol Sci. 2020;416:117003. https://doi.org/10.1016/j.jns.2020.117003.
PRISMA. PRISMA for Scoping Reviews. https://prisma-statement.org/Extensions/ScopingReviews. Accessed date 2018.
Peters MDJ GC, McInerney P, Munn Z, Tricco AC, Khalil, H. JBI Manual for Evidence Synthesis. https://synthesismanual.jbi.global. Accessed Date 2020.
Rudd KD, Lawler K, Callisaya ML, Alty J. Methods used to investigate the association of cognitive impairment with upper limb motor function in adults: a scoping review protocol. Figshare; 2022. https://doi.org/10.6084/m9.figshare.19406981.v1
Ott BR, Ellias SA, Lannon MC. Quantitative assessment of movement in Alzheimer’s disease. J Geriatr Psychiatry Neurol. 1995;8(1):71–5.
Welch LW, Cunningham AT, Eckardt MJ, et al. Fine motor speed deficits in alcoholic Korsakoff’s syndrome. Alcoholism: Clin Exp Res. 1997;21(1):134–8.
Camicioli R, Howieson D, Oken B, et al. Motor slowing precedes cognitive impairment in the oldest old. Neurology. 1998;50(5):1496–8.
Goldman WP, Baty JD, Buckles VD, et al. Cognitive and motor functioning in Parkinson disease: subjects with and without questionable dementia. Arch Neurol. 1998;55(5):674–80. https://doi.org/10.1001/archneur.55.5.674.
Willis L, Behrens M, Mack W, et al. Ideomotor apraxia in early Alzheimer’s disease: time and accuracy measures. Brain Cogn. 1998;38(2):220–33. https://doi.org/10.1006/brcg.1998.1029.
Goldman W, Baty J, Buckles V, et al. Motor dysfunction in mildly demented AD individuals without extrapyramidal signs. Neurology. 1999;53(5):956–956.
Schröter A, Mergl R, Bürger K, et al. Kinematic analysis of handwriting movements in patients with Alzheimer’s disease, mild cognitive impairment, depression and healthy subjects. Dement Geriatr Cogn Disord. 2003;15(3):132–42.
Amieva H, Letenneur L, Dartigues JF, et al. Annual rate and predictors of conversion to dementia in subjects presenting mild cognitive impairment criteria defined according to a population-based study. Dement Geriatr Cogn Disord. 2004;18(1):87–93. https://doi.org/10.1159/000077815.
Muhlack S, Przuntek H, Müller T. Transdermal Rivastigmine treatment does not worsen impaired performance of complex motions in patients with Alzheimer’s disease. Pharmacopsychiatry. 2006;39(1):16–9. https://doi.org/10.1055/s-2006-931473.
Bramell-Risberg E, Jarnlo G-B, Elmståhl S. Slowing of alternating forearm movements is associated with cognitive impairment in community-dwelling older people. Dement Geriatr Cogn Disord. 2010;29(5):457–66.
Ameli M, Kemper F, Sarfeld A-S, et al. Arbitrary visuo-motor mapping during object manipulation in mild cognitive impairment and Alzheimer’s disease: A pilot study. Clin Neurol Neurosurg. 2011;113(6):453–8. https://doi.org/10.1016/j.clineuro.2011.01.011.
Rousseaux M, Rénier J, Anicet L, et al. Gesture comprehension, knowledge and production in Alzheimer’s disease. Eur J Neurol. 2012;19(7):1037–44. https://doi.org/10.1111/j.1468-1331.2012.03674.x.
Rabinowitz I, Lavner Y. Association between finger tapping, attention, memory, and cognitive diagnosis in elderly patients. Percept Mot Skills. 2014;119(1):259–78. https://doi.org/10.2466/10.22.PMS.119c12z3.
Henley SMD, Downey LE, Nicholas JM, et al. Degradation of cognitive timing mechanisms in behavioural variant frontotemporal dementia. Neuropsychologia. 2014;65:88–101. https://doi.org/10.1016/j.neuropsychologia.2014.10.009.
Johnen A, Tokaj A, Kirschner A, Wiendl H, Lueg G, Duning T, Lohmann H. Apraxia profile differentiates behavioural variant frontotemporal from Alzheimer's dementia in mild disease stages. J Neurol Neurosurg Psychiatry. 2015;86(7):809–15. https://doi.org/10.1136/jnnp-2014-308773.
Ward M, Cecato JF, Aprahamian I, et al. Assessment for apraxia in Mild Cognitive Impairment and Alzheimer’s disease. Dement Neuropsychol. 2015;9:71–5.
Nagahama Y, Okina T, Suzuki N. Impaired imitation of gestures in mild dementia: comparison of dementia with Lewy bodies, Alzheimer’s disease and vascular dementia. J Neurol Neurosurg Psychiatry. 2015;86(11):1248. https://doi.org/10.1136/jnnp-2014-309436.
Lin YC, Hsu WC, Wu CK, et al. Comparison of motor performance of upper and lower extremities in dual-task tests in patients with mild Alzheimer’s dementia. Aging Clin Exp Res. 2016;28(3):491–6. https://doi.org/10.1007/s40520-015-0441-1.
Toosizadeh N, Najafi B, Reiman EM, et al. Upper-extremity dual-task function: an innovative method to assess cognitive impairment in older adults. Front Aging Neurosci. 2016;8:167. https://doi.org/10.3389/fnagi.2016.00167.
Fritz NE, Kegelmeyer DA, Kloos AD, et al. Motor performance differentiates individuals with Lewy body dementia, Parkinson’s and Alzheimer’s disease. Gait Posture. 2016;50:1–7. https://doi.org/10.1016/j.gaitpost.2016.08.009.
Souza CP, Oliveira GN, Foss MP, et al. The interlocking finger test in patients with Parkinson’s disease and healthy subjects. J Clin Neurosci. 2016;29:145–8. https://doi.org/10.1016/j.jocn.2015.09.026.
Dahdal P, Meyer A, Chaturvedi M, et al. Fine motor function skills in patients with Parkinson disease with and without mild cognitive impairment. Dement Geriatr Cogn Disord. 2016;42(3–4):127–34. https://doi.org/10.1159/000448751.
Kay CD, Seidenberg M, Durgerian S, et al. Motor timing intraindividual variability in amnestic mild cognitive impairment and cognitively intact elders at genetic risk for Alzheimer’s disease. J Clin Exp Neuropsychol. 2017;39(9):866–75. https://doi.org/10.1080/13803395.2016.1273321.
Bartoli E, Caso F, Magnani G, et al. Low-cost robotic assessment of Visuo-motor deficits in Alzheimer’s disease. IEEE Trans Neural Syst Rehabil Eng. 2017;25(7):852–60. https://doi.org/10.1109/TNSRE.2017.2708715.
Sanin GN, Benke T. Bimanual gesture imitation in Alzheimer’s disease. J Alzheimers Dis. 2017;57(1):53–9. https://doi.org/10.3233/jad-160680.
Garre-Olmo J, Faúndez-Zanuy M, López-de-Ipiña K, et al. Kinematic and pressure features of handwriting and drawing: preliminary results between patients with mild cognitive impairment, Alzheimer disease and healthy controls. Curr Alzheimer Res. 2017;14(9):960–8. https://doi.org/10.2174/1567205014666170309120708.
Suzumura S, Osawa A, Maeda N, et al. Differences among patients with Alzheimer’s disease, older adults with mild cognitive impairment and healthy older adults in finger dexterity. Geriatr Gerontol Int. 2018;18(6):907–14. https://doi.org/10.1111/ggi.13277.
Roalf DR, Rupert P, Mechanic-Hamilton D, et al. Quantitative assessment of finger tapping characteristics in mild cognitive impairment, Alzheimer’s disease, and Parkinson’s disease. J Neurol. 2018;265(6):1365–75. https://doi.org/10.1007/s00415-018-8841-8.
Gupta A, Murthy P, Rao S. Brief screening for cognitive impairment in addictive disorders. Indian J Psychiatry. 2018;60(Suppl 4).
Rycroft SS, Quach LT, Ward RE, et al. The relationship between cognitive impairment and upper extremity function in older primary care patients. J Gerontol A Biol Sci Med Sci. 2018;74(4):568–74. https://doi.org/10.1093/gerona/gly246.
Gulde P, Leippold K, Armstrong A, Kohl S, Grimmer T, Diehl-Schmid J, Hermsdörfer J. An explorative note on apraxia tests. Front Neurol. 2018;8(9). https://doi.org/10.3389/fneur.2018.00660.
Jeppesen Kragh F, Bruun M, Budtz-Jørgensen E, et al. Quantitative measurements of motor function in Alzheimer’s disease, frontotemporal dementia, and dementia with Lewy bodies: a proof-of-concept study. Dement Geriatr Cogn Disord. 2018;46(3–4):168–79. https://doi.org/10.1159/000492860.
Zhang L, Zhao Y, Shen C, Lei L, Dong J, Zou D, Zou J, Wang M. Can long-term regular practice of physical exercises including taichi improve finger tapping of patients presenting with mild cognitive impairment? Front Physiol. 2018;28(9). https://doi.org/10.3389/fphys.2018.01396.
Fadda L, Corona F, Floris G, et al. Upper limb movements in dementia with Lewy body: a quantitative analysis. Exp Brain Res. 2019;237(8):2105–10. https://doi.org/10.1007/s00221-019-05575-2.
Toosizadeh N, Ehsani H, Wendel C, et al. Screening older adults for amnestic mild cognitive impairment and early-stage Alzheimer’s disease using upper-extremity dual-tasking. Sci Rep. 2019;9(1):10911. https://doi.org/10.1038/s41598-019-46925-y.
Mollica MA, Tort-Merino A, Navarra J, et al. Early detection of subtle motor dysfunction in cognitively normal subjects with amyloid-beta positivity. Cortex. 2019;121:117–24. https://doi.org/10.1016/j.cortex.2019.07.021.
Tomita Y, Tanaka S, Takahashi S, et al. Detecting cognitive decline in community-dwelling older adults using simple cognitive and motor performance tests. Geriatr Gerontol Int. 2020;20(3):212–7. https://doi.org/10.1111/ggi.13863.
Bologna M, Guerra A, Colella D, et al. Bradykinesia in Alzheimer’s disease and its neurophysiological substrates. Clin Neurophysiol. 2020;131(4):850–8. https://doi.org/10.1016/j.clinph.2019.12.413.
Liou W-C, Chan L, Hong C-T, et al. Hand fine motor skill disability correlates with dementia severity. Arch Gerontol Geriatr. 2020;90:104168. https://doi.org/10.1016/j.archger.2020.104168.
San Martín-Valenzuela C, Borras-Barrachina A, Gallego JJ, Urios A, Mestre-Salvador V, Correa-Ghisays P, Ballester MP, Escudero-García D, Tosca J, Montón C, Ríos MP. Motor and cognitive performance in patients with liver cirrhosis with minimal hepatic encephalopathy. J Clin Med. 2020;9(7). https://doi.org/10.3390/2Fjcm9072154.
Ntracha A, Iakovakis D, Hadjidimitriou S, et al. Detection of mild cognitive impairment through natural language and touchscreen typing processing. Front Digit Health. 2020;2:567158. https://doi.org/10.3389/fdgth.2020.567158.
Ehsani H, Parvaneh S, Mohler J, et al. Can motor function uncertainty and local instability within upper-extremity dual-tasking predict amnestic mild cognitive impairment and early-stage Alzheimer’s disease? Comput Biol Med. 2020;120:103705. https://doi.org/10.1016/j.compbiomed.2020.103705.
Zhang Y, Chen Y, Yu H, et al. What can “drag & drop” tell? Detecting mild cognitive impairment by hand motor function assessment under dual-task paradigm. Int J Hum Comput Stud. 2021;145:102547. https://doi.org/10.1016/j.ijhcs.2020.102547.
Paixão C, Tavares A, Marques A. Respiratory function and upper extremity functional activity performance in people with dementia: a shout for attention. J Aging Phys Act. 2021;29(1):89–98. https://doi.org/10.1123/japa.2020-0005.
Mancioppi G, Fiorini L, Rovini E, et al. Innovative motor and cognitive dual-task approaches combining upper and lower limbs may improve dementia early detection. Sci Rep. 2021;11(1):7449. https://doi.org/10.1038/s41598-021-86579-3.
Nagahama Y, Okina T, Suzuki N. Neuropsychological basis of impaired gesture imitations in patients with Alzheimer's disease and dementia with Lewy bodies. Int J Geriatr Psychiatry. 2021;37(1). https://doi.org/10.1002/gps.5622.
Uwa-Agbonikhena IF, Gryb VA, Gerasymchuk VR. Associations between the upper extremity function and cognition in post-stroke patients. Wiad Lek. 2021;74(8):1917–20.
Beeri MS, Leurgans SE, Bennett DA, et al. Diverse motor performances are related to incident cognitive impairment in community-dwelling older adults. Front Aging Neurosci. 2021;13. https://doi.org/10.3389/fnagi.2021.717139.
Zhao L, Liu G, Zhang L, et al. Long-term physical exercise improves finger tapping of patients with Alzheimer’s disease. Curr Alzheimer Res. 2021;18(14):1077–86. https://doi.org/10.2174/1567205018666211215150157.
Suzumura S, Kanada Y, Osawa A, et al. Assessment of finger motor function that reflects the severity of cognitive function. Fujita Med J. 2021;7(4):122–9. https://doi.org/10.20407/fmj.2020-013.
Colella D, Guerra A, Paparella G, et al. Motor dysfunction in mild cognitive impairment as tested by kinematic analysis and transcranial magnetic stimulation. Clin Neurophysiol. 2021;132(2):315–22. https://doi.org/10.1016/j.clinph.2020.10.028.
Cosgrove J, Hinder MR, St George RJ, et al. Significant cognitive decline in Parkinson’s disease exacerbates the reliance on visual feedback during upper limb reaches. Neuropsychologia. 2021;157:107885. https://doi.org/10.1016/j.neuropsychologia.2021.107885.
Davoudi A, Dion C, Amini S, et al. Classifying non-dementia and Alzheimer’s disease/vascular dementia patients using kinematic, time-based, and visuospatial parameters: the digital clock drawing test. J Alzheimers Dis. 2021;82(1):47–57. https://doi.org/10.3233/jad-201129.
Kutz DF, Fröhlich S, Rudisch J, et al. Finger tapping as a biomarker to classify cognitive status in 80+-Year-Olds. J Personalized Med. 2022;12(2):286.
Schmidt N, Strohmaier T, Witt K. A modified version of the interlocking finger test as a bedside screening test for visuospatial deficits and dementia in Parkinson’s disease. Brain Behav. 2022;12(4):e2516. https://doi.org/10.1002/brb3.2516.
Acknowledgements
The Wicking Dementia Research and Education Centre is supported by J.O. and J.R. Wicking Trust (Equity Trustees) and the University of Tasmania.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. KR is supported by a Research Training Program living allowance and University of Tasmania tuition fee scholarship. The Wicking Dementia Research and Education Centre is supported by the J.O. and J.R. Wicking Trust (Equity Trustees). The funding bodies have no direct role in the study design, data collection, analysis, and interpretation or manuscript preparation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no conflict of interest.
Standard of reporting: This review was conducted in line with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses -extension for scoping reviews (PRISMA-ScR) guidelines.
Disclosure
Authors have no conflict of interests to disclose.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix 1
Appendix 1
Key search terms for PubMed Search performed on 14/03/2022
Search | Query |
|---|---|
#4 | ((#1) AND (#2)) AND (#3) |
#3 | (Motor impairment[Title/Abstract]) OR (motor function[Title/Abstract])) OR (movement[Title/Abstract])) OR (motor test[Title/Abstract])) OR (motor dysfunction[Title/Abstract])) OR (Motor test[Title/Abstract]))) OR (fine motor test[Title/Abstract])) OR (motor performance[Title/Abstract])) OR (movement test[Title/Abstract])) OR (gross motor function[Title/Abstract])) OR (gross motor impairment[Title/Abstract])) OR (motor dysfunction[Title/Abstract])) OR (motor decline[Title/Abstract])) OR (finger tapping[Title/Abstract])) OR (apraxia[Title/Abstract])) OR (dyspraxia[Title/Abstract])) OR (dexterity[Title/Abstract])) OR (grasp*[Title/Abstract])) OR (grip*[Title/Abstract])) OR (tap*[Title/Abstract])) OR (hand tapping[Title/Abstract])) OR (keyboard tapping[Title/Abstract])) OR (holding[Title/Abstract])) OR (draw*[Title/Abstract])) OR (writ*[Title/Abstract])) OR (Purdue pegboard test[Title/Abstract]) |
#2 | Hand*[Title/Abstract] OR Forearm*[Title/Abstract] OR Finger*[Title/Abstract] OR Upper limb*[Title/Abstract] OR "Upper limb"[Title/Abstract] |
#1 | Dementia*[Title/Abstract] OR "Cognitive impairment"[Title/Abstract] OR "Cognitive decline"[Title/Abstract] OR "Alzheimer’s Disease"[Title/Abstract] OR "Mild Cognitive Impairment"[Title/Abstract] OR Cognition*[Title/Abstract] OR "Cognitive domains"[Title/Abstract] |
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Rudd, K.D., Lawler, K., Callisaya, M.L. et al. Investigating the associations between upper limb motor function and cognitive impairment: a scoping review. GeroScience 45, 3449–3473 (2023). https://doi.org/10.1007/s11357-023-00844-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11357-023-00844-z