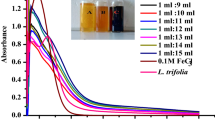
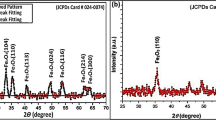
Abstract
The synthesis of iron-based nanoparticles (Fe NPs) using traditional preparation methods suffered from the disadvantages of high cost, environmental harm, and easy agglomeration. In this study, a novel eco-friendly method was proposed for the synthesis of iron nanomaterials (ML-Fe NPs): using antioxidant components extracted from mulberry leaf to reduce divalent iron (II). The preparation conditions of ML-Fe NPs were optimized by orthogonal tests. The prepared ML-Fe NPs exhibited an amorphous core–shell structure, displaying excellent dispersion and stability. During the synthesis process of ML-Fe NPs, the polyphenol molecules in mulberry leaf extract played a dominant role. A possible synthetic mechanism involving complexation, reduction, and encapsulation was proposed. Furthermore, the ML-Fe NPs were utilized to construct an ML-Fe NPs/peroxymonosulfate catalytic system for the degradation of Rhodamine B dye wastewater. The ML-Fe NPs demonstrated remarkable catalytic potential, achieving a 99% degradation efficiency for Rhodamine B within a span of 40 min.









Similar content being viewed by others
Data Availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
References
Agnihotri A, Gupta P, Dwivedi A, Seth CS (2018) Counteractive mechanism (s) of salicylic acid in response to lead toxicity in Brassica juncea (L.) Czern. cv. Varuna Planta 248:49–68. https://doi.org/10.1007/s00425-018-2867-0
Ballesteros JI, Caleja-Ballesteros HJR, Villena MC (2021) Digital image-based method for iron detection using green tea (Camellia sinensis) extract as natural colorimetric reagent. Microchem J 160:105652. https://doi.org/10.1016/j.microc.2020.105652
Bao T, Xu Y, Gowd V et al (2016) Systematic study on phytochemicals and antioxidant activity of some new and common mulberry cultivars in China. J Funct Foods 25:537–547. https://doi.org/10.1016/j.jff.2016.07.001
Chen L, Ni R, Yuan T et al (2020) Effects of green synthesis, magnetization, and regeneration on ciprofloxacin removal by bimetallic nZVI/Cu composites and insights of degradation mechanism. J Hazard Mater 382:121008. https://doi.org/10.1016/j.jhazmat.2019.121008
Chen W, Wu J, Weng X et al (2022) One-step green synthesis of hybrid Fe-Mn nanoparticles: Methodology, characterization and mechanism. J Clean Prod 363:132406. https://doi.org/10.1016/j.jclepro.2022.132406
Cheng H, Sun Y, Wang X et al (2020) Hierarchical porous carbon fabricated from cellulose-degrading fungus modified rice husks: ultrahigh surface area and impressive improvement in toluene adsorption. J Hazard Mater 392:122298. https://doi.org/10.1016/j.jhazmat.2020.122298
Davarazar M, Kamali M, Venâncio C et al (2023) Activation of persulfate using copper oxide nanoparticles for the degradation of Rhodamine B containing effluents: degradation efficiency and ecotoxicological studies. Chem Eng J 453:139799. https://doi.org/10.1016/j.cej.2022.139799
Du R, Guo W, Shen Y et al (2023) In situ assay of the reducing sugars in hydrophilic natural deep eutectic solvents by a modified DNS method. J Mol Liq 385:122286. https://doi.org/10.1016/j.molliq.2023.122286
Fazlzadeh M, Rahmani K, Zarei A et al (2017) A novel green synthesis of zero valent iron nanoparticles (NZVI) using three plant extracts and their efficient application for removal of Cr(VI) from aqueous solutions. Adv Powder Technol 28:122–130. https://doi.org/10.1016/j.apt.2016.09.003
Garg A, Chopra L (2022) Dye waste: a significant environmental hazard. Mater Today: Proc 48:1310–1315. https://doi.org/10.1016/j.matpr.2021.09.003
Guo Y, Sun Q, Wu F-G et al (2021) Polyphenol-containing nanoparticles: synthesis, properties, and therapeutic delivery. Adv Mater 33:2007356. https://doi.org/10.1002/adma.202007356
Hou X, Zhan G, Huang X et al (2020) Persulfate activation induced by ascorbic acid for efficient organic pollutants oxidation. Chem Eng J 382:122355. https://doi.org/10.1016/j.cej.2019.122355
Hu Y, Yang L, Liang Z et al (2023) Comparative analysis of flavonoids extracted from Dendrobium chrysotoxum flowers by supercritical fluid extraction and ultrasonic cold extraction. Sustain Chem Pharm 36:101267. https://doi.org/10.1016/j.scp.2023.101267
Jain R, Mendiratta S, Kumar L, Srivastava A (2021) Green synthesis of iron nanoparticles using Artocarpus heterophyllus peel extract and their application as a heterogeneous Fenton-like catalyst for the degradation of Fuchsin Basic dye. Curr Res Green Sustain Chem 4:100086. https://doi.org/10.1016/j.crgsc.2021.100086
Jawed A, Golder AK, Pandey LM (2023) Synthesis of iron oxide nanoparticles mediated by Camellia sinensis var. Assamica for Cr(VI) adsorption and detoxification. Bioresourc Technol 376:128816. https://doi.org/10.1016/j.biortech.2023.128816
Jin X, Liu Y, Tan J et al (2018) Removal of Cr(VI) from aqueous solutions via reduction and absorption by green synthesized iron nanoparticles. J Clean Prod 176:929–936. https://doi.org/10.1016/j.jclepro.2017.12.026
Khatami M, Alijani HQ, Fakheri B et al (2019) Super-paramagnetic iron oxide nanoparticles (SPIONs): greener synthesis using Stevia plant and evaluation of its antioxidant properties. J Clean Prod 208:1171–1177. https://doi.org/10.1016/j.jclepro.2018.10.182
Kim E-J, Kim J-H, Azad A-M, Chang Y-S (2011) Facile synthesis and characterization of Fe/FeS nanoparticles for environmental applications. ACS Appl Mater Interfaces 3:1457–1462. https://doi.org/10.1021/am200016v
Kim D-Y, Patel SKS, Rasool K et al (2024) Bioinspired silver nanoparticle-based nanocomposites for effective control of plant pathogens: a review. Sci Total Environ 908:168318. https://doi.org/10.1016/j.scitotenv.2023.168318
Kuang Y, Wang Q, Chen Z et al (2013) Heterogeneous Fenton-like oxidation of monochlorobenzene using green synthesis of iron nanoparticles. J Colloid Interface Sci 410:67–73. https://doi.org/10.1016/j.jcis.2013.08.020
Kumar D, Dhankher OP, Tripathi RD, Seth CS (2023) Titanium dioxide nanoparticles potentially regulate the mechanism(s) for photosynthetic attributes, genotoxicity, antioxidants defense machinery, and phytochelatins synthesis in relation to hexavalent chromium toxicity in Helianthus annuus L. J Hazard Mater 454:131418. https://doi.org/10.1016/j.jhazmat.2023.131418
Kumar D, Singh R, Upadhyay SK et al (2024) Review on interactions between nanomaterials and phytohormones: novel perspectives and opportunities for mitigating environmental challenges. Plant Sci 340:111964. https://doi.org/10.1016/j.plantsci.2023.111964
Kumari T, Phogat D, Shukla V (2023) Exploring the multipotentiality of plant extracts for the green synthesis of iron nanoparticles: a study of adsorption capacity and dye degradation efficiency. Environ Res 229:116025. https://doi.org/10.1016/j.envres.2023.116025
Kumkoon T, Srisaisap M, Boonserm P (2023) Biosynthesized silver nanoparticles using Morus alba (white mulberry) leaf extract as potential antibacterial and anticancer agents. Molecules 28:1213. https://doi.org/10.3390/molecules28031213
Levy L, Radian A (2023) Catechol-iron-clay surface complexation enriches radical formation and efficiency in heterogeneous Fenton reactions. Appl Clay Sci 232:106802. https://doi.org/10.1016/j.clay.2022.106802
Li P, Wang Y, Ma R, Zhang X (2005) Separation of tea polyphenol from green tea Leaves by a combined CATUFM-adsorption resin process. J Food Eng 67:253–260. https://doi.org/10.1016/j.jfoodeng.2004.04.009
Li Z, Zhang X, Li G et al (2023) Hollow Co/CoO/Carbon nanofibers promoted PMS decomposition for the degradation of Rhodamine B. J Mater Sci Technol 157:120–129. https://doi.org/10.1016/j.jmst.2022.10.079
Lin Z, Weng X, Owens G, Chen Z (2020) Simultaneous removal of Pb(II) and rifampicin from wastewater by iron nanoparticles synthesized by a tea extract. J Clean Prod 242:118476. https://doi.org/10.1016/j.jclepro.2019.118476
Liu Y, Jin X, Chen Z (2018) The formation of iron nanoparticles by Eucalyptus leaf extract and used to remove Cr(VI). Sci Total Environ 627:470–479. https://doi.org/10.1016/j.scitotenv.2018.01.241
Lu H, Zhu F, Xu H et al (2023) Insights of mechanism into enhanced removal of Cr(VI) by Ginkgo biloba leaves synthesized bimetallic nano-zero-valent iron/copper. Colloids Surf, A 675:132094. https://doi.org/10.1016/j.colsurfa.2023.132094
Luo F, Yang D, Chen Z et al (2016) Characterization of bimetallic Fe/Pd nanoparticles by grape leaf aqueous extract and identification of active biomolecules involved in the synthesis. Sci Total Environ 562:526–532. https://doi.org/10.1016/j.scitotenv.2016.04.060
Ma G, Chai X, Hou G et al (2022) Phytochemistry, bioactivities and future prospects of mulberry leaves: a review. Food Chem 372:131335. https://doi.org/10.1016/j.foodchem.2021.131335
Machado S, Pinto SL, Grosso JP et al (2013) Green production of zero-valent iron nanoparticles using tree leaf extracts. Sci Total Environ 445–446:1–8. https://doi.org/10.1016/j.scitotenv.2012.12.033
Machado S, Pacheco JG, Nouws HPA et al (2015) Characterization of green zero-valent iron nanoparticles produced with tree leaf extracts. Sci Total Environ 533:76–81. https://doi.org/10.1016/j.scitotenv.2015.06.091
Matar GH, Andac M (2023) Green synthesis of iron oxide nanoparticles using brown Egyptian propolis extract for evaluation of their antibacterial activity and degradation of dyes. Inorg Chem Commun 153:110889. https://doi.org/10.1016/j.inoche.2023.110889
Memete AR, Timar AV, Vuscan AN et al (2022) Phytochemical composition of different botanical parts of Morus species, health benefits and application in food industry. Plants 11:152. https://doi.org/10.3390/plants11020152
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428. https://doi.org/10.1021/ac60147a030
Mittal AK, Chisti Y, Banerjee UC (2013) Synthesis of metallic nanoparticles using plant extracts. Biotechnol Adv 31:346–356. https://doi.org/10.1016/j.biotechadv.2013.01.003
Mohammadpour A, Karami N, Zabihi R et al (2023) Green synthesis, characterization, and application of Fe3O4 nanoparticles for methylene blue removal: RSM optimization, kinetic, isothermal studies, and molecular simulation. Environ Res 225:115507. https://doi.org/10.1016/j.envres.2023.115507
Moran JF, Klucas RV, Grayer RJ et al (1997) Complexes of iron with phenolic compounds from soybean nodules and other legume tissues: prooxidant and antioxidant properties. Free Radical Biol Med 22:861–870. https://doi.org/10.1016/S0891-5849(96)00426-1
Nabi BG, Mukhtar K, Ahmed W et al (2023) Natural pigments: anthocyanins, carotenoids, chlorophylls, and betalains as colorants in food products. Food Biosci 52:102403. https://doi.org/10.1016/j.fbio.2023.102403
Nasiri J, Motamedi E, Naghavi MR, Ghafoori M (2019) Removal of crystal violet from water using β-cyclodextrin functionalized biogenic zero-valent iron nanoadsorbents synthesized via aqueous root extracts of Ferula persica. J Hazard Mater 367:325–338. https://doi.org/10.1016/j.jhazmat.2018.12.079
Nikitha M, Elanchezhiyan SSD, Meenakshi S (2023) Photodegradation of rhodamine-B in aqueous environment using visible-active gC3N4@CS-MoS2 nanocomposite. Environ Res 238:117032. https://doi.org/10.1016/j.envres.2023.117032
Ouyang Q, Kou F, Tsang PE et al (2019) Green synthesis of Fe-based material using tea polyphenols and its application as a heterogeneous Fenton-like catalyst for the degradation of lincomycin. J Clean Prod 232:1492–1498. https://doi.org/10.1016/j.jclepro.2019.06.043
Pan Y, Qin R, Hou M et al (2022) The interactions of polyphenols with Fe and their application in Fenton/Fenton-like reactions. Sep Purif Technol 300:121831. https://doi.org/10.1016/j.seppur.2022.121831
Poguberović SS, Krčmar DM, Maletić SP et al (2016) Removal of As(III) and Cr(VI) from aqueous solutions using “green” zero-valent iron nanoparticles produced by oak, mulberry and cherry leaf extracts. Ecol Eng 90:42–49. https://doi.org/10.1016/j.ecoleng.2016.01.083
Prajapati P, Gupta P, Kharwar RN, Seth CS (2023) Nitric oxide mediated regulation of ascorbate-glutathione pathway alleviates mitotic aberrations and DNA damage in Allium cepa L. under salinity stress. Int J Phytorem 25:403–414. https://doi.org/10.1080/15226514.2022.2086215
Rajendran A, Alsawalha M, Alomayri T (2021) Biogenic synthesis of husked rice-shaped iron oxide nanoparticles using coconut pulp (Cocos nucifera L.) extract for photocatalytic degradation of Rhodamine B dye and their in vitro antibacterial and anticancer activity. J Saudi Chem Soc 25:101307. https://doi.org/10.1016/j.jscs.2021.101307
Rana A, Kumari N, Tyagi M, Jagadevan S (2018) Leaf-extract mediated zero-valent iron for oxidation of Arsenic (III): preparation, characterization and kinetics. Chem Eng J 347:91–100. https://doi.org/10.1016/j.cej.2018.04.075
Rizvi M, Bhatia T, Gupta R (2022) Green & sustainable synthetic route of obtaining iron oxide nanoparticles using Hylocereus undantus (pitaya or dragon fruit). Mater Today: Proc 50:1100–1106. https://doi.org/10.1016/j.matpr.2021.07.469
Saatci M, Deliormanlı AM (2023) Synthesis, characterization, and 5-fluorouracil release behavior of superparamagnetic γ-Fe2O3/ZnO hetero-nanostructures for biomedical applications. Ceram Int 49:12934–12949. https://doi.org/10.1016/j.ceramint.2022.12.165
Samadi Z, Yaghmaeian K, Mortazavi-Derazkola S et al (2021) Facile green synthesis of zero-valent iron nanoparticles using barberry leaf extract (GnZVI@BLE) for photocatalytic reduction of hexavalent chromium. Bioorg Chem 114:105051. https://doi.org/10.1016/j.bioorg.2021.105051
Sánchez-Salcedo EM, Tassotti M, Del Rio D et al (2016) (Poly)phenolic fingerprint and chemometric analysis of white (Morus alba L.) and black (Morus nigra L.) mulberry leaves by using a non-targeted UHPLC–MS approach. Food Chem 212:250–255. https://doi.org/10.1016/j.foodchem.2016.05.121
Selvaraj R, Pai S, Vinayagam R et al (2022) A recent update on green synthesized iron and iron oxide nanoparticles for environmental applications. Chemosphere 308:136331. https://doi.org/10.1016/j.chemosphere.2022.136331
Shafaghatlonbar M, Bagherzade G (2023) Dual role of chlorogenic acid as an influential precursor in synthesizing nano-sized Cu(II) complexes and investigating its catalytic role in the oxidation of alcohols and its antibacterial activity. J Organomet Chem 996:122758. https://doi.org/10.1016/j.jorganchem.2023.122758
Shaker Ardakani L, Alimardani V, Tamaddon AM et al (2021) Green synthesis of iron-based nanoparticles using Chlorophytum comosum leaf extract: methyl orange dye degradation and antimicrobial properties. Heliyon 7:e06159. https://doi.org/10.1016/j.heliyon.2021.e06159
Shet VB, Kumar PS, Vinayagam R et al (2023) Cocoa pod shell mediated silver nanoparticles synthesis, characterization, and their application as nanocatalyst and antifungal agent. Appl Nanosci 13:4235–4245. https://doi.org/10.1007/s13204-023-02873-8
Soliemanzadeh A, Fekri M (2017) Synthesis of clay-supported nanoscale zero-valent iron using green tea extract for the removal of phosphorus from aqueous solutions. Chin J Chem Eng 25:924–930. https://doi.org/10.1016/j.cjche.2016.12.006
Song M, Nguyen QB, Kim C, Hwang I (2023) Sustained activation of persulfate by slow release of Fe(II) from silica-coated nanosized zero-valent iron for in situ chemical oxidation. Water Res 246:120715. https://doi.org/10.1016/j.watres.2023.120715
Sridevi H, Bhat MR, Kumar PS et al (2023) Structural characterization of cuboidal α-Fe2O3 nanoparticles synthesized by a facile approach. Appl Nanosci 13:5605–5613. https://doi.org/10.1007/s13204-023-02780-y
Tesfaldet ZO, van Staden JF, Stefan RI (2004) Sequential injection spectrophotometric determination of iron as Fe(II) in multi-vitamin preparations using 1,10-phenanthroline as complexing agent. Talanta 64:1189–1195. https://doi.org/10.1016/j.talanta.2004.02.044
Turkmen N, Sari F, Velioglu YS (2006) Effects of extraction solvents on concentration and antioxidant activity of black and black mate tea polyphenols determined by ferrous tartrate and Folin-Ciocalteu methods. Food Chem 99:835–841. https://doi.org/10.1016/j.foodchem.2005.08.034
Vendemiatti JAS, Camparotto NG, Vidal C et al (2021) New benzotriazoles generated during textile dyeing process: synthesis, hazard, water occurrence and aquatic risk assessment. J Hazard Mater 403:123732. https://doi.org/10.1016/j.jhazmat.2020.123732
Vinayagam R, Patnaik Y, Brijesh P et al (2022) Superparamagnetic hematite spheroids synthesis, characterization, and catalytic activity. Chemosphere 294:133730. https://doi.org/10.1016/j.chemosphere.2022.133730
Vinayagam R, Hebbar A, Senthil Kumar P et al (2023) Green synthesized cobalt oxide nanoparticles with photocatalytic activity towards dye removal. Environ Res 216:114766. https://doi.org/10.1016/j.envres.2022.114766
Wang Z, Yu C, Fang C, Mallavarapu M (2014) Dye removal using iron–polyphenol complex nanoparticles synthesized by plant leaves. Environ Technol Innov 1–2:29–34. https://doi.org/10.1016/j.eti.2014.08.003
Wang X, Wang A, Ma J, Fu M (2017) Facile green synthesis of functional nanoscale zero-valent iron and studies of its activity toward ultrasound-enhanced decolorization of cationic dyes. Chemosphere 166:80–88. https://doi.org/10.1016/j.chemosphere.2016.09.056
Wang J, Gao M, Shen T et al (2019) Insights into the efficient adsorption of rhodamine B on tunable organo-vermiculites. J Hazard Mater 366:501–511. https://doi.org/10.1016/j.jhazmat.2018.12.031
Wang B, Zhu C, Ai D, Fan Z (2021) Activation of persulfate by green nano-zero-valent iron-loaded biochar for the removal of p-nitrophenol: performance, mechanism and variables effects. J Hazard Mater 417:126106. https://doi.org/10.1016/j.jhazmat.2021.126106
Wang C, Wang X, Wu W et al (2023) A biochar-based catalyst prepared by potassium ferrate oxidation coupled high-temperature pyrolysis and its application for the activation of peroxymonosulfate towards the degradation of norfloxacin: performance and mechanism insight. Sep Purif Technol 323:124302. https://doi.org/10.1016/j.seppur.2023.124302
Wu Z, Su X, Lin Z et al (2021) Removal of As(V) by iron-based nanoparticles synthesized via the complexation of biomolecules in green tea extracts and an iron salt. Sci Total Environ 764:142883. https://doi.org/10.1016/j.scitotenv.2020.142883
Wu J, Wu P, Weng X et al (2022) Mechanistic insight into the one step green synthesis of hybrid rGO/Fe NPs. Materials Today Nano 18:100193. https://doi.org/10.1016/j.mtnano.2022.100193
Xiao Z, Yuan M, Yang B et al (2016) Plant-mediated synthesis of highly active iron nanoparticles for Cr (VI) removal: investigation of the leading biomolecules. Chemosphere 150:357–364. https://doi.org/10.1016/j.chemosphere.2016.02.056
Xiao C, Li H, Zhao Y et al (2020) Green synthesis of iron nanoparticle by tea extract (polyphenols) and its selective removal of cationic dyes. J Environ Manage 275:111262. https://doi.org/10.1016/j.jenvman.2020.111262
Yu X, Shang T, Zheng G et al (2022) Metal-polyphenol-coordinated nanomedicines for Fe(II) catalyzed photoacoustic-imaging guided mild hyperthermia-assisted ferroustherapy against breast cancer. Chin Chem Lett 33:1895–1900. https://doi.org/10.1016/j.cclet.2021.10.021
Yuan X, Yu S, Xue N et al (2023) Activation of iron based persulfate heterogeneous nano catalyst using plant extract for removal of tetrabromobisphenol A from soil. J Environ Chem Eng 11:109493. https://doi.org/10.1016/j.jece.2023.109493
Zhishen J, Mengcheng T, Jianming W (1999) The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem 64:555–559. https://doi.org/10.1016/S0308-8146(98)00102-2
Zhou H, Ma M, Zhao Y et al (2022) Integrated green complexing agent and biochar modified nano zero-valent iron for hexavalent chromium removal: a characterisation and performance study. Sci Total Environ 834:155080. https://doi.org/10.1016/j.scitotenv.2022.155080
Zhu F, Ma S, Liu T, Deng X (2018) Green synthesis of nano zero-valent iron/Cu by green tea to remove hexavalent chromium from groundwater. J Clean Prod 174:184–190. https://doi.org/10.1016/j.jclepro.2017.10.302
Funding
This work was supported by the Natural Science Foundation of Anhui Province (Grant No. 2108085ME187), University Natural Science Research Key Project of Anhui Province (Grant No. 2022AH050338), and the Open Project of Engineering Research Center of Biofilm Water Purification and Utilization Technology of Ministry of Education (Grant No. BWPU2020KF01).
Author information
Authors and Affiliations
Contributions
The contributions of each author to this study are as follows: Xinxiang Wang: methodology, investigation, and writing (original draft preparation, editing); Jinwei Zhang: investigation, data curation; Yiqi Liu: conceptualization, writing ((original draft editing); Yan Li: investigation; Yuntao Zhu: investigation; Zhiqiang Dong: investigation; Dongxiao Sun: supervision; Lei Ding: conceptualization, writing (editing) and supervision.
Corresponding author
Ethics declarations
Ethics approval
All authors have read and agreed to the published version of the manuscript.
Consent to participate
Not applicable.
Consent for publication
All authors approved this for publication.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: George Z. Kyzas
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, X., Zhang, J., Liu, Y. et al. Green synthesis of iron nanoparticles using mulberry leaf extract: characterization, identification of active biomolecules, and catalytic activity. Environ Sci Pollut Res 31, 20311–20329 (2024). https://doi.org/10.1007/s11356-024-32405-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-024-32405-y