Abstract
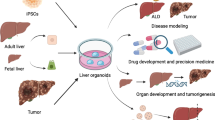
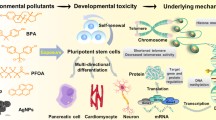
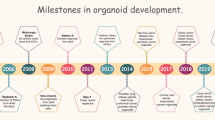
As the economy rapidly develops, chemicals are widely produced and used. This has exacerbated the problems associated with environmental pollution, raising the need for efficient toxicological evaluation techniques to investigate the toxic effects and mechanisms of toxicity of environmental pollutants. The progress in the techniques of cell culture in three dimensions has resulted in the creation of models that are more relevant in terms of biology and physiology. This enables researchers to study organ development, toxicology, and drug screening. Adult stem cells (ASCs) and induced pluripotent stem cells (iPSCs) can be obtained from various mammalian tissues, including cancerous and healthy tissues. Such stem cells exhibit a significant level of tissue memory and ability to self-assemble. When cultivated in 3D in vitro environments, the resulting organoids demonstrate a remarkable capacity to recapitulate the cellular composition and function of organs in vivo. Recently, many tumors’ tissue-derived organoids have been widely used in research on tumor pathogenesis, drug development, precision medicine, and other fields, including those derived from colon cancer, cholangiocarcinoma, liver cancer, and gastric cancer. However, the application of organoid models for evaluating the toxicity of environmental pollutants is still in its infancy. This review introduces the characteristics of the toxicity responses of organoid models upon exposure to pollutants from the perspectives of organoid characteristics, tissue types, and their applications in toxicology; discusses the feasibility of using organoid models in evaluating the toxicity of pollutants; and provides a reference for future toxicological studies on environmental pollutants based on organoid models.





Similar content being viewed by others
Data availability
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
References
Antoni D, Burckel H, Josset E, Noel G (2015) Three-dimensional cell culture: a breakthrough in vivo. Int J Mol Sci 16(3):5517–5527. https://doi.org/10.3390/ijms16035517
Asakawa K, Toyoshima K-e, Ishibashi N, Tobe H, Iwadate A, Kanayama T, Hasegawa T, Nakao K, Toki H, Noguchi S, Ogawa M, Sato A, Tsuji T (2012) Hair organ regeneration via the bioengineered hair follicular unit transplantation. Sci Rep 2:424. https://doi.org/10.1038/srep00424
Astashkina A, Mann B, Grainger DW (2012) A critical evaluation of in vitro cell culture models for high-throughput drug screening and toxicity. Pharmacol Ther 134(1):82–106. https://doi.org/10.1016/j.pharmthera.2012.01.001
Baldassi D, Gabold B, Merkel OM (2021) Air-liquid interface cultures of the healthy and diseased human respiratory tract: Promises, challenges, and future directions. Adv Nanobiomed Res 1(6):2000111. https://doi.org/10.1002/anbr.202000111
Ballard DH, Boyer CJ, Alexander JS (2019) Organoids—preclinical models of human disease. N Engl J Med 380(20):1981–1982. https://doi.org/10.1056/NEJMc1903253
Baxter M, Withey S, Harrison S, Segeritz C-P et al (2015) Phenotypic and functional analyses show stem cell-derived hepatocyte-like cells better mimic fetal rather than adult hepatocytes. J Hepatol 62(3):581–589. https://doi.org/10.1016/j.jhep.2014.10.016
Bhatia SN, Underhill GH, Zaret KS, Fox IJ (2014) Cell and tissue engineering for liver disease. Sci Transl Med 6(245):245sr2. https://doi.org/10.1126/scitranslmed.3005975
Boonekamp KE, Kretzschmar K, Wiener DJ, Asra P et al (2019) Long-term expansion and differentiation of adult murine epidermal stem cells in 3d organoid cultures. Proc Natl Acad Sci U S A 116(29):14630–14638. https://doi.org/10.1073/pnas.1715272116
Broutier L, Mastrogiovanni G, Verstegen MMA, Francies HE et al (2017) Human primary liver cancer-derived organoid cultures for disease modeling and drug screening. Nat Med 23(12):1424–1435. https://doi.org/10.1038/nm.4438
Chang Y, Siddens LK, Heine LK, Sampson DA et al (2019) Comparative mechanisms of pah toxicity by benzo[a]pyrene and dibenzo[def, p]chrysene in primary human bronchial epithelial cells cultured at air-liquid interface. Toxicol Appl Pharmacol 379:114644. https://doi.org/10.1016/j.taap.2019.114644
Chen YX, Xie GC, Pan D, Du YR et al (2018) Three-dimensional culture of human airway epithelium in matrigel for evaluation of human rhinovirus c and bocavirus infections. Biomed Environ Sci 31(2):136–145. https://doi.org/10.3967/bes2018.016
Chen S, Chen Y, Gao Y, Han B et al (2023) Toxic effects and mechanisms of nanoplastics on embryonic brain development using brain organoids model. Sci Total Environ 904:166913–166913. https://doi.org/10.1016/j.scitotenv.2023.166913
Chen YW, Huang SX, Toste Rodrigues, de Carvalho AL, Ho S-H, Islam MN, Volpi S, Notarangelo LD, Ciancanelli M, Casanova J-L, Bhattacharya J, Liang AF, Palermo LM, Porotto M, Moscona A, Snoeck H-W (2017) A three-dimensional model of human lung development and disease from pluripotent stem cells. Nat Cell Biol 19(5):542–549. https://doi.org/10.1038/ncb3510
Cheng W, Li X, Zhou Y, Yu H, Xie Y, Guo H, Wang H, Li Y, Feng Y, Wang Y (2022) Polystyrene microplastics induce hepatotoxicity and disrupt lipid metabolism in the liver organoids. Sci Total Environ 806:150328. https://doi.org/10.1016/j.scitotenv.2021.150328
Cheng W, Zhou Y, Xie Y, Li Y, Zhou R, Wang H, Feng Y, Wang Y (2023) Combined effect of polystyrene microplastics and bisphenol a on the human embryonic stem cells-derived liver organoids: The hepatotoxicity and lipid accumulation. Sci Total Environ 854:158585. https://doi.org/10.1016/j.scitotenv.2022.158585
Cheng W, Zhou Y, Chen H, Wu Q, Li Y, Wang H, Feng Y, Wang Y (2024) The iron matters: Aged microplastics disrupted the iron homeostasis in the liver organoids. Sci Total Environ 906:167529. https://doi.org/10.1016/j.scitotenv.2023.167529
Choi J, Iich E, Lee J-H (2016) Organogenesis of adult lung in a dish: differentiation, disease and therapy. Dev Biol 420(2):278–286. https://doi.org/10.1016/j.ydbio.2016.10.002
Choi S, Kim E-M, Kim S-Y, Choi Y, Choi S, Cho N, Park H-J, Kim KK (2022) Particulate matter exposure exacerbates cellular damage by increasing stress granule formation in respiratory syncytial virus-infected human lung organoids. Environ Pollut 315:120439. https://doi.org/10.1016/j.envpol.2022.120439
Clevers H, Loh KM, Nusse R (2014) An integral program for tissue renewal and regeneration: Wnt signaling and stem cell control.Science 346:1248012. https://doi.org/10.1126/science.1248012
Devall M, Jennelle LT, Bryant J, Bien S, Peters U, Powell S, Casey G (2020) Modeling the effect of prolonged ethanol exposure on global gene expression and chromatin accessibility in normal 3d colon organoids. PLoS One 15(1):e0227116. https://doi.org/10.1371/journal.pone.0227116
Dignass AU, Sturm A (2001) Peptide growth factors in the intestine. Eur J Gastroenterol Hepatol 13(7):763–770. https://doi.org/10.1097/00042737-200107000-00002
Du Z, Zang Z, Luo J, Liu T, Yang L, Cai Y, Wang L, Zhang D, Zhao J, Gao J, Lv K, Wang L, Li H, Gong H, Fan X (2023) Chronic exposure to (2R, 6R)-hydroxynorketamine induces developmental neurotoxicity in hesc-derived cerebral organoids. J Hazard Mater 453:131379. https://doi.org/10.1016/j.jhazmat.2023.131379
Duval K, Grover H, Han L-H, Mou Y, Pegoraro AF, Fredberg J, Chen Z (2017) Modeling physiological events in 2D vs. 3D cell culture. Physiology 32(4):266–277. https://doi.org/10.1152/physiol.00036.2016
Dye BR, Hill DR, Ferguson MAH, Tsai Y-H, Nagy MS, Dyal R, Wells JM, Mayhew CN, Nattiv R, Klein OD, White ES, Deutsch GH, Spence JR (2015) In vitro generation of human pluripotent stem cell derived lung organoids. Elife 4:e05098. https://doi.org/10.7554/eLife.05098
Ebisudani T, Sugimoto S, Haga K, Mitsuishi A, Takai-Todaka R, Fujii M, Toshimitsu K, Hamamoto J, Sugihara K, Hishida T, Asamura H, Fukunaga K, Yasuda H, Katayama K, Sato T (2021) Direct derivation of human alveolospheres for sars-cov-2 infection modeling and drug screening. Cell Rep 35(10):109218. https://doi.org/10.1016/j.celrep.2021.109218
Eiraku M, Takata N, Ishibashi H, Kawada M et al (2011) Self-organizing optic-cup morphogenesis in three-dimensional culture. Nature 472(7341):51-U73. https://doi.org/10.1038/nature09941
Ergir E, Oliver-De la Cruz J, Fernandes S, Cassani M, Niro F, Pereira-Sousa D, Vrbsky J, Vinarsky V, Perestrelo AR, Debellis D, Vadovicova N, Uldrijan S, Cavalieri F, Pagliari S, Redl H, Ertl P, Forte G (2022) Generation and maturation of human ipsc-derived 3d organotypic cardiac microtissues in long-term culture. Sci Rep 12(1):17409. https://doi.org/10.1038/s41598-022-22225-w
Evans KV, Lee J-H (2020) Alveolar wars: the rise of in vitro models to understand human lung alveolar maintenance, regeneration, and disease. Stem Cells Transl Med 9(8):867–881. https://doi.org/10.1002/sctm.19-0433
Fatehullah A, Tan SH, Barker N (2016) Organoids as an in vitro model of human development and disease. Nat Cell Biol 18(3):246–254. https://doi.org/10.1038/ncb3312
Fernandes KJL, McKenzie IA, Mill P, Smith KM et al (2004) A dermal niche for multipotent adult skin-derived precursor cells. Nat Cell Biol 6(11):1082-U16. https://doi.org/10.1038/ncb1181
Forsythe SD, Devarasetty M, Shupe T, Bishop C, Atala A, Soker S, Skardal A (2018) Environmental toxin screening using human-derived 3d bioengineered liver and cardiac organoids. Front Public Health 6:103. https://doi.org/10.3389/fpubh.2018.00103
Fry RC, Bangma J, Szilagyi J, Rager JE (2019) Developing novel in vitro methods for the risk assessment of developmental and placental toxicants in the environment. Toxicol Appl Pharmacol 378:114635. https://doi.org/10.1016/j.taap.2019.114635
Fuchs E (2007) Scratching the surface of skin development. Nature 445(7130):834–42. https://doi.org/10.1038/nature05659
Garreta E, Kamm RD, de Sousa C, Lopes SM, Lancaster MA et al (2021) Rethinking organoid technology through bioengineering. Nat Mater 20(2):145–155. https://doi.org/10.1038/s41563-020-00804-4
Gendoo DMA, Denroche RE, Zhang A, Radulovich N, Jang GH, Lemire M, Fischer S, Chadwick D, Lungu IM, Ibrahimov E, Cao P-J, Stein LD, Wilson JM, Bartlett JMS, Tsao M-S, Dhani N, Hedley D, Gallinger S, Haibe-Kains B (2019) Whole genomes define concordance of matched primary, xenograft, and organoid models of pancreas cancer. PLoS Comput Biol 15(1):e1006596. https://doi.org/10.1371/journal.pcbi.1006596
Giandomenico SL, Mierau SB, Gibbons GM, Wenger LMD et al (2019) Cerebral organoids at the air-liquid interface generate diverse nerve tracts with functional output. Nat Neurosci 22(4):669–679. https://doi.org/10.1038/s41593-019-0350-2
Giandomenico SL, Sutcliffe M, Lancaster MA (2021) Generation and long-term culture of advanced cerebral organoids for studying later stages of neural development. Nat Protoc 16(2):579–602. https://doi.org/10.1038/s41596-020-00433-w
Gibb S (2008) Toxicity testing in the 21st century: a vision and a strategy. Reprod Toxicol 25(1):136–138. https://doi.org/10.1016/j.reprotox.2007.10.013
Grabinger T, Luks L, Kostadinova F, Zimberlin C, Medema JP, Leist M, Brunner T (2014) Ex vivo culture of intestinal crypt organoids as a model system for assessing cell death induction in intestinal epithelial cells and enteropathy. Cell Death Dis 5:e1228. https://doi.org/10.1038/cddis.2014.183
Guan Y, Xu D, Garfin PM, Ehmer U, Hurwitz M, Enns G, Michie S, Wu M, Zheng M, Nishimura T, Sage J, Peltz G (2017) Human hepatic organoids for the analysis of human genetic diseases. Jci Insight 2(17):e94954. https://doi.org/10.1172/jci.insight.94954
Han Y, Duan X, Yang L, Nilsson-Payant BE et al (2021) Identification of sars-cov-2 inhibitors using lung and colonic organoids. Nature 589(7841):270–275. https://doi.org/10.1038/s41586-020-2901-9
Haramis APG, Begthel H, van den Born M, van Es J et al (2004) De novo crypt formation and juvenile polyposis on bmp inhibition in mouse intestine. Science 303(5664):1684–1686. https://doi.org/10.1126/science.1093587
Hines EA, Sun X (2014) Tissue crosstalk in lung development. J Cell Biochem 115(9):1469–1477. https://doi.org/10.1002/jcb.24811
Hofbauer P, Jahnel SM, Papai N, Giesshammer M et al (2021) Cardioids reveal self-organizing principles of human cardiogenesis. Cell 184(12):3299-3317.e22. https://doi.org/10.1016/j.cell.2021.04.034
Hu H, Gehart H, Artegiani B, Lopez-Iglesias C et al (2018) Long-term expansion of functional mouse and human hepatocytes as 3D organoids. Cell 175(6):1591-1606.e19. https://doi.org/10.1016/j.cell.2018.11.013
Hu S, Li Z, Lutz H, Huang K et al (2020) Dermal exosomes containing mir-218–5p promote hair regeneration by regulating beta-catenin signaling. Sci Adv 6(30):eaba1685. https://doi.org/10.1126/sciadv.aba1685
Huang Y, Dai Y, Li M, Guo L, Cao C, Huang Y, Ma R, Qiu S, Su X, Zhong K, Huang Y, Gao H, Bu Q (2021) Exposure to cadmium induces neuroinflammation and impairs ciliogenesis in hesc-derived 3d cerebral organoids. Sci Total Environ 797:149043. https://doi.org/10.1016/j.scitotenv.2021.149043
Imamura Y, Mukohara T, Shimono Y, Funakoshi Y et al (2015) Comparison of 2D- and 3D-culture models as drug-testing platforms in breast cancer. Oncol Rep 33(4):1837–43. https://doi.org/10.3892/or.2015.3767
Jiang Y, Lu L, Du C, Li Y, Cheng W, Bi H, Li G, Zhuang M, Ren D, Wang H, Ji X (2023) Human airway organoids as 3d in vitro models for a toxicity assessment of emerging inhaled pollutants: Tire wear particles. Front Bioeng Biotechnol 10:1105710. https://doi.org/10.3389/fbioe.2022.1105710
Kadoshima T, Sakaguchi H, Nakano T, Soen M et al (2013) Self-organization of axial polarity, inside-out layer pattern, and species-specific progenitor dynamics in human es cell-derived neocortex. Proc Natl Acad Sci U S A 110(50):20284–20289. https://doi.org/10.1073/pnas.1315710110
Kaluthantrige Don F, Huch M (2021) Organoids, where we stand and where we go. Trends Mol Med 27(5):416–418. https://doi.org/10.1016/j.molmed.2021.03.001
Kapalczynska M, Kolenda T, Przybyla W, Zajaczkowska M et al (2018) 2d and 3d cell cultures—a comparison of different types of cancer cell cultures. Arch Med Sci 14(4):910–919. https://doi.org/10.5114/aoms.2016.63743
Katsuda T, Kawamata M, Hagiwara K, Takahashi R-u et al (2017) Conversion of terminally committed hepatocytes to culturable bipotent progenitor cells with regenerative capacity. Cell Stem Cell 20(1):41–55. https://doi.org/10.1016/j.stem.2016.10.007
Khademhosseini A, Eng G, Yeh J, Kucharczyk PA et al (2007) Microfluidic patterning for fabrication of contractile cardiac organoids. Biomed Microdevice 9(2):149–57. https://doi.org/10.1007/s10544-006-9013-7
Korinek V, Barker N, Moerer P, van Donselaar E et al (1998) Depletion of epithelial stem-cell compartments in the small intestine of mice lacking tcf-4. Nat Genet 19(4):379–83. https://doi.org/10.1038/1270
Kostadinova R, Boess F, Applegate D, Suter L et al (2013) A long-term three dimensional liver co-culture system for improved prediction of clinically relevant drug-induced hepatotoxicity. Toxicol Appl Pharmacol 268(1):1–16. https://doi.org/10.1016/j.taap.2013.01.012
Kozlovsky P, Bryson-Richardson RJ, Jaffa AJ, Rosenfeld M, Elad D (2016) The driving mechanism for unidirectional blood flow in the tubular embryonic heart. Ann Biomed Eng 44(10):3069–3083. https://doi.org/10.1007/s10439-016-1620-8
Lancaster MA, Knoblich JA (2014) Organogenesis in a dish: modeling development and disease using organoid technologies. Science 345(6194):1247125. https://doi.org/10.1126/science.1247125
Lancaster MA, Renner M, Martin CA, Wenzel D et al (2013) Cerebral organoids model human brain development and microcephaly. Nature 501(7467):373–379. https://doi.org/10.1038/nature12517
Lancaster MA, Corsini NS, Wolfinger S, Gustafson EH et al (2017) Guided self-organization and cortical plate formation in human brain organoids. Nat Biotechnol 35(7):659–666. https://doi.org/10.1038/nbt.3906
Langhans SA (2018) Three-dimensional in vitro cell culture models in drug discovery and drug repositioning. Front Pharmacol 9:6–6. https://doi.org/10.3389/fphar.2018.00006
Lee J, Boscke R, Tang P-C, Hartman BH, Heller S, Koehler KR (2018) Hair follicle development in mouse pluripotent stem cell-derived skin organoids. Cell Rep 22(1):242–254. https://doi.org/10.1016/j.celrep.2017.12.007
Lee J, Sutani A, Kaneko R, Takeuchi J, Sasano T, Kohda T, Ihara K, Takahashi K, Yamazoe M, Morio T, Furukawa T, Ishino F (2020) In vitro generation of functional murine heart organoids via fgf4 and extracellular matrix. Nat Commun 11(1):4283. https://doi.org/10.1038/s41467-020-18031-5
Lim X, Nusse R (2013) Wnt signaling in skin development, homeostasis, and disease. Cold Spring Harb Perspect Biol 5(2):a008029. https://doi.org/10.1101/cshperspect.a008029
Liu Y, Luo H, Wang X, Takemura A, Fang YR, Jin Y, Suwa F (2013) In vitro construction of scaffold-free bilayered tissue-engineered skin containing capillary networks. BioMed Res Int 2013:561410. https://doi.org/10.1155/2013/561410
Lou YR, Leung AW (2018) Next generation organoids for biomedical research and applications. Biotechnol Adv 36(1):132–149. https://doi.org/10.1016/j.biotechadv.2017.10.005
MacArthur Clark J (2018) The 3rs in research: a contemporary approach to replacement, reduction and refinement. Br J Nutr 120(s1):S1–S7. https://doi.org/10.1017/S0007114517002227
Mariani J, Simonini MV, Palejev D, Tomasini L et al (2012) Modeling human cortical development in vitro using induced pluripotent stem cells. Proc Natl Acad Sci U S A 109(31):12770–12775. https://doi.org/10.1073/pnas.1202944109
Meli L, Jordan ET, Clark DS, Linhardt RJ, Dordick JS (2012) Influence of a three-dimensional, microarray environment on human cell culture in drug screening systems. Biomaterials 33(35):9087–9096. https://doi.org/10.1016/j.biomaterials.2012.08.065
Mittal R, Woo FW, Castro CS, Cohen MA et al (2019) Organ-on-chip models: implications in drug discovery and clinical applications. J Cell Physiol 234(6):8352–8380. https://doi.org/10.1002/jcp.27729
Miyamoto M, Nam L, Kannan S, Kwon C (2021) Heart organoids and tissue models for modeling development and disease. Semin Cell Dev Biol 118:119–128. https://doi.org/10.1016/j.semcdb.2021.03.011
Moon S-H, Baez D, Jung T-H, Chung E-B et al (2017) From bench to market: preparing human pluripotent stem cells derived cardiomyocytes for various applications. Int J Stem Cells 10(1):1–11. https://doi.org/10.15283/ijsc17024
Naruse M, Masui R, Ochiai M, Maru Y, Hippo Y, Imai T (2020) An organoid-based carcinogenesis model induced by in vitro chemical treatment. Carcinogenesis 41(10):1444–1453. https://doi.org/10.1093/carcin/bgaa011
Nikolic MZ, Caritg O, Jeng Q, Johnson J-A, Sun D, Howell KJ, Brady JL, Laresgoiti U, Allen G, Butler R, Zilbauer M, Giangreco A, Rawlins EL (2017) Human embryonic lung epithelial tips are multipotent progenitors that can be expanded in vitro as long-term self-renewing organoids. Elife 6:e26575. https://doi.org/10.7554/eLife.26575.001
O’Connell L, Winter DC (2020) Organoids: past learning and future directions. Stem Cells Dev 29(5):281–289. https://doi.org/10.1089/scd.2019.0227
Palazzolo S, Caligiuri I, Sfriso AA, Mauceri M, Rotondo R, Campagnol D, Canzonieri V, Rizzolio F (2022) Early warnings by liver organoids on short- and long-chain pfas toxicity. Toxics 10(2):91. https://doi.org/10.3390/toxics10020091
Pampaloni F, Reynaud EG, Stelzer EH (2007) The third dimension bridges the gap between cell culture and live tissue. Nat Rev Mol Cell Biol 8(10):839–45. https://doi.org/10.1038/nrm2236
Park E, Kim HK, Jee J, Hahn S, Jeong S, Yoo J (2019) Development of organoid-based drug metabolism model. Toxicol Appl Pharmacol 385:114790. https://doi.org/10.1016/j.taap.2019.114790
Pinto D, Gregorieff A, Begthel H, Clevers H (2003) Canonical wnt signals are essential for homeostasis of the intestinal epithelium. Genes Dev 17(14):1709–1713. https://doi.org/10.1101/gad.267103
Prior N, Inacio P, Huch M (2019) Liver organoids: from basic research to therapeutic applications. Gut 68(12):2228–2237. https://doi.org/10.1136/gutjnl-2019-319256
Qian X, Ha Nam N, Song MM, Hadiono C et al (2016) Brain-region-specific organoids using mini-bioreactors for modeling zikv exposure. Cell 165(5):1238–1254. https://doi.org/10.1016/j.cell.2016.04.032
Quadrato G, Tuan N, Macosko EZ, Sherwood JL, Yang SM, Berger DR, Maria N, Scholvin J, Goldman M, Kinney JP, Boyden ES, Lichtman JW, Williams ZM, McCarroll SA, Arlotta P (2017) Cell diversity and network dynamics in photosensitive human brain organoids. Nature 545:48–5. https://doi.org/10.1038/nature22047
Quarto N, Wan DC, Kwan MD, Panetta NJ, Li S, Longaker MT (2010) Origin matters: differences in embryonic tissue origin and wnt signaling determine the osteogenic potential and healing capacity of frontal and parietal calvarial bones. J Bone Miner Res 25(7):1680–94. https://doi.org/10.1359/jbmr.091116
Rocco SA, Koneva L, Middleton LYM, Tasha T et al (2018) Cadmium exposure inhibits branching morphogenesis and causes alterations consistent with hif-1 alpha inhibition in human primary breast organoids. Toxicol Sci 164(2):592–602. https://doi.org/10.1093/toxsci/kfy112
Rock JR, Onaitis MW, Rawlins EL, Lu Y et al (2009) Basal cells as stem cells of the mouse trachea and human airway epithelium. Proc Natl Acad Sci U S A 106(31):12771–12775. https://doi.org/10.1073/pnas.0906850106
Rossi G, Manfrin A, Lutolf MP (2018) Progress and potential in organoid research. Nat Rev Genet 19(11):671–687. https://doi.org/10.1038/s41576-018-0051-9
Rowe RG, Daley GQ (2019) Induced pluripotent stem cells in disease modelling and drug discovery. Nat Rev Genet 20(7):377–388. https://doi.org/10.1038/s41576-019-0100-z
Sachs N, Papaspyropoulos A, Zomer-van Ommen DD, Heo I, Bottinger L et al (2019) Long-term expanding human airway organoids for disease modeling. EMBO J 38(4):e100300. https://doi.org/10.15252/embj.2018100300
Saito Y, Muramatsu T, Kanai Y, Ojima H et al (2019) Establishment of patient-derived organoids and drug screening for biliary tract carcinoma. Cell Rep 27(4):1265-1276.e4. https://doi.org/10.1016/j.celrep.2019.03.088
Sakib S, Voigt A, Goldsmith T, Dobrinski I (2019) Three-dimensional testicular organoids as novel in vitro models of testicular biology and toxicology. Environ Epigenet 5(3):dvz011. https://doi.org/10.1093/eep/dvz011
Salahudeen AA, Choi SS, Rustagi A, Zhu J et al (2020) Progenitor identification and sars-cov-2 infection in human distal lung organoids. Nature 588(7839):670–675. https://doi.org/10.1038/s41586-020-3014-1
Sampaziotis F, de Brito MC, Madrigal P, Bertero A et al (2015) Cholangiocytes derived from human induced pluripotent stem cells for disease modeling and drug validation. Nat Biotechnol 33(8):845–852. https://doi.org/10.1038/nbt.3275
Sato T, Clevers H (2013) Growing self-organizing mini-guts from a single intestinal stem cell: mechanism and applications. Science 340(6137):1190–1194. https://doi.org/10.1126/science.1234852
Sato T, Vries RG, Snippert HJ, van de Wetering M et al (2009) Single Lgr5+ stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459(7244):262-U147. https://doi.org/10.1038/nature07935
Sato T, Clevers H (2015) Snapshot: Growing organoids from stem cells. Cell 161(7):1700.e1. https://doi.org/10.1016/j.cell.2015.06.028
Silva-Almeida C, Ewart MA, Wilde C (2020) 3D gastrointestinal models and organoids to study metabolism in human colon cancer. Semin Cell Dev Biol 98:98–104. https://doi.org/10.1016/j.semcdb.2019.05.019
Song Z, Cai J, Liu Y, Zhao D et al (2009) Efficient generation of hepatocyte-like cells from human induced pluripotent stem cells. Cell Res 19(11):1233–1242. https://doi.org/10.1038/cr.2009.107
Souza AG, Silva IBB, Campos-Fernandez E, Barcelos LS et al (2018) Comparative assay of 2D and 3D cell culture models: proliferation, gene expression and anticancer drug response. Curr Pharm Des 24(15):1689–1694. https://doi.org/10.2174/1381612824666180404152304
Stingl J, Eaves CJ, Zandieh I, Emerman JT (2001) Characterization of bipotent mammary epithelial progenitor cells in normal adult human breast tissue. Breast Cancer Res Treat 67(2):93–109. https://doi.org/10.1023/a:1010615124301
Sun T, Jackson S, Haycock JW, MacNeil S (2006) Culture of skin cells in 3D rather than 2D improves their ability to survive exposure to cytotoxic agents. J Biotechnol 122(3):372–81. https://doi.org/10.1016/j.jbiotec.2005.12.021
Szkolnicka D, Farnworth SL, Lucendo-Villarin B, Hay DC (2014) Deriving functional hepatocytes from pluripotent stem cells. Curr Protoc Cell Biol 30:1G.5.1–12. https://doi.org/10.1002/9780470151808.sc01g05s30
Taguchi A, Kaku Y, Ohmori T, Sharmin S et al (2014) Redefining the in vivo origin of metanephric nephron progenitors enables generation of complex kidney structures from pluripotent stem cells. Cell Stem Cell 14(1):53–67. https://doi.org/10.1016/j.stem.2013.11.010
Takasato M, Er PX, Becroft M, Vanslambrouck JM, Stanley EG, Elefanty AG, Little MH (2014) Directing human embryonic stem cell differentiation towards a renal lineage generates a self-organizing kidney. Nat Cell Biol 16:118–126. https://doi.org/10.1038/ncb2894
Tchieu J, Zimmer B, Fattahi F, Amin S, Zeltner N, Chen S, Studer L (2017) A modular platform for differentiation of human pscs into all major ectodermal lineages. Cell Stem Cell 21(3):399–410.e7. https://doi.org/10.1016/j.stem.2017.08.015
van de Wetering M, Francies HE, Francis JM, Bounova G et al (2015) Prospective derivation of a living organoid biobank of colorectal cancer patients. Cell 161(4):933–945. https://doi.org/10.1016/j.cell.2015.03.053
Veninga V, Voest EE (2021) Tumor organoids: opportunities and challenges to guide precision medicine. Cancer Cell 39(9):1190–1201. https://doi.org/10.1016/j.ccell.2021.07.020
Wang Y, Wang L, Zhu Y, Qin J (2018) Human brain organoid-on-a-chip to model prenatal nicotine exposure. Lab Chip 18(6):851–860. https://doi.org/10.1039/c7lc01084b
Wang Z, Walker GW, Muir DCG, Nagatani-Yoshida K (2020) Toward a global understanding of chemical pollution: a first comprehensive analysis of national and regional chemical inventories. Environ Sci Technol 54(5):2575–2584. https://doi.org/10.1021/acs.est.9b06379
Wang X, Wang S, Guo B, Su Y, Tan Z, Chang M, Diao J, Zhao Y, Wang Y (2021) Human primary epidermal organoids enable modeling of dermatophyte infections. Cell Death Dis 12(1):35. https://doi.org/10.1038/s41419-020-03330-y
Wang R, Kang N, Zhang W, Chen B, Xu S, Wu L (2023) The developmental toxicity of pm2.5 on the early stages of fetal lung with human lung bud tip progenitor organoids. Environ Pollut 330:121764. https://doi.org/10.1016/j.envpol.2023.121764
Winkler AS, Cherubini A, Rusconi F, Santo N, Madaschi L, Pistoni C, Moschetti G, Sarnicola ML, Crosti M, Rosso L, Tremolada P, Lazzari L, Bacchetta R (2022) Human airway organoids and microplastic fibers: A new exposure model for emerging contaminants. Environ Int 163:107200. https://doi.org/10.1016/j.envint.2022.107200
Wu F, Wu D, Ren Y, Huang Y et al (2019) Generation of hepatobiliary organoids from human induced pluripotent stem cells. J Hepatol 70(6):1145–1158. https://doi.org/10.1016/j.jhep.2018.12.028
Xiang Y, Tanaka Y, Cakir B, Patterson B et al (2019) Hesc-derived thalamic organoids form reciprocal projections when fused with cortical organoids. Cell Stem Cell 24(3):487–497. https://doi.org/10.1016/j.stem.2018.12.015
Xie S, Jiang L, Wang M, Sun W, Yu S, Turner JR, Yu Q (2020) Cadmium ingestion exacerbates salmonella infection, with a loss of goblet cells through activation of notch signaling pathways by ros in the intestine. J Hazard Mater 391:122262. https://doi.org/10.1016/j.jhazmat.2020.122262
Yamamoto J, Udono M, Miura S, Sekiya S, Suzuki A (2018) Cell aggregation culture induces functional differentiation of induced hepatocyte-like cells through activation of hippo signaling. Cell Rep 25(1):183–198. https://doi.org/10.1016/j.celrep.2018.09.010
Yang L, Zou J, Zang Z, Wang L, Du Z, Zhang D, Cai Y, Li M, Li Q, Gao J, Xu H, Fan X (2023) Di-(2-ethylhexyl) phthalate exposure impairs cortical development in hesc-derived cerebral organoids. Sci Total Environ 865:161251. https://doi.org/10.1016/j.scitotenv.2022.161251
Yin F, Zhu Y, Wang Y, Qin J (2018) Engineering brain organoids to probe impaired neurogenesis induced by cadmium. ACS Biomater Sci Eng 4(5):1908–1915. https://doi.org/10.1021/acsbiomaterials.8b00160
Zepp JA, Zacharias WJ, Frank DB, Cavanaugh CA et al (2017) Distinct mesenchymal lineages and niches promote epithelial self-renewal and myofibrogenesis in the lung. Cell 170(6):1134–1148. https://doi.org/10.1016/j.cell.2017.07.034
Zhang B, Montgomery M, Chamberlain MD, Ogawa S et al (2016) Biodegradable scaffold with built-in vasculature for organ-on-a-chip engineering and direct surgical anastomosis. Nat Mater 15(6):669–678. https://doi.org/10.1038/nmat4570
Zhong X, Gutierrez C, Xue T, Hampton C, Vergara MN, Cao L-H, Peters A, Park TS, Zambidis ET, Meyer JS, Gamm DM, Yau K-W, Canto-Soler MV (2014) Generation of three-dimensional retinal tissue with functional photoreceptors from human ipscs. Nat Commun 5:4047. https://doi.org/10.1038/ncomms5047
Zhou Y, Wu Q, Li Y, Feng Y, Wang Y, Cheng W (2023) Low-dose of polystyrene microplastics induce cardiotoxicity in mice and human-originated cardiac organoids. Environ Int 179:108171. https://doi.org/10.1016/j.envint.2023.108171
Funding
This work was supported by the National Natural Science Foundation of China (42077430), the Natural Science Foundation of Jiangsu Province, China (BK20221325), the Qing Lan Project of Jiangsu Province, and the Special Science and Technology Innovation Program for Carbon Peak and Carbon Neutralization of Jiangsu Province (BE2022612). The figures of this work were drawn by Figdraw (www.figdraw.com).
Author information
Authors and Affiliations
Contributions
Yuanhang Zhang: writing—original draft, literature search, and having the idea for the article. Kai Liu: data curation and literature search. Huan He: supervision and conceptualization. Hui Xiao: data curation. Zhihong Fang: data collection. Xianxian Chen: investigation. Huiming Li: writing—review and editing and funding acquisition. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
All authors approved the final manuscript and the submission to this journal.
Consent for publication
This research work does not contain any individual person’s data in the form of individual details, images, or videos. This work was based on published literature.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Lotfi Aleya
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, Y., Liu, K., He, H. et al. Innovative explorations: unveiling the potential of organoids for investigating environmental pollutant exposure. Environ Sci Pollut Res 31, 16256–16273 (2024). https://doi.org/10.1007/s11356-024-32256-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-024-32256-7