Abstract
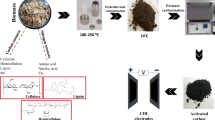
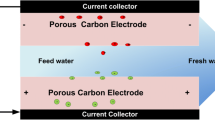
This study produced a biomass-based cylindrical electrode containing sucrose (an organic binder). The Cu2+ removal performance of the synthesized sucrose-bonded cylindrical electrode was evaluated in a 3-phase 3-dimensional electro-oxidation reactor (3D-EO) and the classical electro-oxidation method (2D-EO). Sodium Dodecyl Sulfate (SDs) was grafted onto activated carbon and used as microelectrode in 3D-EO reactors. SDs grafting resulted in a 57% reduction in the micropores of activated carbon. Therefore, the surface area of carbon after grafting decreased from 1328 m2/g to 580 m2/g. The sucrose-bonded cylindrical electrode has a rich carbon structure and consists of 84.04 wt% C, 12.10 wt% O and 3.20 wt%Si. According to CV measurement, the sucrose-bonded cylindrical electrode gives a surface reaction against Cu2+ at voltages lower than -0.62 V. Increasing the potential difference from 1V to 3V in 2D-EO and 3D-EO processes led to the removal of Cu2+ from the solution. The 3D-EO reactor achieved a removal rate of 87.12% at 3V. The 100 ppm solution was treated with a 3D-EO reactor containing 6 g/L of PC/SDs400Ws for 60 min, successfully removing 91.22% of Cu2+.








Similar content being viewed by others
Data availability
Not applicable
References
Ahn CK, Park D, Woo SH, Park JM (2009) Removal of cationic heavy metal from aqueous solution by activated carbon impregnated with anionic surfactants. J Hazard Mater 164:1130–1136. https://doi.org/10.1016/j.jhazmat.2008.09.036
Alslaibi TM, Abustan I, Ahmad MA, Foul AA (2013) Cadmium removal from aqueous solution using microwaved olive stone activated carbon. J Environ Chem Eng 1:589–599. https://doi.org/10.1016/j.jece.2013.06.028
Andrade AIASS, Stigter TY (2009) Multi-method assessment of nitrate and pesticide contamination in shallow alluvial groundwater as a function of hydrogeological setting and land use. Agric Water Manag 96:1751–1765. https://doi.org/10.1016/j.agwat.2009.07.014
Ansari T, Marr IL, Narejo N (2004) Heavy metals in marine pollution perspective-A mini review. J Appl Sci 4. https://doi.org/10.3923/jas.2004.1.20
Antonio Pineda Arellano C, Martínez SS (2007) Indirect electrochemical oxidation of cyanide by hydrogen peroxide generated at a carbon cathode. Int J Hydrog Energy 32:3163–3169. https://doi.org/10.1016/j.ijhydene.2006.04.011
Araya M, Olivares M, Pizarro F et al (2004) Community-based randomized double-blind study of gastrointestinal effects and copper exposure in drinking water. Environ Health Perspect 112:1068–1073. https://doi.org/10.1289/ehp.6913
Babar N, Joya KS, Ehsan MA et al (2019) Noble-metal-free colloidal-copper based Low overpotential water oxidation electrocatalyst. ChemCatChem 11:6022–6030. https://doi.org/10.1002/cctc.201900202
Bandmann O, Weiss KH, Kaler SG (2015) Wilson’s disease and other neurological copper disorders. Lancet Neurol 14:103–113. https://doi.org/10.1016/S1474-4422(14)70190-5
Biesinger MC (2017) Advanced analysis of copper X-ray photoelectron spectra: Advanced analysis of copper X-ray photoelectron spectra. Surf Interface Anal 49:1325–1334. https://doi.org/10.1002/sia.6239
Biesinger MC, Lau LWM, Gerson AR, Smart RStC (2010) Resolving surface chemical states in XPS analysis of first row transition metals, oxides and hydroxides: Sc, Ti, V, Cu and Zn. Appl Surf Sci 257:887–898. https://doi.org/10.1016/j.apsusc.2010.07.086
Boonamnuayvitaya V, Sae-ung S, Tanthapanichakoon W (2005) Preparation of activated carbons from coffee residue for the adsorption of formaldehyde. Sep Purif Technol 42:159–168. https://doi.org/10.1016/j.seppur.2004.07.007
Borandegi M, Nezamzadeh-Ejhieh A (2015) Enhanced removal efficiency of clinoptilolite nano-particles toward Co(II) from aqueous solution by modification with glutamic acid. Colloids Surf A Physicochem Eng Asp 479:35–45. https://doi.org/10.1016/j.colsurfa.2015.03.040
Carneiro PA, Umbuzeiro GA, Oliveira DP, Zanoni MVB (2010) Assessment of water contamination caused by a mutagenic textile effluent/dyehouse effluent bearing disperse dyes. J Hazard Mater 174:694–699. https://doi.org/10.1016/j.jhazmat.2009.09.106
Chen H, Feng Y, Suo N et al (2019) Preparation of particle electrodes from manganese slag and its degradation performance for salicylic acid in the three-dimensional electrode reactor (TDE). Chemosphere 216:281–288. https://doi.org/10.1016/j.chemosphere.2018.10.097
Cho S, Kim C, Hwang I (2020) Electrochemical degradation of ibuprofen using an activated-carbon-based continuous-flow three-dimensional electrode reactor (3DER). Chemosphere 259:127382. https://doi.org/10.1016/j.chemosphere.2020.127382
Costa CR, Montilla F, Morallón E, Olivi P (2009) Electrochemical oxidation of acid black 210 dye on the boron-doped diamond electrode in the presence of phosphate ions: Effect of current density, pH, and chloride ions. Electrochim Acta 54:7048–7055. https://doi.org/10.1016/j.electacta.2009.07.027
Demiral İ, Demiral H (2010) Surface characterization of activated carbons obtained from olive bagasse by chemical activation. Surf Interface Anal 42:1347–1350. https://doi.org/10.1002/sia.3294
Deng H, Yang L, Tao G, Dai J (2009) Preparation and characterization of activated carbon from cotton stalk by microwave assisted chemical activation—Application in methylene blue adsorption from aqueous solution. J Hazard Mater 166:1514–1521. https://doi.org/10.1016/j.jhazmat.2008.12.080
Derikvandi H, Nezamzadeh-Ejhieh A (2017) Increased photocatalytic activity of NiO and ZnO in photodegradation of a model drug aqueous solution: Effect of coupling, supporting, particles size and calcination temperature. J Hazard Mater 321:629–638. https://doi.org/10.1016/j.jhazmat.2016.09.056
Dickler E, Schafermeyer S (1993) Guidelines for ıntegrated production of pome fruits in Europe. Acta Hortic 83–96. 10.17660/ActaHortic.1993.347.9
D’Souza S, Miller JE, Ahn J et al (2019) The Antibiotic Trimethoprim Displays Strong Mutagenic Synergy with 2-Aminopurine. Antimicrob Agents Chemother 63:e01577–e01518. https://doi.org/10.1128/AAC.01577-18
Eshghi A, Kheirmand M (2017) Graphene/Ni–Fe layered double hydroxide nano composites as advanced electrode materials for glucose electro oxidation. Int J Hydrog Energy 42:15064–15072. https://doi.org/10.1016/j.ijhydene.2017.04.288
Foroughi M, Rahmani AR, Asgari G et al (2020) Optimization and modeling of tetracycline removal from wastewater by three-dimensional electrochemical system: application of response surface methodology and least squares support vector machine. Environ Model Assess 25:327–341. https://doi.org/10.1007/s10666-019-09675-9
García Torres E, Pérez Morales R, González Zamora A et al (2022) Consumption of water contaminated by nitrate and its deleterious effects on the human thyroid gland: a review and update. Int J Environ Health Res 32:984–1001. https://doi.org/10.1080/09603123.2020.1815664
Gottipati R, Mishra S (2016) Preparation of microporous activated carbon from Aegle Marmelos fruit shell and its application in removal of chromium(VI) from aqueous phase. J Ind Eng Chem 36:355–363. https://doi.org/10.1016/j.jiec.2016.03.005
Gray JP, Suhali-Amacher N, Ray SD (2017) Chapter 19 - Metals and Metal Antagonists. In: Ray SD (ed) Side Effects of Drugs Annual. Elsevier, pp 197–208
Harvey LJ, McArdle HJ (2008) Biomarkers of copper status: a brief update. Br J Nutr 99:S10–S13. https://doi.org/10.1017/S0007114508006806
Hassanien R, Almaky MM, Houlton A, Horrocks BR (2016) Preparation and electrical properties of a copper-conductive polymer hybrid nanostructure. RSC Adv 6:99422–99432. https://doi.org/10.1039/C6RA20325F
Heidari-Chaleshtori M, Nezamzadeh-Ejhieh A (2015) Clinoptilolite nano-particles modified with aspartic acid for removal of Cu(II) from aqueous solutions: isotherms and kinetic aspects. New J Chem 39:9396–9406. https://doi.org/10.1039/C5NJ01631B
Klink MJ, Makgae ME, Crouch AM (2010) Physico-chemical and electrochemical characterization of Ti/RhOx–IrO2 electrodes using sol–gel technology. Mater Chem Phys 124:73–77. https://doi.org/10.1016/j.matchemphys.2010.05.016
Kumar A, Sharma CB (1987) Hematological indices in copper-poisoned rats. Toxicol Lett 38:275–278. https://doi.org/10.1016/0378-4274(87)90009-9
Kumar PS, Raja MJS, Kumaresan M et al (2014) A new electrode reactor with in-built recirculation mode for the enhancement of methylene blue dye removal from the aqueous solution: Comparison of adsorption, electrolysis and combined effect. Korean J Chem Eng 31:276–283. https://doi.org/10.1007/s11814-013-0209-2
Kumar S, Singh S, Srivastava VC (2015) Electro-oxidation of nitrophenol by ruthenium oxide coated titanium electrode: Parametric, kinetic and mechanistic study. Chem Eng J 263:135–143. https://doi.org/10.1016/j.cej.2014.11.051
Lei J, Duan P, Liu W et al (2020) Degradation of aqueous cefotaxime in electro-oxidation — electro-Fenton —persulfate system with Ti/CNT/SnO2–Sb–Er anode and Ni@NCNT cathode. Chemosphere 250:126163. https://doi.org/10.1016/j.chemosphere.2020.126163
Li C, Zhou K, Qin W et al (2019) A Review on heavy metals contamination in soil: Effects, sources, and remediation techniques. Soil Sediment Contam Int J 28:380–394. https://doi.org/10.1080/15320383.2019.1592108
Li P, Karunanidhi D, Subramani T, Srinivasamoorthy K (2021) Sources and consequences of groundwater contamination. Arch Environ Contam Toxicol 80:1–10. https://doi.org/10.1007/s00244-020-00805-z
Liang S, Lin H, Yan X, Huang Q (2018) Electro-oxidation of tetracycline by a Magnéli phase Ti4O7 porous anode: Kinetics, products, and toxicity. Chem Eng J 332:628–636. https://doi.org/10.1016/j.cej.2017.09.109
Liu G, Chai X, Shao Y et al (2011) Toxicity of copper, lead, and cadmium on the motility of two marine microalgae Isochrysis galbana and Tetraselmis chui. J Environ Sci 23:330–335. https://doi.org/10.1016/S1001-0742(10)60410-X
Mackay DM, Roberts PV, Cherry JA (1985) Transport of organic contaminants in groundwater. Environ Sci Technol 19:384–392. https://doi.org/10.1021/es00135a001
Mainali K (2020) Phenolic Compounds Contaminants in Water: A Glance. CTCSE 4:. 10.33552/CTCSE.2020.04.000593
Mercer SW, Wang J, Burke R (2017) In vivo modeling of the pathogenic effect of copper transporter mutations that cause menkes and wilson diseases, motor neuropathy, and susceptibility to alzheimer’s disease. J Biol Chem 292:4113–4122. https://doi.org/10.1074/jbc.M116.756163
Momcilovic M, Purenovic M, Bojic A et al (2011) Removal of lead(II) ions from aqueous solutions by adsorption onto pine cone activated carbon. Desalination 276:53–59. https://doi.org/10.1016/j.desal.2011.03.013
Moradi SE (2014) Microwave assisted preparation of sodium dodecyl sulphate (SDS) modified ordered nanoporous carbon and its adsorption for MB dye. J Ind Eng Chem 20:208–215. https://doi.org/10.1016/j.jiec.2013.04.005
Moulya KP, Manjunatha JG, Aljuwayid AM et al (2023) Polymer modified carbon paste electrode for the electrochemical analysis of Tartrazine. Results Chem 5:100809. https://doi.org/10.1016/j.rechem.2023.100809
Munoz-Morales M, Saez C, Canizares P, Rodrigo MA (2020) Improvement of electrochemical oxidation efficiency through combination with adsorption processes. J Environ Manag 262:110364. https://doi.org/10.1016/j.jenvman.2020.110364
Nasiri-Ardali M, Nezamzadeh-Ejhieh A (2020) A comprehensive study on the kinetics and thermodynamic aspects of batch and column removal of Pb(II) by the clinoptilolite–glycine adsorbent. Mater Chem Phys 240:122142. https://doi.org/10.1016/j.matchemphys.2019.122142
Nezamzadeh-Ejhieh A, Moazzeni N (2013) Sunlight photodecolorization of a mixture of Methyl Orange and Bromocresol Green by CuS incorporated in a clinoptilolite zeolite as a heterogeneous catalyst. J Ind Eng Chem 19:1433–1442. https://doi.org/10.1016/j.jiec.2013.01.006
Nezamzadeh-Ejhieh A, Nematollahi Z (2011) Surfactant modified zeolite carbon paste electrode (SMZ-CPE) as a nitrate selective electrode. Electrochim Acta 56:8334–8341. https://doi.org/10.1016/j.electacta.2011.07.013
Niknezhadi A, Nezamzadeh-Ejhieh A (2017) A novel and sensitive carbon paste electrode with clinoptilolite nano-particles containing hexadecyltrimethyl ammonium surfactant and dithizone for the voltammetric determination of Sn(II). J Colloid Interface Sci 501:321–329. https://doi.org/10.1016/j.jcis.2017.04.068
Ntakirutimana S, Tan W, Wang Y (2019) Enhanced surface activity of activated carbon by surfactants synergism. RSC Adv 9:26519–26531. https://doi.org/10.1039/C9RA04521J
Ozcan L, Unlusoy B, Yurdakal S (2023) Voltammetric determination of arbutin using carbon paste electrode modified with low crystalline home-prepared rutile TiO2 nanoparticles. Mater Chem Phys 301:127588. https://doi.org/10.1016/j.matchemphys.2023.127588
Pal D, Maiti SK (2020) An approach to counter sediment toxicity by immobilization of heavy metals using waste fish scale derived biosorbent. Ecotoxicol Environ Saf 187:109833. https://doi.org/10.1016/j.ecoenv.2019.109833
Pan G, Jing X, Ding X et al (2019) Synergistic effects of photocatalytic and electrocatalytic oxidation based on a three-dimensional electrode reactor toward degradation of dyes in wastewater. J Alloys Compd 809:151749. https://doi.org/10.1016/j.jallcom.2019.151749
Pavithra KG, Jaikumar V, Kumar PS, Sundarrajan P (2020) Cleaner strategies on the effective elimination of toxic chromium from wastewater using coupled electrochemical/biological systems. Environ Prog Sustain Energy 39. https://doi.org/10.1002/ep.13399
Peryea FJ (2001) Heavy Metal Contamınatıon In Decıduous Tree Fruıt Orchards: Implıcatıons For Mıneral Nutrıent Management. Acta Hortic 564:31–39. https://doi.org/10.17660/ActaHortic.2001.564.2
Rawat K, Pathak B (2021) Chapter 3 - Contamination of groundwater by fly ash heavy metals at landfill sites. In: Ahamad A, Siddiqui SI, Singh P (eds) Contamination of Water. Academic Press, pp 31–48
Reddy YVM, Shin JH, Palakollu VN et al (2022) Strategies, advances, and challenges associated with the use of graphene-based nanocomposites for electrochemical biosensors. Adv Colloid Interf Sci 304:102664. https://doi.org/10.1016/j.cis.2022.102664
Ryoo R, Joo SH, Jun S (1999) Synthesis of highly ordered carbon molecular sieves via template-mediated structural transformation. J Phys Chem B 103:7743–7746. https://doi.org/10.1021/jp991673a
Saadat M, Nezamzadeh-Ejhieh A (2016) Clinoptilolite nanoparticles containing HDTMA and Arsenazo III as a sensitive carbon paste electrode modifier for indirect voltammetric measurement of Cesium ions. Electrochim Acta 217:163–170. https://doi.org/10.1016/j.electacta.2016.09.084
Saratale RG, Rajesh Banu J, Shin H-S, et al (2020) Textile Industry Wastewaters as Major Sources of Environmental Contamination: Bioremediation Approaches for Its Degradation and Detoxification. In: Saxena G, Bharagava RN (eds) Bioremediation of Industrial Waste for Environmental Safety: Volume I: Industrial Waste and Its Management. Springer, Singapore, pp 135–167
Schafer H, Wenzel A, Fritsche U et al (1993) Long-term effects of selected xenobiotica on freshwater green algae: development of a flow-through test system. Sci Total Environ 134:735–740. https://doi.org/10.1016/S0048-9697(05)80077-4
Shen DK, Gu S, Bridgwater AV (2010) Study on the pyrolytic behaviour of xylan-based hemicellulose using TG–FTIR and Py–GC–FTIR. J Anal Appl Pyrolysis 87:199–206. https://doi.org/10.1016/j.jaap.2009.12.001
Shetti NP, Malode SJ, Bukkitgar SD et al (2019) Electro-oxidation and determination of nimesulide at nanosilica modified sensor. Mater Sci Energy Technol 2:396–400. https://doi.org/10.1016/j.mset.2019.03.005
Singla S, Sharma S, Basu S et al (2021) Photocatalytic water splitting hydrogen production via environmental benign carbon based nanomaterials. Int J Hydrog Energy 46:33696–33717. https://doi.org/10.1016/j.ijhydene.2021.07.187
Stern BR (2010) Essentiality and Toxicity in Copper Health Risk Assessment: Overview, Update and Regulatory Considerations. J Toxic Environ Health A 73:114–127. https://doi.org/10.1080/15287390903337100
Su C, Jiang L, Zhang W (2014) A review on heavy metal contamination in the soil worldwide: Situation, impact and remediation techniques
Sun W, Sun Y, Shah KJ et al (2019) Electrocatalytic oxidation of tetracycline by Bi-Sn-Sb/γ-Al2O3 three-dimensional particle electrode. J Hazard Mater 370:24–32. https://doi.org/10.1016/j.jhazmat.2018.09.085
Szpyrkowicz L, Kaul SN, Neti RN, Satyanarayan S (2005) Influence of anode material on electrochemical oxidation for the treatment of tannery wastewater. Water Res 39:1601–1613. https://doi.org/10.1016/j.watres.2005.01.016
Tamiji T (2018) A comprehensive study on the kinetic aspects and experimental design for the voltammetric response of a Sn(IV)-clinoptilolite carbon paste electrode towards Hg(II). J Electroanal Chem
Tchounwou PB, Yedjou CG, Patlolla AK, Sutton DJ (2012) Heavy Metal Toxicity and the Environment. In: Luch A (ed) Molecular, Clinical and Environmental Toxicology. Springer Basel, Basel, pp 133–164
Teshale F, Karthikeyan R, Sahu O (2020) Synthesized bioadsorbent from fish scale for chromium (III) removal. Micron 130:102817. https://doi.org/10.1016/j.micron.2019.102817
Vasconcelos VM, Ponce-de-León C, Nava JL, Lanza MRV (2016) Electrochemical degradation of RB-5 dye by anodic oxidation, electro-Fenton and by combining anodic oxidation–electro-Fenton in a filter-press flow cell. J Electroanal Chem 765:179–187. https://doi.org/10.1016/j.jelechem.2015.07.040
Wang T, Song Y, Ding H et al (2020) Insight into synergies between ozone and in-situ regenerated granular activated carbon particle electrodes in a three-dimensional electrochemical reactor for highly efficient nitrobenzene degradation. Chem Eng J 394:124852. https://doi.org/10.1016/j.cej.2020.124852
Wang X, Zhang B, Zhang W et al (2017) Super-light Cu@Ni nanowires/graphene oxide composites for significantly enhanced microwave absorption performance. Sci Rep 7:1584. https://doi.org/10.1038/s41598-017-01529-2
World Health Organization (2008) Chemical Fact Sheets
World Health Organization (2022) Guidelines for drinking-water quality: fourth edition incorporating the first and second addenda. World Health Organization
World Health Organization (ed) (2004) Guidelines for drinking-water quality, 3rd edn. World Health Organization, Geneva
Wu X, Song X, Chen H, Yu J (2021) Treatment of phenolic compound wastewater using CuFe2O4/Al2O3 particle electrodes in a three-dimensional electrochemical oxidation system. Environ Technol 42:4393–4404. https://doi.org/10.1080/09593330.2020.1760356
Wu Z, Liu Y, Wang S et al (2019) A novel integrated system of three-dimensional electrochemical reactors (3DERs) and three-dimensional biofilm electrode reactors (3DBERs) for coking wastewater treatment. Bioresour Technol 284:222–230. https://doi.org/10.1016/j.biortech.2019.03.123
Zhang Y, Chang G, Shu H et al (2014) Synthesis of Pt–Pd bimetallic nanoparticles anchored on graphene for highly active methanol electro-oxidation. J Power Sources 262:279–285. https://doi.org/10.1016/j.jpowsour.2014.03.127
Zheng T, Wang Q, Shi Z et al (2016) Advanced treatment of wet-spun acrylic fiber manufacturing wastewater using three-dimensional electrochemical oxidation. J Environ Sci 50:21–31. https://doi.org/10.1016/j.jes.2016.03.020
Acknowledgements
This study has been funded by the Scientific Research Foundation, Eskisehir Osmangazi University under grant numbers FBA-2021-1591.
Funding
The author declares that this study has been funded by the Scientific Research Foundation, Eskisehir Osmangazi University under grant numbers FBA-2021-1591.
Author information
Authors and Affiliations
Contributions
Material preparation, data collection, analysis, execution of experiments and manuscript were carried out by Canan SAMDAN.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose
Ethics approval
All ethical rules have been read and applied as written.
Consent to participate
The study does not contain any information that would require consent from any individual.
Consent for publication
The study is not a case study. It does not contain information that would require an individual's consent.
Additional information
Responsible Editor: Guilherme Luiz Dotto
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 867 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Samdan, C. Synthesis and characterization of cylindrical electrode with sucrose binder as advanced electrode materials for copper 3D-electro-oxidation. Environ Sci Pollut Res 30, 99511–99528 (2023). https://doi.org/10.1007/s11356-023-29388-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-29388-7