Abstract
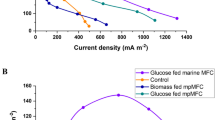
This study investigated the effect of co-culturing the photobiont and mycobiont in the microbial fuel cell (MFC) cathode on biomass production, lipid generation, and power output. Chlorella vulgaris provides oxygen and nutrients for the yeast Cystobasidium oligophagum JRC1, while the latter offers CO2 and quench oxygen for higher algal growth. The MFC with co-culture enhanced the lipid output of biomass by 28.33%, and the total yield and productivity were 1.47 ± 0.18 g/l and 0.123 g/l/day, respectively. Moreover, with co-culture, the open circuit voltage of 685 ± 11 mV was two times higher than algae alone. The specific growth rate (day−1) at the cathode was 0.367 ± 0.04 in co-culture and 0.288 ± 0.05 with C. vulgaris only. The power density of the system was 5.37 ± 0.21 mW/m2 with 75.88 ± 1.89% of COD removal. The co-culture thus proved beneficial at the MFC cathode in terms of total energy output as 11.5 ± 0.035 kWh/m3, which was 1.4-fold higher than algae alone.





Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Abed RM (2010) Interaction between cyanobacteria and aerobic heterotrophic bacteria in the degradation of hydrocarbons. Int Biodeterior Biodegradation 64(1):58–64. https://doi.org/10.1016/j.ibiod.2009.10.008
Arora N, Patel A, Mehtani J, Pruthi PA, Pruthi V, Poluri KM (2019) Co-culturing of oleaginous microalgae and yeast: a paradigm shift towards enhanced lipid productivity. Environ Sci Pollut Res 26:6952–16973. https://doi.org/10.1007/s11356-019-05138-6
Bligh EG, Dyer WJ (1959) A rapid total lipid extraction and purification method. Can J Biochem Physiol 37(8):911–917. https://doi.org/10.1139/o59-099
Chaijak P, Sukkasem C, Lertworapreecha M, Boonsawang P, Wijasika S, Sato C (2018) Enhancing electricity generation using a laccase-based microbial fuel cell with yeast Galactomyces reessii on the cathode. J Microbiol Biotechnol 28(8):1360–1366. https://doi.org/10.4014/jmb.1803.03015
Cheirsilp B, Suwannarat W, Niyomdecha R (2011) Mixed culture of oleaginous yeast Rhodotorula glutinis and microalga Chlorella vulgaris for lipid production from industrial wastes and its use as biodiesel feedstock. New Biotechnol 28(4):362–368. https://doi.org/10.1016/j.nbt.2011.01.004
Das S, Nath K, Chowdhury R (2021) Comparative studies on biomass productivity and lipid content of novel blue-green algae during autotrophic and heterotrophic growth. Environ Sci Pollut Res 28:12107–12118. https://doi.org/10.1007/s11356-020-09577-4
Dumas C, Basseguy R, Bergel A (2008) DSA to grow electrochemically active biofilms of Geobacter sulfurreducens. Electrochim Acta 53(7):3200–3209. https://doi.org/10.1016/j.electacta.2007.10.066
Escher A, Characklis WG (1982) Algal-bacterial interactions within aggregates. Biotechnol Bioeng 24:2283–2290. https://doi.org/10.1002/bit.260241017
Ewusi-Mensah D, Huang J, Chaparro LK, Rodenas P, Ramírez-Moreno M, Ortiz JM, Esteve-Núñez A (2021) Algae-assisted microbial desalination cell. Analysis of cathode performance and desalination efficiency assessment. Processes 9(11):2011. https://doi.org/10.3390/pr9112011
Ge Z, Zhang F, Grimaud J, Hurst J, He Z (2013) Long-term investigation of microbial fuel cells treating primary sludge or digested sludge. Bioresour Technol 136:509–514. https://doi.org/10.1016/j.biortech.2013.03.016
Ghasemi M, Daud WRW, Ismail AF, Jafari Y, Ismail M, Mayahi A, Othman J (2013) Simultaneous wastewater treatment and electricity generation by microbial fuel cell: performance comparison and cost investigation of using Nafion 117 and SPEEK as separators. Desalination 325:1–6. https://doi.org/10.1016/j.desal.2013.06.013
Islam MA, Ethiraj B, Cheng CK, Yousuf A, Thiruvenkadam S, Prasad R, Rahman Khan MM (2018) Enhanced current generation using mutualistic interaction of yeast-bacterial coculture in dual chamber microbial fuel cell. Ind Eng Chem Res 57(3):813–821. https://doi.org/10.1021/acs.iecr.7b01855
Karmakar B, Mukherjee S, Sengupta SL, Halder G (2021) Recirculating used cooking oil and Nagkesar seed shells in dual-stage catalytic biodiesel synthesis with C1-C3 alcohols. Environ Sci Pollut Res 28(41):58154–58169. https://doi.org/10.1007/s11356-021-14309-3
Khandelwal A, Vijay A, Dixit A, Chhabra M (2018) Microbial fuel cell powered by lipid extracted algae: a promising system for algal lipids and power generation. Bioresour Technol 247:520–527. https://doi.org/10.1016/j.biortech.2017.09.119
Khandelwal A, Chhabra M, Yadav P (2020) Performance evaluation of algae assisted microbial fuel cell under outdoor conditions. Bioresour Technol 310:123418. https://doi.org/10.1016/j.biortech.2020.123418
Lefebvre O, Tan Z, Kharkwal S, Ng HY (2012) Effect of increasing anodic NaCl concentration on microbial fuel cell performance. Bioresour Technol 112:336–340. https://doi.org/10.1016/j.biortech.2012.02.048
Li T, Li CT, Butler K, Hays SG, Guarnieri MT, Oyler GA, Betenbaugh MJ (2017) Mimicking lichens: incorporation of yeast strains together with sucrose-secreting cyanobacteria improves survival, growth, ROS removal, and lipid production in a stable mutualistic co-culture production platform. Biotechnol Biofuels 10(1):1–11. https://doi.org/10.1186/s13068-017-0736-x
Li Y, Zhao ZK, Bai F (2007) High-density cultivation of oleaginous yeast Rhodosporidium toruloides Y4 in fed-batch culture. Enzyme Microb Technol 41(3):312–317. https://doi.org/10.1016/j.enzmictec.2007.02.008
Liu L, Chen J, Lim PE, Wei D (2018) Enhanced single cell oil production by mixed culture of Chlorella pyrenoidosa and Rhodotorula glutinis using cassava bagasse hydrolysate as carbon source. Bioresour Technol 255:40–148. https://doi.org/10.1016/j.biortech.2018.01.114
Logan BE, Hamelers B, Rozendal R, Schröder U, Keller J, Freguia S, Rabaey K (2006) Microbial fuel cells: methodology and technology. Environ Sci Technol 40(17):5181–5192. https://doi.org/10.1021/es0605016
McFeters GA, Stuart SA, Olson SB (1978) Growth of heterotrophic bacteria and algal extracellular products in oligotrophic waters. Appl Environ Microbial 35:383–391. https://doi.org/10.1128/aem.35.2.383-391.1978
Mohan SV, Raghavulu SV, Sarma PN (2008) Influence of anodic biofilm growth on bioelectricity production in a single chambered mediator less microbial fuel cell using mixed anaerobic consortia. Biosens Bioelectron 24(1):41–47. https://doi.org/10.1016/j.bios.2008.03.010
Mouget JL, Dakhama A, Lavoie MC, de la Noüe J (1995) Algal growth enhancement by bacteria: is consumption of photosynthetic oxygen involved? FEMS Microbiol Ecol 18(1):35–43. https://doi.org/10.1111/j.1574-6941.1995.tb00159.x
Mishra A, Chhabra M (2022) Performance of photo-microbial fuel cell with Dunaliella salina at the saline cathode. Bioresour Technol Rep 19:101199. https://doi.org/10.1016/j.biteb.2022.101199
Ou X, Yan X, Zhang X, Zhang X (2013) Life-cycle energy use and greenhouse gas emissions analysis for bio-liquid jet fuel from open pond-based micro-algae under China conditions. Energies 6(9):4897–4923. https://doi.org/10.3390/en6094897
Park Y, Je KW, Lee K, Jung SE, Choi TJ (2008) Growth promotion of Chlorella ellipsoidea by co-inoculation with Brevundimonas sp. isolated from the microalga. Hydrobiologia 598(1):219–228. https://doi.org/10.1007/s10750-007-9152-8
Raposo F, De la Rubia MA, Borja R, Alaiz M (2008) Assessment of a modified and optimized method for determining chemical oxygen demand of solid substrates and solutions with high suspended solid content. Talanta 76(2):448–453. https://doi.org/10.1016/j.talanta.2008.03.030
Rasouli Z, Valverde-Pérez B, D’Este M, De Francisci D, Angelidaki I (2018) Nutrient recovery from industrial wastewater as single cell protein by a co-culture of green microalgae and methanotrophs. Biochem Eng J 134:129–135. https://doi.org/10.1016/j.bej.2018.03.010
Sarpal AS, Silva PR, Martins JL, Amaral JJ, Monnerat MM, Cunha VS, de Souza W (2014) Biodiesel potential of oleaginous yeast biomass by NMR spectroscopic techniques. Energy fuels 28(6):3766–3777. https://doi.org/10.1021/ef402516x
Schneider K, Thorne RJ, Cameron PJ (2016) An investigation of anode and cathode materials in photo microbial fuel cells. Philosophical Transactions of the Royal Society A: Mathematical. Phys. Eng Sci 374(2061):20150080. https://doi.org/10.1098/rsta.2015.0080
Schröder U (2007) Anodic electron transfer mechanisms in microbial fuel cells and their energy efficiency. Phys Chem Chem Phys 9(21):2619–2629. https://doi.org/10.1039/b703627m
Sun H, Gao Z, Zhang L, Wang X, Gao M, Wang Q (2023) A comprehensive review of microbial lipid production from wastes: research updates and tendencies. Environ Sci Pollut Res 1–22. https://doi.org/10.1007/s11356-023-28123-6
Takaya N, Antonina MB, Sakairi C, Sakaguchi Y, Kato I, Zhou Z, Shoun H (2003) Aerobic denitrifying bacteria that produce low levels of nitrous oxide. Appl Environ Microbiol 69:3152–3157. https://doi.org/10.1128/aem.69.6.3152-3157.2003
Tariq M, Ali S, Ahmad F, Ahmad M, Zafar M, Khalid N, Khan MA (2011) Identification, FT-IR, NMR (1H and 13C) and GC/MS studies of fatty acid methyl esters in biodiesel from rocket seed oil. Fuel Process Technol 92(3):336–341. https://doi.org/10.1016/j.fuproc.2010.09.025
Vijay A, Vaishnava M, Chhabra M (2016) Microbial fuel cell assisted nitrate nitrogen removal using cow manure and soil. Environ Sci Pollut Res 23:7744–7756. https://doi.org/10.1007/s11356015-5934-0
Vijay A, Arora S, Gupta S, Chhabra M (2018) Halophilic starch degrading bacteria isolated from Sambhar Lake, India, as potential anode catalyst in microbial fuel cell: a promising process for saline water treatment. Bioresour Technol 256:391–398. https://doi.org/10.1016/j.biortech.2018.02.044
Vyas S, Chhabra M (2017) Isolation, identification and characterization of Cystobasidium oligophagum JRC1: a cellulase and lipase producing oleaginous yeast. Bioresour Technol 223:250–258. https://doi.org/10.1016/j.biortech.2016.10.039
Vyas S, Chhabra M (2019) Assessing oil accumulation in the oleaginous yeast Cystobasidium oligophagum JRC1 using dairy waste cheese whey as a substrate. 3 Biotech 9:1–13. https://doi.org/10.1007/s13205-019-1701-0
Wágner DS, Valverde-Perez B, Sæbø M, de la Sotilla MB, Van Wagenen J, Smets BF, Plosz BG (2016) Towards a consensus-based biokinetic model for green microalgae: the ASM-A. Water Res 103:485–499. https://doi.org/10.1016/j.watres.2016.07.026
Wang S, Wu Y, Wang X (2016) Heterotrophic cultivation of Chlorella pyrenoidosa using sucrose as the sole carbon source by co-culture with Rhodotorula glutinis. Bioresour Technol 220:615–620. https://doi.org/10.1016/j.biortech.2016.09.010
Watanabe K, Takihana N, Aoyagi H, Hanada S, Watanabe Y, Ohmura N, Tanaka H (2005) Symbiotic association in Chlorella culture. FEMS Microbiol Ecol 51(2):187–196. https://doi.org/10.1016/j.femsec.2004.08.004
Acknowledgements
The authors thank the Ministry of Human Resource and Development for the fellowship and the Indian Institute of Technology Jodhpur (IITJ) for its research facilities.
Author information
Authors and Affiliations
Contributions
Both the authors contributed to the study’s conception and design. Material preparation, data collection, and analysis were performed by Akanksha Mishra. The first draft of the manuscript was written by Akanksha Mishra, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. The project was supervised by Meenu Chhabra.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable
Consent for publication
Not applicable
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Ta Yeong Wu
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mishra, ., Chhabra, M. Co-culturing Chlorella vulgaris and Cystobasidium oligophagum JRC1 in the microbial fuel cell cathode for lipid biosynthesis. Environ Sci Pollut Res (2023). https://doi.org/10.1007/s11356-023-29232-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11356-023-29232-y