Abstract
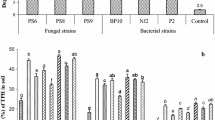
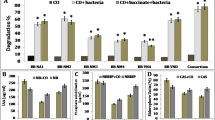
The present study investigated the concerted effort of Eisenia fetida and rhamnolipid JBR-425 in combination with a five-member bacterial consortium exhibiting elevated degradation levels of low and high molecular weight polycyclic aromatic hydrocarbons (PAH) from soil contaminated with Digboi crude oil. Application of bacterial consortium (G2) degraded 30–89% of selected PAH from the artificial soil after a 45-day post-exposure, in which chrysene showed the highest level of degradation with 89% and benzo(a)pyrene is the lowest with 30%, respectively. Moreover, an acute exposure study observed that earthworm biomass decreased, and mortality rates increased with increasing crude oil concentrations (0.25 to 2%). Earthworms with a 100% survival rate at 1% crude oil exposure suggest the tolerance potential and its mutual involvement in the bioremediation of crude oil with selected bacterial consortia. Bacterial consortium assisted with E. fetida (G3) showed 98% chrysene degradation with a slight change in benzo(a)pyrene degradation (35%) in crude oil spiked soil. Besides, the most dominant PAH in crude oil found in the current work, fluoranthene, undergoes 93% and 70% degradation in G3 and G5 groups, respectively. However, rhamnolipid JBR-425 coupled with the bacterial consortium (G5) has resulted in 97% degradation of chrysene and 33% for benzo(a)pyrene. Overall, bacterial consortium assisted with earthworm group has shown better degradation of selected PAH than bacterial consortium with biosurfactant. Catalase (CAT), glutathione reductase (GST) activity and MDA content was found to be reduced in earthworms after sub-lethal exposure, suggesting oxidative stress prevalence via reactive oxygen species (ROS). Hence, the findings of the present work suggest that the application of a bacterial consortium, along with earthworm E. fetida, has huge potential for field restoration of contaminated soil with PAH and ecosystem sustainability.




Similar content being viewed by others
Data availability
The raw data used and analysed during the present work are available from the corresponding author on request.
References
Abdel-Shafy HI, Mansour MSM (2016) A review on polycyclic aromatic hydrocarbons: source, environmental impact, effect on human health and remediation. Egypt J Pet 25(1):107-123. https://doi.org/10.1016/j.ejpe.2015.03.011
Adeyi AA, Oyeleke P (2017) Heavy metals and polycyclic aromatic hydrocarbons in soil from e-waste dumpsites in Lagos and Ibadan, Nigeria. J Heal Pollut. https://doi.org/10.5696/2156-9614-7.15.71
Aebi H (1984) Catalase in vitro. In Methods in enzymology (Vol. 105, pp. 121-126). Academic press. https://doi.org/10.1016/S0076-6879(84)05016-3
Almutairi M (2019) Vermiremediation strategy for remediation of Kuwaiti oil contaminated soil. SN Appl Sci. https://doi.org/10.1007/s42452-019-1297-3
Asgharnia H, Jafari AJ, Kalantary RR et al (2014) Influence of bioaugmentation on biodegradation of phenanthrene-contaminated soil by earthworm in lab scale. J Environ Heal Sci Eng. https://doi.org/10.1186/s40201-014-0150-2
Bernard JM (1963) Forest floor moisture capacity of the New Jersey pine barrens. Ecology. https://doi.org/10.2307/1932538
Bing XIA, Huang D, Mao YE, Hao QIU, Hongfeng CHEN, Keqiang ZHAO, Rongliang QIU, Rongrong YING (2022) Effects of rhamnolipids on bacterial communities in contaminated soil and earthworm guts. Pedosphere. https://doi.org/10.1016/j.pedsph.2022.11.005
Caliani I, Campani T, Conti B et al (2021) Multi-biomarker approach and IBR index to evaluate the effects of different contaminants on the ecotoxicological status of Apis mellifera. Ecotoxicol Environ Saf 208. https://doi.org/10.1016/j.ecoenv.2020.111486
Ceccanti B, Masciandaro G, Garcia C et al (2006) Soil bioremediation: combination of earthworms and compost for the ecological remediation of a hydrocarbon polluted soil. Water Air Soil Pollut. https://doi.org/10.1007/s11270-006-9180-4
Enerijiofi KE (2021) Bioremediation of environmental contaminants: a sustainable alternative to environmental management. In: Bioremediation for Environmental Sustainability. https://doi.org/10.1016/B978-0-12-820524-2.00019-5
Fischer J, Talal GDA, Schnee LS et al (2022) Clay types modulate the toxicity of low concentrated copper oxide nanoparticles toward springtails in artificial test soils. Environ Toxicol Chem 41(10):2454-2465. https://doi.org/10.1002/etc.5440
Freitas JS, Teresa FB, de Almeida EA (2017) Influence of temperature on the antioxidant responses and lipid peroxidation of two species of tadpoles (Rhinella schneideri and Physalaemus nattereri) exposed to the herbicide sulfentrazone (Boral 500SC®). Comp Biochem Physiol Part - C Toxicol Pharmacol. https://doi.org/10.1016/j.cbpc.2017.04.005
Gautam K, Seth M, Dwivedi S et al (2022) Soil degradation kinetics of oxybenzone (benzophenone-3) and toxicopathological assessment in the earthworm, Eisenia fetida. Environ Res 213:113689. https://doi.org/10.1016/j.envres.2022.113689
Gautam K, Anbumani S (2020) Ecotoxicological effects of organic micro-pollutants on the environment. Curr Dev Biotechnol Bioeng 481–501. https://doi.org/10.1016/B978-0-12-819594-9.00019-X
Ghavidel A, Rad SN, Alikhani HA et al (2018) Presence of Eisenia fetida enhanced phytoremediation of anthracene by Lolium perenne. Biosci J. https://doi.org/10.14393/BJ-v34n1a2018-39861
Ghosal D, Ghosh S, Dutta TK, Ah Y (2016) Current state of knowledge in microbial degradation of polycyclic aromatic hydrocarbons (PAHs): a review. Front Microbiol. https://doi.org/10.3389/fmicb.2016.01369
Govindasamy P, Mahawer SK, Mowrer J, Bagavathiannan M, Prasad M, Ramakrishnan S, Chandra A (2023) Comparison of Low-Cost Methods for Soil Water Holding Capacity. Commun Soil Sci Plant Analysis. https://doi.org/10.1080/00103624.2022.2112216
Hennessee CT, Li QX (2016) Effects of polycyclic aromatic hydrocarbon mixtures on degradation, gene expression, and metabolite production in four Mycobacterium species. Appl Environ Microbiol. https://doi.org/10.1128/AEM.00100-16
Jiang Y, Wang J, Muhammad S et al (2018) How do earthworms affect decomposition of residues with different quality apart from fragmentation and incorporation? Geoderma. https://doi.org/10.1016/j.geoderma.2018.04.013
Joy S, Rahman PK, Sharma S (2017) Biosurfactant production and concomitant hydrocarbon degradation potentials of bacteria isolated from extreme and hydrocarbon contaminated environments. Chem Eng J. https://doi.org/10.1016/j.cej.2017.02.054
Khomarbaghi Z, Shavandi M, Amoozegar MA, Dastgheib SMM (2019) Bacterial community dynamics during bioremediation of alkane-and PAHs-contaminated soil of Siri Island, Persian Gulf: a microcosm study. Int J Environ Sci Technol. https://doi.org/10.1007/s13762-018-02198-y
Koolivand A, Saeedi R, Coulon F et al (2020) Bioremediation of petroleum hydrocarbons by vermicomposting process bioaugmentated with indigenous bacterial consortium isolated from petroleum oily sludge. Ecotoxicol Environ Saf. https://doi.org/10.1016/j.ecoenv.2020.110645
Kumari S, Regar RK, Manickam N (2018) Improved polycyclic aromatic hydrocarbon degradation in a crude oil by individual and a consortium of bacteria. Bioresour Technol. https://doi.org/10.1016/j.biortech.2018.01.075
Kumari S, Rajput VD, Sushkova S, Minkina T (2022) Microbial electrochemical system: An emerging technology for remediation of polycyclic aromatic hydrocarbons from soil and sediments. Environ Geochem Health. https://doi.org/10.1007/s10653-022-01356-z
Li M, Ma X, Saleem M et al (2020a) Biochemical response, histopathological change and DNA damage in earthworm (Eisenia fetida) exposed to sulfentrazone herbicide. Ecol Indic 115. https://doi.org/10.1016/j.ecolind.2020.106465
Li Y, Wang X, Sun Z (2020b) Ecotoxicological effects of petroleum-contaminated soil on the earthworm Eisenia fetida. J Hazard Mater 393. https://doi.org/10.1016/j.jhazmat.2020.122384
Liu T, Wang X, Chen D et al (2018) Growth, reproduction and biochemical toxicity of chlorantraniliprole in soil on earthworms (Eisenia fetida). Ecotoxicol Environ Saf. https://doi.org/10.1016/j.ecoenv.2017.12.010
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem. https://doi.org/10.1016/s0021-9258(19)52451-6
Manickam N, Bajaj A, Saini HS, Shanker R (2012) Surfactant mediated enhanced biodegradation of hexachlorocyclohexane (HCH) isomers by Sphingomonas sp. NM05. Biodegradation. https://doi.org/10.1007/s10532-012-9543-z
Mohanty I, Rath A (2020) Bioremediation: current research trends and applications. In: Waste to energy: prospects and applications. https://doi.org/10.1007/978-981-33-4347-4_5
Muller EB, Nisbet RM, Berkley HA (2010) Sublethal toxicant effects with dynamic energy budget theory: model formulation. Ecotoxicology. https://doi.org/10.1007/s10646-009-0385-3
Mulligan CN, Yong RN, Gibbs BF (2001) Surfactant-enhanced remediation of contaminated soil: a review. Eng Geol. https://doi.org/10.1016/S0013-7952(00)00117-4
Muñiz S, Gonzalvo P, Valdehita A et al (2017) Ecotoxicological assessment of soils polluted with chemical waste from lindane production: use of bacterial communities and earthworms as bioremediation tools. Ecotoxicol Environ Saf. https://doi.org/10.1016/j.ecoenv.2017.07.070
Nagla AMA, Sulieman HNA, Ahmed MAH et al (2020) Bacterial and vermi-remediation of soil contaminated with chlorpyrifos insecticide. African J Biotechnol. https://doi.org/10.5897/ajb2019.16962
Nam TH, Jeon HJ, Mo Hh et al (2015) Determination of biomarkers for polycyclic aromatic hydrocarbons (PAHs) toxicity to earthworm (Eisenia fetida). Environ Geochem Health. https://doi.org/10.1007/s10653-015-9706-z
OECD (Organisation for Economic Co-operation and Development) (1984) Earthworm, acute toxicity tests. Guideline for Testing Chemicals (207)
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem. https://doi.org/10.1016/0003-2697(79)90738-3
Okoh E, Yelebe ZR, Oruabena B et al (2020) Clean-up of crude oil-contaminated soils: bioremediation option. Int J Environ Sci Technol. https://doi.org/10.1007/s13762-019-02605-y
Ossai IC, Ahmed A, Hassan A, Hamid FS (2020) Remediation of soil and water contaminated with petroleum hydrocarbon: a review. Environ Technol Innov. https://doi.org/10.1016/j.eti.2019.100526
Pande V, Pandey SC, Sati D et al (2020) Bioremediation: an effective emerging approach towards environment restoration. Environ Sustain. https://doi.org/10.1007/s42398-020-00099-w
Patel AB, Shaikh S, Jain KR et al (2020) Polycyclic aromatic hydrocarbons: sources, toxicity, and remediation approaches. Front Microbiol. https://doi.org/10.3389/fmicb.2020.562813
Ramadass K, Megharaj M, Venkateswarlu K, Naidu R (2017) Ecotoxicity of measured concentrations of soil-applied diesel: effects on earthworm survival, dehydrogenase, urease and nitrification activities. Appl Soil Ecol. https://doi.org/10.1016/j.apsoil.2017.05.017
Rasheed T, Shafi S, Bilal M et al (2020) Surfactants-based remediation as an effective approach for removal of environmental pollutants—a review. J Mol Liq. https://doi.org/10.1016/j.molliq.2020.113960
Ravindra K, Sokhi R, Van Grieken R (2008) Atmospheric polycyclic aromatic hydrocarbons: source attribution, emission factors and regulation. Atmos Environ. https://doi.org/10.1016/j.atmosenv.2007.12.010
Rodrigues AP, Lehtonen KK, Guilhermino L, Guimarães L (2013) Exposure of Carcinus maenas to waterborne fluoranthene: accumulation and multibiomarker responses. Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2012.10.077
Rong H, Wang C, Liu H et al (2020) Biochemical toxicity and potential detoxification mechanisms in earthworms Eisenia fetida exposed to sulfamethazine and copper. Bull Environ Contam Toxicol. https://doi.org/10.1007/s00128-020-02927-5
Saint-Denis M, Narbonne JF, Arnaud C, Ribera D (2001) Biochemical responses of the earthworm Eisenia fetida andrei exposed to contaminated artificial soil: effects of lead acetate. Soil Biol Biochem. https://doi.org/10.1016/S0038-0717(00)00177-2
Sammarco PW, Kolian SR, Warby RAF et al (2013) Distribution and concentrations of petroleum hydrocarbons associated with the BP/Deepwater Horizon Oil Spill, Gulf of Mexico. Mar Pollut Bull. https://doi.org/10.1016/j.marpolbul.2013.05.029
Sanchez W, Burgeot T, Porcher JM (2013) A novel “Integrated Biomarker Response” calculation based on reference deviation concept. Environ Sci Pollut Res 20:2721–2725. https://doi.org/10.1007/s11356-012-1359-1
Shakir Hanna SH, Weaver RW (2002) Earthworm survival in oil contaminated soil. Plant Soil. https://doi.org/10.1023/A:1015816315477
Shi Z, Wang C, Zhao Y (2020) Effects of surfactants on the fractionation, vermiaccumulation, and removal of fluoranthene by earthworms in soil. Chemosphere. https://doi.org/10.1016/j.chemosphere.2020.126332
Singh A, Singh DP, Tiwari R et al (2015) Taxonomic and functional annotation of gut bacterial communities of Eisenia foetida and Perionyx excavatus. Microbiol Res. https://doi.org/10.1016/j.micres.2015.03.003
Singh J (2018) 3 - Role of earthworm in sustainable agriculture. In: Galanakis CMBT-SFS from A to I (ed). Academic Press. https://doi.org/10.1016/B978-0-12-811935-8.00003-2
Sinha RK, Bharambe G, Ryan D (2008) Converting wasteland into wonderland by earthworms - a low-cost nature’s technology for soil remediation: a case study of vermiremediation of PAHs contaminated soil. Environmentalist. https://doi.org/10.1007/s10669-008-9171-7
Song JA, Choi CY (2021) Exposure to benzo[α]pyrene causes oxidative stress and cell damage in bay scallop Argopecten irradians. Aquac Reports. https://doi.org/10.1016/j.aqrep.2021.100860
Sonune N (2021) Microbes: a potential tool for bioremediation. In: Rhizobiont in bioremediation of hazardous waste. https://doi.org/10.1007/978-981-16-0602-1_17
Tang J, Wang M, Wang F et al (2011) Ecotoxicity of petroleum hydrocarbon contaminated soil. J Environ Sci. https://doi.org/10.1016/S1001-0742(10)60517-7
Tripathi V, Gaur VK, Dhiman N et al (2019) Characterisation and properties of the biosurfactant produced by PAH-degrading bacteria isolated from contaminated oily sludge environment. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-019-05591-3
Van Herwijnen R, Wattiau P, Bastiaens L et al (2003) Elucidation of the metabolic pathway of fluorene and cometabolic pathways of phenanthrene, fluoranthene, anthracene and dibenzothiophene by Sphingomonas sp. LB126. Res Microbiol. https://doi.org/10.1016/S0923-2508(03)00039-1
van Gestel CA (2012) Soil ecotoxicology: state of the art and future directions. ZooKeys. https://doi.org/10.3897/zookeys.176.2275
Varjani S, Upasani VN (2019) Evaluation of rhamnolipid production by a halotolerant novel strain of Pseudomonas aeruginosa. Bioresour Technol. https://doi.org/10.1016/j.biortech.2019.121577
Wang H, Zhang X, Wang L et al (2020a) Biochemical responses and DNA damage induced by herbicide QYR301 in earthworm (Eisenia fetida). Chemosphere. https://doi.org/10.1016/j.chemosphere.2019.125512
Wang T, Su D, Wang X, He Z (2020b) Adsorption-degradation of polycyclic aromatic hydrocarbons in soil by immobilized mixed bacteria and its effect on microbial communities. J Agric Food Chem. https://doi.org/10.1021/acs.jafc.0c04752
Wang S, Wang D, Yu Z et al (2021) Advances in research on petroleum biodegradability in soil. Environ Sci Process Impacts. https://doi.org/10.1039/D0EM00370K
Yang W, Hadibarata T, Mahmoud AH, Yuniarto A (2020) Biotransformation of pyrene in soil in the presence of earthworm Eisenia fetida. Environ Technol Innov. https://doi.org/10.1016/j.eti.2020.100701
Zhang W, Xia X, Wang JJ et al (2021) Oxidative stress and genotoxicity of nitenpyram to earthworms (Eisenia foetida). Chemosphere 264. https://doi.org/10.1016/j.chemosphere.2020.128493
Acknowledgements
The authors are thankful to the Director CSIR- Indian Institute of Toxicology Research (CSIR-IITR) for his support. SK acknowledges UGC, University Grants Commission (UGC), India, for providing the fellowship and Academy of Science and Innovative Research (AcSIR), India, for PhD enrolment. KG is thankful to CSIR, and MS gratefully acknowledges ICMR, New Delhi, for research fellowships. SK, KG, and MS acknowledge other lab members for their support.
Funding
SA and NM are thankful to the Council of Scientific and Industrial Research (CSIR), New Delhi, for financial support. This manuscript carries the CSIR-IITR communication number: IITR/SEC/2021–2022/65.
Author information
Authors and Affiliations
Contributions
SK: Study design, methodology standardisation, experimentation and observation, sample extraction and analysis for analytical measurements, writing of original draft. KG: writing of original draft, data acquisition and statistical analysis. MS: experimentation of ecotoxicological studies. NM and SA: conceptualization, supervision of experiments, data verification, analysis, and interpretation, funding acquisition, reviewing and editing the original draft.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
All authors read and approved the final manuscript.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Chris Lowe
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kumari, S., Gautam, K., Seth, M. et al. Bioremediation of polycyclic aromatic hydrocarbons in crude oil by bacterial consortium in soil amended with Eisenia fetida and rhamnolipid. Environ Sci Pollut Res 30, 82517–82531 (2023). https://doi.org/10.1007/s11356-023-28082-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-28082-y