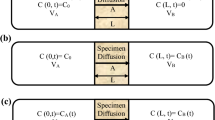
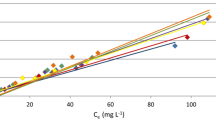
Abstract
Bentonite was applied in diffusive studies for selenium, an emerging contaminant. The planar source method was used to determine the apparent and effective diffusion coefficients and assess the mobility of the selenium species. A double Gaussian function described the results. Different diffusion coefficients were associated with different mobilities, and consequently, to the coexistence of two selenium species: selenite and selenate. Apparent diffusion coefficients were higher for selenate, around 10− 10 m2 s− 1, than for selenite, around 10− 12 m2 s− 1. Results from sequential extraction and distribution coefficient justified selenate’s greater mobility than selenite. Since the increase in redox potential from 448 to 511 mV may be associated with selenite oxidation in an interconversion process, the diffusion in bentonite demonstrates that applications in geological barriers deserve attention regarding the mobilization of selenium species. Interconversions can mobilize selenium, as reduced species can shift to more oxidized and mobile species, enhancing environmental contamination.





Similar content being viewed by others
Notes
The main numerical results in this section can be generated from the MatLab script available at https://data.mendeley.com/datasets/d8mjk92n8g/1; Angelita Ribeiro, Barbara Kroetz, Cesar Tarley, Taufik Abrao, Paulo Parreira, and Maria Santos (2021), “Separating selenium species by diffusion in Brazilian bentonite: a mathematical modeling approach”, Mendeley Data, 16 December 2021/Version 1, https://doi.org/10.17632/d8mjk92n8g.1
References
Ahonen L, Tiljander M, Kivikoski H, Laaksonen R (2008) Quality assurance of the bentonite material. Working Report 33 Posiva WR. Olkiluoto, Finland
Aldaba D, Gutiérrez MG, Abrão T, Rigol A, Vidal M (2015) Transport of radioselenium oxyanions by diffusion in unsaturated soils. Radiochim Acta 103:501–511. https://doi.org/10.1515/ract-2014-2318
Aldaba D, Gutiérrez MG, Rigol A, Vidal M (2010a) Comparison of laboratory methodologies for evaluating radiostrontium diffusion in soils: Planar-source versus half-cell methods. Sci Total Environ 408:5966–5971. https://doi.org/10.1016/j.scitotenv.2010.05.035
Aldaba D, Rigol A, Vidal M (2010b) Diffusion experiments for estimating radiocesium and radiostrontium sorption in unsaturated soils from Spain comparison with batch sorption data. J Hazard Mater 181:1072–1079. https://doi.org/10.1016/j.jhazmat.2010.05.124
Alonso U, Missana T, Gutiérrez M, Patelli A, Siitari-Kauppi M, Rigato V (2009) Diffusion coefficient measurements in consolidated clays determined by RBS micro-scale profiling. Appl Clay Sci 43:477–484. https://doi.org/10.1016/j.clay.2008.12.005
Appelo CAJ, Van Loon LR, Wersin P (2010) Multicomponent diffusion of a suite of tracers (HTO, Cl, Br, I, Na, Sr, Cs) in a single sample of Opalinus Clay. Geochim Cosmochim Acta 74:1201–1219. https://doi.org/10.1016/j.gca.2009.11.013
Barrett EP, Joyner LG, Halenda PP (1951) The determination of pore volume and area distributions in porous substances. I. Computations from nitrogen isotherms. J Am Chem Soc 73:373–380
Beauwens T, De Canniere P, Moors H, Wang L, Maes N (2005) Studying the migration behaviour of selenate in boom clay by electromigration. Eng Geol 77:285–293. https://doi.org/10.1016/j.enggeo.2004.07.019
Brunauer S, Emmett PH (1938) Adsorption of gases in multimolecular layers. J Am Chem Soc 60:309–319
Cabon JY, Erler W (1998) Determination of selenium species in seawater by flow injection hydride generation in situ trapping followed by electrothermal atomic absorption spectrometry. Analyst 123:1565–1569. https://doi.org/10.1039/A802541J
Constantino LV, Quirino JN, Monteiro AM, Abrão T., Parreira PS, Urbano A, Santos MJ (2017) Sorption-desorption of selenite and selenate on Mg-Al layered double hydroxide in competition with nitrate, sulfate and phosphate. Chemosphere 181:627–634. https://doi.org/10.1016/j.chemosphere.2017.04.071
Cormenzana JL, Gutiérrez MG, Missana T, Junghanns A (2003) Simultaneous estimation of effective and apparent diffusion coefficients in compacted bentonite. J Contam Hydrol 61:63–72. https://doi.org/10.1016/S0169-7722(02)00113-4
Crank J (1975) The mathematics of diffusion, 2nd edn. Formerly of Brunel University Uxbridge, New York
Curti E, Aimoz L, Kitamura A (2013) Selenium uptake onto natural pyrite. J Radioanal Nuclear Chem 295:1655–1665. https://doi.org/10.1007/s10967-012-1966-9
Essington ME (2003) Soil and water chemistry: An integrative approach. CRC Press, United States of America
Gutiérrez MG, Cormenzana JL, Missana T, Alonso U, Mingarro M (2011) Diffusion of strongly sorbing cations (60Co and 152Eu) in compacted FEBEX bentonite. Phys Chem Earth 36:1708–1713. https://doi.org/10.1016/j.pce.2011.10.013
Idemitsu K, Kozaki H, Yuhara M, Arima T, Inagaki Y (2016) Diffusion behavior of selenite in purified bentonite. Prog Nucl Energy 92:279–285
Iida Y, Yamaguchi T, Tanaka T (2011) Experimental and modeling study on diffusion of selenium under variable bentonite content and porewater salinity. J Nucl Sci Technol 48:1170–1183. https://doi.org/10.1080/18811248.2011.9711805
Jasielec JJ, Stec J, Szyszkiewicz-Warzecha K, Lagosz A, Deja J, Lewenstam A, Filipek R (2020) Effective and apparent diffusion coefficients of chloride ions and chloride binding kinetics parameters in mortars: Non-stationary diffusion-reaction model and the inverse problem. Materials 13:5522. https://doi.org/10.3390/ma13235522
Kim SS, Min JH, Baik MH, N KG (2012) The sorption of selenite onto Ca-bentonite. Decontamination Decommissioning Res 1:17–18
Kong J, Lee CP, Sun Y, Hua R, Liu W, Wang Z, Li Y, Wang Y (2021) Anion exclusion and sorption effect for compacted bentonite: the dependency of diffusion coefficients and capacity of HTO and Se(IV). J Radioanal Nucl Chem, 717–725. https://doi.org/10.1007/s10967-021-07688-x
Kozaki T, Sato H, Sato S, Ohashi H (1999) Diffusion mechanism of cesium ions in compacted montmorillonite. Eng Geol 34:223–230. https://doi.org/10.1016/S0013-7952(99)00077-0
Kumar AR, Riyazuddin P (2011) Speciation of selenium in groundwater: Seasonal variations and redox transformations. J Hazard Mater 192:263–269. https://doi.org/10.1016/j.jhazmat.2011.05.013
Lavrentyeva E, Vassiliev S, Levin E, AA T, Polyakov SN, Leonid M, Napolskii K, Petrii O, Tsirlina G (2012) Smectite clays as the quasi-templates for platinum electrodeposition. Electrochim Acta 61:94–106. https://doi.org/10.1016/j.electacta.2011.11.105
Leroy P, Tournassat C, Bernard O, Devau N, Azaroual M (2015) The electrophoretic mobility of montmorillonite. zeta potential and surface conductivity effects. J Colloid Interface Sci 451:21–39. https://doi.org/10.1016/j.jcis.2015.03.047
McNaught AD, Wilkinson A, et al. (1997) Compendium of chemical terminology. vol 1669. Blackwell Science Oxford
Oliveira CIR, ROCHA MCG, Silva ALN, Bertolino LC (2016) Characterization of bentonite clays from Cubati, Paraiba (Northeast of Brazil). Cerâmica 62:272–277. https://doi.org/10.1590/0366-69132016623631970
Rabenhorst MC, Hively WD, James BR (2007) Measurements of soil redox potential. Sci Soc Am J 73:668–674. https://doi.org/10.2136/sssaj2007.0443
Rihayat T, Salim S, Arlina A, Fona Z, Jalal R, Alam PN, Sami ZM, Syarif J, Juhan N (2018) Determination of CEC value (cation exchange capacity) of Bentonites from North Aceh and Bener Meriah, Aceh Province Indonesia using three methods. In: IOP Conf Series: Materials science and engineering. https://doi.org/10.1088/1757-899X/334/1/012054, pp 1–1
Rollins MB, Pool DL (1968) Measurement of exchangeable cations in bentonites. Clays Clay Miner 16:165–172. https://doi.org/10.1346/CCMN.1968.0160207
Rouquerol F, Rouquerol J, Sing K, Lhewellyn P, Maurin G (2014) Adsorption by powders and porous solids principles, methodology and applications, 2nd edn. Academic Press, Marseille, France
Rovira M, Giménez J, Martínez M, Martínez-Lladó X, Pablo J, Martínez (2008) Sorption of selenium(IV), and selenium(VI) onto natural iron oxides: Goethite and hematite. J Hazard Mater 150:279–284. https://doi.org/10.1016/j.jhazmat.2007.04.098
Samper J, Daí Z, Molinero J., Gutiérrez MG, Missana T, Migarro M (2006) Inverse modeling of tracer experiments in FEBEX compacted Ca-bentonite. Phys Chem Earth 31:640–648. https://doi.org/10.1016/j.pce.2006.04.013
Sandy T, DiSante C (2010) Review of available technologies for the removal of Selenium from water. Working Report. CH2MHILL. North American Metals Council
Santos HD, Jacomine PKT, Anjos LD, Oliveira VD, Oliveira JD, Coelho MR, Cunha TD (2006) Sistema Brasileiro de Classificação de Solos. 2 ed. Empresa Brasileira de Pesquisa Agropecuária Embrapa Solos. Ministério da Agricultura, Pecuária e Abastecimento. Available in https://www.embrapa.br/
Sato H, Miyamoto S (2004) Diffusion behaviour of selenite and hydroselenide in compacted bentonite. Appl Clay Sci 26:47–55. https://doi.org/10.1016/j.clay.2003.10.007
Satterfield CN, Sherwood TK (1963) The role of diffusion in catalysis. Universidade de Michigan: Addison-Wesley Pub. Co, Michigan
Schaefer C, Arands R, Sloot V, Kosson D (1995) Prediction and experimental validation of liquid-phase diffusion resistance in unsaturated soils. J Contam Hydrol 20:145–166. https://doi.org/10.1016/0169-7722(95)00020-V
Shackelford CD, Moore S (2013) Fickian diffusion of radionuclides for engineered containment barriers: Diffusion coefficients, porosities, and complicating issues. Eng Geol 152:133–147. https://doi.org/10.1016/j.enggeo.2012.10.014
Sharmasarkar S, Vance GF (2002) Selenite-selenate sorption in surface coal mine environment. Adv Environ Res 7:87–95. https://doi.org/10.1016/S1093-0191(01)00110-1
Sharmasarkar S, Vance GF, Cassel-Sharmasarkar F (1996) Selenite-selenate speciation in mine soils and salt solutions: A comparison of atomic absorption spectroscopy and ion chromatography https://www.osti.gov/biblio/588812
Sing KSW (1982) Reporting physisorption data for gas/solid systems - with special reference to the determination of surface area and porosity. Pure Appl Chem 54:2201–2218
Tachi Y, Shibutani T, Sato H, Yui M (1998) Sorption and diffusion behavior of selenium in tuff. J Contam Hydrol 35:77–89. https://doi.org/10.1016/S0169-7722(98)00117-X
Tan L, Nancharaiah Y, van Hullebusch E, Lens N (2016) Selenium: environmental significance, pollution, and biological treatment technologies. Biotechnol Adv 34:886–907. https://doi.org/10.1016/j.biotechadv.2016.05.005
Teixeira PC, Donagemma GK, Fontana A, Teixeira WG (2017) Manual de Métodos de Análise de Solo. 3 ed. Empresa Brasileira de Pesquisa Agropecuária Embrapa Solos. Ministério da Agricultura, Pecuária e Abastecimento. Available in https://www.embrapa.br/
Thompson M, Stephen L, Wood R (2002) Harmonized guidelines for single laboratory validation of methods and analysis - IUPAC Technical Report. Pure Appl Chem 74:835–855
Tournassat C, Appelo CAJ (2011) Modelling approaches for anion-exclusion in compacted Na-bentonite. Geochim Cosmochim Acta 75:3698–3710. https://doi.org/10.1016/j.gca.2011.04.001
Van Loon L, Baeyens B, Bradbury M (2005) Diffusion and retention of sodium and strontium in Opalinus Clay: Comparison of sorption data from diffusion and batch sorption measurements, and geochemical calculations. Appl Geochem 20:2351–2363. https://doi.org/10.1016/j.apgeochem.2005.08.008
Van Loon LR, Glaus MA, Muller W (2007) Anion exclusion effects in compacted bentonites: Towards a better undesstanding of anion diffusion. Appl Geochem 22:2536–2552. https://doi.org/10.1016/j.apgeochem.2007.07.008
Wang S, Liang D, Wang D, Wei W, Fu D, Lin Z (2012) Selenium fractionation and speciation in agriculture soils and accumulation in corn (Zea mays L.) under field conditions in Shaanxi Province, China. Sci Total Environ, 159–164. https://doi.org/10.1016/j.scitotenv.2012.03.091
Wang Z, Wang H, Li Q, Xu M, Guo Y, Li J, Jinying Y (2016) ph effect on Re(VII) and Se(IV) diffusion in compacted GMZ bentonite. Appl Geochem 73:1–7. https://doi.org/10.1016/j.apgeochem.2016.07.015
Wu T, Wang H, Zheng Q, Zhao YL, Van Loon LR (2014) Diffusion behavior of Se(IV) and Re(VII) in GMZ bentonite. Appl Clay Sci 101:136–140. https://doi.org/10.1016/j.clay.2014.07.028
Wu T, Wang Z, Tonga Y, Wang Y, Van Loon LR (2018) Investigation of Re(VII) diffusion in bentonite by through-diffusion and modeling techniques. Appl Clay Sci 166:223–229. https://doi.org/10.1016/j.clay.2018.08.023
Acknowledgements
The authors would like to thank CNPq (The National Council for Scientific and Technological Development), under Grants 309927/2015-3 and 304066/2015-0 (Researcher grants), Capes (Coordination of Superior Level Staff Improvement) and Araucaria Foundation under Grants 507/2014 (Researcher grant) for their financial support and fellowships.
Funding
Grants 309927/2015-3 and Researcher grants 304066/2015-0 from CNPq, Capes (Coordination of Superior Level Staff Improvement) and Araucaria Foundation under Grants 507/2014 (Researcher grants) for their financial support and fellowships.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
Not applicable
Consent for publication
Not applicable
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Ioannis A. Katsoyiannis
Author contributions
All authors whose names appear on the submission: (i) made substantial contributions to the conception or design of the work; the acquisition, analysis, and interpretation of data, (ii) drafted the work or revised it critically for important intellectual content, (iii) approved the version to be published, and (iv) agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Data availability
The main numerical results in this section can be generated from the MatLab script available at https://data.mendeley.com/datasets/d8mjk92n8g/1; Angelita Ribeiro, Barbara Kroetz, Cesar Tarley, Taufik Abrao, Paulo Parreira, and Maria Santos (2021), “Separating selenium species by diffusion in Brazilian bentonite: a mathematical modeling approach”, Mendeley Data, 16 December 2021/Version 1, https://doi.org/10.17632/d8mjk92n8g.1
Code availability
Not applicable
Consent to participate
Not applicable
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ribeiro, A., Kroetz, B.L., Tarley, C.R.T. et al. Separating selenium species by diffusion in Brazilian bentonite: a mathematical modeling approach. Environ Sci Pollut Res 29, 88119–88130 (2022). https://doi.org/10.1007/s11356-022-21529-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-21529-8