Abstract
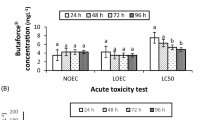
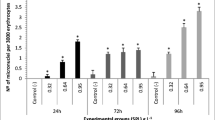
Malachite green (MG) is a triphenylmethane dye that is widely used in aquaculture as a fungicide, bactericide, ectoparasiticide, and antiprotozoal. There is great debate regarding the potential for this compound to trigger adverse effects. Here, we review the previous findings and then evaluate the lethal and sublethal effects of MG in the species Hemichromis bimaculatus (jewelfish). The lethal concentration for 50% of the fish in 96 h was 1 mg/L. We observed a dose-dependent increase in the percentage of fish mortality as well as physical and behavioral changes. We further found that the highest tested sublethal dose significantly increased the DNA damage index identified using the comet assay (74.97 ± 13.8 at a significant level of P < 0.05 for the 0.75 mg/L concentration), but did not significantly alter the results of the micronucleus test. Although our results suggest that MG confers risks on exposed fish, the findings were significant only at the highest exposure concentration (0.75 mg/L). At lower concentrations (0.25 mg/L and 0.5 mg/L), no adverse effect was observed. The maximum MG concentration recommended for use in ornamental fish farming is 0.2 mg/L. Therefore, our results suggest that, specifically for the parameters analyzed in this work, MG does not have any adverse effect when users strictly adhere to the recommended concentration criteria for ornamental fish.





Similar content being viewed by others
References
Alberts B, Wilson J, Hunt T (2008) Molecular biology of the cell. Garland Science
Ayres M, Ayres Jr M, Ayres DL, Santos AS (2007) BioEstat 5.0 – Aplicações estatísticas nas áreas das Ciências Biológicas e Médicas. Belém: Sociedade Civil Mamirauá, 5th edn. p 290
Bassleer G (2009) The new illustrated guide to fish diseases in ormental tropical and pond fish, 3rd Edition. Bassleer Biofish, Westmeerbeek
Bell DM (1982) Physiological and psychophysical spectral sensitivities of the cichlid fish, Hemichromis bimaculatus. J Exp Zool 223:29–32. https://doi.org/10.1002/jez.1402230105
Bergwerff AA, Kuiper RV, Scherpenisse P (2004) Persistence of residues of malachite green in juvenile eels (Anguilla anguilla). Aquaculture 233:55–63. https://doi.org/10.1016/j.aquaculture.2003.11.013
Bhosale MM, Felix S, Aruna S, et al (2017) Induced breeding of African jewelfish Hemichromis bimaculatus (Gill, 1862) using gonadotropic signaling molecular analogue WOVA-FH. ~ 2547 ~ J Entomol Zool Stud 5
Bills TD, Marking LL, Chandler JH Jr (1977) Malachite green: its toxicity to aquatic organisms, persistence, and removal with activated carbon. US Fish Wildl Serv
Bolognesi C, Perrone E, Roggieri P, Pampanin DM, Sciutto A (2006) Assessment of micronuclei induction in peripheral erythrocytes of fish exposed to xenobiotics under controlled conditions. Aquat Toxicol 78:S93–S98. https://doi.org/10.1016/j.aquatox.2006.02.015
Bose B, Motiwale L, Rao KVK (2005) DNA damage and G2/M arrest in Syrian hamster embryo cells during malachite green exposure are associated with elevated phosphorylation of ERK1 and JNK1. Cancer Lett 230:260–270. https://doi.org/10.1016/j.canlet.2005.01.001
Carrasco KR, Tilbury KL, Myers MS (1990) Assessment of the piscine micronucleus test as an in situ biological indicator of chemical contaminant effects. Can J Fish Aquat Sci 47:2123–2136. https://doi.org/10.1139/f90-237
Cavalcante DGSM, Martinez CBR, Sofia SH (2008) Genotoxic effects of roundup® on the fish Prochilodus lineatus. Mutat Res Genet Toxicol Environ Mutagen 655:41–46. https://doi.org/10.1016/j.mrgentox.2008.06.010
Çavaş T (2008) In vivo genotoxicity of mercury chloride and lead acetate: micronucleus test on acridine orange stained fish cells. Food Chem Toxicol 46:352–358. https://doi.org/10.1016/j.fct.2007.08.015
Collins AR, Ai-guo A, Duthie SJ (1995) The kinetics of repair of oxidative DNA damage (strand breaks and oxidised pyrimidines) in human cells. Mutat Res 336:69–77
Collins AR, Dusinská M, Horská A (2001) Detection of alkylation damage in human lymphocyte DNA with the comet assay. Acta Biochim Pol 48:611–614
Culp SJ, Blankenship LR, Kusewitt DF, Doerge DR, Mulligan LT, Beland FA (1999) Toxicity and metabolism of malachite green and leucomalachite green during short-term feeding to Fischer 344 rats and B6C3F1 mice. Chem Biol Interact 122:153–170. https://doi.org/10.1016/S0009-2797(99)00119-2
Culp SJ, Beland FA, Heflich RH, Benson RW, Blankenship LR, Webb PJ, Mellick PW, Trotter RW, Shelton SD, Greenlees KJ, Manjanatha MG (2002) Mutagenicity and carcinogenicity in relation to DNA adduct formation in rats fed leucomalachite green. Mutat Res Fundam Mol Mech Mutagen 506–507:55–63. https://doi.org/10.1016/S0027-5107(02)00152-5
Da Silva ST, Fontanetti CS (2006) Micronucleus test and observation of nuclear alterations in erythrocytes of Nile tilapia exposed to waters affected by refinery effluent. Mutat Res Toxicol Environ Mutagen 605:87–93. https://doi.org/10.1016/j.mrgentox.2006.02.010
de Campos Ventura B, de Angelis DF, Marin-Morales AMA (2008) Mutagenic and genotoxic effects of the Atrazine herbicide in Oreochromis niloticus (Perciformes, Cichlidae) detected by the micronuclei test and the comet assay. Pestic Biochem Phys 90(1):42–51
Doerge DR, Churchwell MI, Gehring TA, Pu YM, Plakas SM (1998) Analysis of malachite green and metabolites in fish using liquid chromatography atmospheric pressure chemical ionization mass spectrometry. Rapid Commun Mass Spectrom 12:1625–1634. https://doi.org/10.1002/(SICI)1097-0231(19981115)12:21<1625::AID-RCM373>3.0.CO;2-I
Fernandes TCC, Mazzeo DEC, Marin-Morales MA (2007) Mechanism of micronuclei formation in polyploidizated cells of Allium cepa exposed to trifluralin herbicide. Pestic Biochem Physiol 88:252–259. https://doi.org/10.1016/j.pestbp.2006.12.003
Ferraro MVM, Fenocchio AS, Mantovani MS, Ribeiro CO, Cestari MM (2004) Mutagenic effects of tributyltin and inorganic lead (Pb II) on the fish H. malabaricus as evaluated using the comet assay and the piscine micronucleus and chromosome aberration tests. Genet Mol Biol 27:103–107. https://doi.org/10.1590/S1415-47572004000100017
Fessard V, Godard T, Huet S, Mourot A, Poul JM (1999) Mutagenicity of malachite green and leucomalachite green in in vitro tests. J Appl Toxicol 19:421–430. https://doi.org/10.1002/(SICI)1099-1263(199911/12)19:6<421::AID-JAT595>3.0.CO;2-6
Gluth G, Hanke W (1983) The effect of temperature on physiological changes in carp, Cyprinus carpio L., induced by phenol. Ecotoxicol Environ Saf 7:373–389. https://doi.org/10.1016/0147-6513(83)90003-9
Goldacre RJ, Phillips JN (1949) 370. The ionization of basic triphenylmethane dyes. J Chem Soc 0:1724. https://doi.org/10.1039/jr9490001724
Grisolia CK, Oliveira-Filho EC, Ramos FR, Lopes MC, Muniz DHF, Monnerat RG (2009) Acute toxicity and cytotoxicity of Bacillus thuringiensis and Bacillus sphaericus strains on fish and mouse bone marrow. Ecotoxicology 18:22–26. https://doi.org/10.1007/s10646-008-0252-7
Hartmann A, Speit G (1997) The contribution of cytotoxicity to DNA-effects in the single cell gel test (comet assay). Toxicol Lett 90:183–188. https://doi.org/10.1016/S0378-4274(96)03847-7
Jang GH, Park IS, Lee SH, Huh TL, Lee YM (2009) Malachite green induces cardiovascular defects in developing zebrafish (Danio rerio) embryos by blocking VEGFR-2 signaling. Biochem Biophys Res Commun 382:486–491. https://doi.org/10.1016/j.bbrc.2009.01.118
Kousar S, Javed M (2015) Studies on induction of nuclear abnormalities in peripheral blood erythrocytes of fish exposed to copper. 886:879–886. https://doi.org/10.4194/1303-2712-v15
Kubitza F, Kubtiza L (2004) Principais parasitoses e doenças dos peixes cultivados, 4a Edição
Manjanatha MG, Shelton SD, Bishop M, Shaddock JG, Dobrovolsky VN, Heflich RH, Webb PJ, Blankenship LR, Beland FA, Greenlees KJ, Culp SJ (2004) Analysis of mutations and bone marrow micronuclei in big blue® rats fed leucomalachite green. Mutat Res Fundam Mol Mech Mutagen 547:5–18. https://doi.org/10.1016/j.mrfmmm.2003.11.009
Melo KM, Oliveira R, Grisolia CK, Domingues I, Pieczarka JC, de Souza Filho J, Nagamachi CY (2015) Short-term exposure to low doses of rotenone induces developmental, biochemical, behavioral, and histological changes in fish. Environ Sci Pollut Res 22:13926–13938. https://doi.org/10.1007/s11356-015-4596-2
Mittelstaedt RA, Mei N, Webb PJ, Shaddock JG, Dobrovolsky VN, McGarrity LJ, Morris SM, Chen T, Beland FA, Greenlees KJ, Heflich RH (2004) Genotoxicity of malachite green and leucomalachite green in female big blue B6C3F1 mice. Mutat Res Genet Toxicol Environ Mutagen 561:127–138. https://doi.org/10.1016/j.mrgentox.2004.04.003
Nebbia C, Girolami F, Carletti M, Gasco L, Zoccarato I, Giuliano Albo A (2017) In vitro interactions of malachite green and leucomalachite green with hepatic drug-metabolizing enzyme systems in the rainbow trout (Onchorhyncus mykiss). Toxicol Lett 280:41–47. https://doi.org/10.1016/j.toxlet.2017.07.900
Niska K, Korkea-aho T, Lindfors E, Kiuru T, Tuomainen M, Taskinen J, Peltonen K (2009) Disappearance of malachite green residues in fry of rainbow trout (Oncorhynchus mykiss) after treatment of eggs at the hatching stage. Aquaculture 297:25–30. https://doi.org/10.1016/j.aquaculture.2009.08.037
OECD - (Organisation for Economic Co-operation and Development) (1992) Guideline for Testing of Chemicals, 203. Fish, acute toxicity test. OECD, Paris, France Available at: <www.oecd.org>
Omoregie E, Ofojekwu PC, Anosike JC, Adeleye AO (1998) Acute toxicity of malachite green to the Nile tilapia, Oreochromis niloticus (L.). J Aquac Tropics 13:233–237
Özkan F, Gündüz SG, Berköz M, Özlüer Hunt A (2011) Induction of micronuclei and other nuclear abnormalities in peripheral erythrocytes of Nile tilapia, Oreochromis niloticus, following exposure to sublethal cadmium doses. Turkish J Zool 35:585–592. https://doi.org/10.3906/zoo-0907-77
Rao KVK, Mahudawala DM, Redkar AA (1998) Malignant transformation of Syrian hamster embryo (SHE) cells in primary culture by malachite green: transformation is associated with abrogation of G2/M checkpoint control. Cell Biol Int 22:581–589. https://doi.org/10.1006/cbir.1998.0300
Ross LG, Ward KMH, Ross B (1985) The effects of formalin, malachite green and suspended solids on the respiratory activity of rainbow trout, Salmo gairdneri Richardson. Aquac Res 16:129–138. https://doi.org/10.1111/j.1365-2109.1985.tb00302.x
Rowland WJ (1975) The effects of dummy size and color on behavioral interaction in the Jewel cichlid, Hemichromis bimaculatus gill. Behaviour 53:109–125
Ruider S, Schmahl G, Mehlhorn H, Schmidt H, Ritter G (1997) Effects of different malachite green derivatives and metabolites on the fish ectoparasite, Ichthyophthirius multifiliis, Fouquet 1876 (Hymenostomatida, Ciliophora). Eur J Protistol 33:375–388. https://doi.org/10.1016/S0932-4739(97)80049-1
Schmahl G, Ruider S, Mehlhorn H et al (1992) Treatment of fish parasites - 9. Effects of a medicated food containing malachite green on Ichthyophthirius multifiliis Fouquet, 1876 (Hymenostomatida, Ciliophora) in ornamental fish. Parasitol Res 78:183–192. https://doi.org/10.1007/BF00931725
Schoettger RA (1970) Toxicology of thiodan in several fish and aquatic invertebrates. Investig Fish Control 35:2–31
Silva-Pereira LC, Cardoso PCS, Leite DS, Bahia MO, Bastos WR, Smith MAC, Burbano RR (2005) Mercury genotoxicity on human lymphocyte cultures. Braz J Med Biol Res 38:901–907
Smith MJ, Heath AG (1979) Acute toxicity of copper, chromate, zinc, and cyanide to freshwater fish: effect of different temperatures. Bull Environ Contam Toxicol 22:113–119. https://doi.org/10.1007/BF02026917
Srivastava SJ, Singh ND, Srivastava AK, Sinha R (1995) Acute toxicity of malachite green and its effects on certain blood parameters of a catfish, Heteropneustes fossilis. Aquat Toxicol 31:241–247. https://doi.org/10.1016/0166-445X(94)00061-T
Stammati A, Nebbia C, De Angelis I et al (2005) Effects of malachite green (MG) and its major metabolite, leucomalachite green (LMG), in two human cell lines. Toxicol in Vitro 19:853–858. https://doi.org/10.1016/j.tiv.2005.06.021
Strunjak-Perovic I, Coz-Rakovac R, Topic Popovic N, Jadan M (2009) Seasonality of nuclear abnormalities in gilthead sea bream Sparus aurata (L.) erythrocytes. Fish Physiol Biochem 35:287–291. https://doi.org/10.1007/s10695-008-9208-3
Sundarrajan M, Fernandis AZ, Subrahmanyam G, Prabhudesai S, Krishnamurthy SC, Rao KVK (2000) Overexpression of G1/S cyclins and PCNA and their relationship to tyrosine phosphorylation and dephosphorylation during tumor promotion by metanil yellow and malachite green. Toxicol Lett 116:119–130. https://doi.org/10.1016/S0378-4274(00)00216-2
Wan H, Weng S, Liang L, Lu Q, He J (2011) Evaluation of the developmental toxicity of leucomalachite green administered orally to rats. Food Chem Toxicol 49:3031–3037. https://doi.org/10.1016/j.fct.2011.10.003
Werth G, Boiteux A (1968) Toxicity of triphenylmethane dyes. Malachite green as an uncoupling agent of oxidative phosphorylation in vivo and in vitro. Arch Toxikol 23:82–103
Yonar ME, Yonar SM (2010) Changes in selected immunological parameters and antioxidant status of rainbow trout exposed to malachite green (Oncorhynchus mykiss, Walbaum, 1792). Pestic Biochem Physiol 97:19–23. https://doi.org/10.1016/j.pestbp.2009.11.009
Funding
This study was funded by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES; Edital Pro-Amazônia Biodiversidade e Sustentabilidade 047/2012) and Banco Nacional de Desenvolvimento Econômico e Social–BNDES (Operation 2.318.697.0001) in projects coordinated by Julio Cesar Pieczarka. This study is part of the doctorship thesis of Augusto Cesar Paes de Souza in PhD in Biodiversity and Biotechnology–PPG-BIONORTE.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Souza, A.C.P., Melo, K.M., de Azevedo, L.F.C. et al. Lethal and sublethal exposure of Hemichromis bimaculatus (Gill, 1862) to malachite green and possible implications for ornamental fish. Environ Sci Pollut Res 27, 33215–33225 (2020). https://doi.org/10.1007/s11356-020-09615-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-09615-1