Abstract
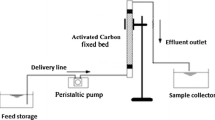
The performance of activated carbon (AC) for the adsorption of Acid Orange 7 (AO7) was investigated in both batch and column studies. The optimal conditions for adsorption process in batch study were found to be a stirring speed of 500 rpm, AC dosage of 5 g/L, and initial AO7 concentration of 100 mg/L. The spent AC was then treated with peroxydisulfate (PDS), and the regenerated AC was used again to adsorb AO7. Both pseudo-first-order and pseudo-second-order rate models for adsorption kinetics were investigated, and the results showed that the latter model was more appropriate. The effects of regeneration time, PDS concentration, and stirring speed on AO7-spent AC regeneration were investigated in batch studies, and the optimal conditions were time 2 h, stirring speed 700 rpm, and PDS concentration 10 g/L. Under the same adsorption conditions, 89% AO7 could be decolorized by adsorption using regenerated AC. In the column studies, the effect of flow rate was investigated and the adsorption capacity was nearly the same when the flow rate rose from 7.9 to 11.4 mL/min, but it decreased significantly when the flow rate was increased to 15.2 mL/min. The performance of regenerated AC in the column was also investigated, and a slight increase in the adsorption capacity was observed in the second adsorption cycle. However, the adsorption capacity decreased to some extent in the third cycle due to the consumption of C-OH group on the AC surface during PDS regeneration.









Similar content being viewed by others
References
An D, Westerhoff P, Zheng MX, Wu MY, Yang Y, Chiu CA (2015) UV-activated persulfate oxidation and regeneration of NOM-saturated granular activated carbon. Water Res 73:304–310
Banuelos JA, Rodriguez FJ, Rocha JM, Bustos E, Rodriguez A, Cruz JC, Arriaga LG, Godinez LA (2013) Novel electro-Fenton approach for regeneration of activated carbon. Environ Sci Technol 47:7927–7933
Banuelos JA, Garcia-Rodriguez O, Rodriguez-Valadez FJ, Manriquez J, Bustos E, Rodriguez A, Godinez LA (2015) Cathodic polarization effect on the electro-Fenton regeneration of activated carbon. J Appl Electrochem 45:523–531
Cai C, Zhang H, Zhong X, Hou LW (2015) Ultrasound enhanced heterogeneous activation of peroxymonosulfate by a bimetallic Fe-Co/SBA-15 catalyst for the degradation of Orange II in water. J Hazard Mater 283:70–79
Chang ST, Lin KYA, CS L (2014) Efficient adsorptive removal of tetramethylammonium hydroxide (TMAH) from water using graphene oxide. Sep Purif Technol 133:99–107
Duan XH, Srinivasakannan C, Liang JS (2014) Process optimization of thermal regeneration of spent coal based activated carbon using steam and application to methylene blue dye adsorption. J Taiwan Inst Chem Eng 45:1618–1627
Fang GD, Liu C, Gao J, Zhou DM (2014) New insights into the mechanism of the catalytic decomposition of hydrogen peroxide by activated carbon: implications for degradation of diethyl phthalate. Ind Eng Chem Res 53:19925–19933
Gao YW, Guo YZ, Zhang H (2016) Iron modified bentonite: enhanced adsorption performance for organic pollutant and its regeneration by heterogeneous visible light photo-Fenton process at circumneutral pH. J Hazard Mater 302:105–113
Gupta VK, Mittal A, Gajbe V, Mittal J (2006) Removal and recovery of the hazardous azo dye acid Orange 7 through adsorption over waste materials: bottom ash and de-oiled soya. Ind Eng Chem Res 45:1446–1453
Gupta VK, Gupta B, Rastogi A, Agarwal S, Nayak A (2011) A comparative investigation on adsorption performances of mesoporous activated carbon prepared from waste rubber tire and activated carbon for a hazardous azo dye-acid blue 113. J Hazard Mater 186:891–901
Hamzeh Y, Ashor A, Azadeh E, Abdulkhani A (2012) Removal of Acid Orange 7 and Remazol Black 5 reactive dyes from aqueous solutions using a novel biosorbent. Mater Sci Eng C 32:1394–1400
Horng RS, Tseng IC (2008) Regeneration of granular activated carbon saturated with acetone and isopropyl alcohol via a recirculation process under H2O2/UV oxidation. J Hazard Mater 154:366–372
Huling SG, Kan E, Wingo C (2009) Fenton-driven regeneration of MTBE-spent granular activated carbon—effects of particle size and iron amendment procedures. Appl Catal B 89:651–658
Hutson A, Ko S, Huling SG (2012) Persulfate oxidation regeneration of granular activated carbon: reversible impacts on sorption behavior. Chemosphere 89:1218–1223
Li HY, Li YL, Xiang LJ, Huang QQ, Qiu JJ, Zhang H, Sivaiah MV, Baron F, Barrault J, Petit S, Valange S (2015a) Heterogeneous photo-Fenton decolorization of Orange II over Al-pillared Fe-smectite: response surface approach, degradation pathway, and toxicity evaluation. J Hazard Mater 287:32–41
Li QM, Qi YS, Gao CZ (2015b) Chemical regeneration of spent powdered activated carbon used in decolorization of sodium salicylate for the pharmaceutical industry. J Clean Prod 86:424–431
Li J, Lin H, Yang L, Zhang H (2016) Copper-spent activated carbon as a heterogeneous peroxydisulfate catalyst for the degradation of Acid Orange 7 in an electrochemical reactor. Water Sci Technol 73:1802–1808
Liang CJ, Lin YT, Shin WH (2009a) Persulfate regeneration of trichloroethylene spent activated carbon. J Hazard Mater 168:187–192
Liang CJ, Lin YT, Shin WH (2009b) Treatment of trichloroethylene by adsorption and persulfate oxidation in batch studies. Ind Eng Chem Res 48:8373–8380
Lim CK, Bay HH, Neoh CH, Ai A, Majid ZA, Ibrahim Z (2013) Application of zeolite-activated carbon macrocomposite for the adsorption of Acid Orange 7: isotherm, kinetic and thermodynamic studies. Environ Sci Pollut Res 20:7243–7255
Mezohegyi G, van der Zee FP, Font J, Fortuny A, Fabregat A (2012) Towards advanced aqueous dye removal processes: a short review on the versatile role of activated carbon. J Environ Manag 102:148–164
Muranaka CT, Julcour C, Wilhelm AM, Delmas H, Nascimento CAO (2010) Regeneration of activated carbon by (photo)-Fenton oxidation. Ind Eng Chem Res 49:989–995
Nath K, Bhakhar MS (2011) Microbial regeneration of spent activated carbon dispersed with organic contaminants: mechanism, efficiency, and kinetic models. Environ Sci Pollut Res 18:534–546
Purkait MK, Maiti A, DasGupta S, De S (2007) Removal of Congo red using activated carbon and its regeneration. J Hazard Mater 145:287–295
Puziy AM, Poddubnaya OI, Socha RP, Gurgul J, Wisniewski M (2008) XPS and NMR studies of phosphoric acid activated carbon. Carbon 46:2113–2123
Rodríguez A, García J, Ovejero G, Mestanza M (2009) Adsorption of anionic and cationic dyes on activated carbon from aqueous solution: equilibrium and kinetics. J Hazard Mater 172:1311–1320
Saputra E, Muhammad S, Sun HQ, Wang SB (2013) Activated carbons as green and effective catalysts for generation of reactive radicals in degradation of aqueous phenol. RSC Adv 3:21905–21910
Sheshmani S, Ashori A, Hasanzadeh S (2014) Removal of Acid Orange 7 from aqueous solution using magnetic graphene/chitosan: a promising nano-adsorbent. Int J Biol Macromol 68:218–224
Silva JP, Sousa S, Rodrigues J, Antunes H, Porter JJ, Gonçalves I, Ferreira-Dias S (2004) Adsorption of acid orange 7 dye in aqueous solutions by spent brewery grains. Sep Purif Technol 40:309–315
Sotelo JL, Uguina MA, Delgdo JA, Celemin LI (2004) Adsorption of methyl ethyl ketone and trichloroethene from aqueous solutions onto activated carbon fixed-bed adsorbers. Sep Purif Technol 37:149–160
Tan KB, Vakili M, Horri BA, Poh PE, Abdullah AZ, Salamatinia B (2015) Adsorption of dyes by nanomaterials: recent developments and adsorption mechanisms. Sep Purif Technol 150:229–242
Tang SF, Lu N, Li J, Wu Y (2012) Design and application of an up-scaled dieletric barrier discharge plasma reactor for regeneration of phenol-saturated granular activated carbon. Sep Purif Technol 95:73–79
Wang X, Wang LG, Li JB, Qiu JJ, Cai C, Zhang H (2014) Degradation of Acid Orange 7 by persulfate activated with zero valent iron in the presence of ultrasonic irradiation. Sep Purif Technol 122:41–46
Wei MC, Wang KS, Lin IC, Hsiao TE, Lin YN, Tang CT, Chen JC, Chang SH (2012) Rapid regeneration of sulfanilic acid-sorbed activated carbon by microwave with persulfate. Chem Eng J 193:366–371
Weng CH, Hsu MC (2008) Regeneration of granular activated carbon by an electrochemical process. Sep Purif Technol 64:227–236
Weng CH, Lin YT, Hsu SC (2014) Electrochemical regeneration of Zn-saturated granular activated carbon from electroplating wastewater plant. Sep Purif Technol 49(4):506–512
Xia HY, Wu J, Xia Y, Zhang LB, Peng JH, Wang SX, Zheng ZQ, Zhang SZ (2015) Microwave assisted regeneration of spent activated carbon from petrochemical plant using response surface methodology. J Porous Mater 22:137–146
Xu SP, Ng JW, Zhang XW, Bai HW, Sun D (2011) Adsorption and photocatalytic degradation of Acid Orange 7 over hydrothermally synthesized mesoporous TiO2 nanotube. Colloid Surface A 379:169–175
Yang SY, Yang X, Shao XT, Niu R, Wang LL (2011) Activated carbon catalyzed persulfate oxidation of azo dye acid orange 7 at ambient temperature. J Hazard Mater 186:659–666
Ye ZH, Wang JW, Sun LY, Zhang DB, Zhang H (2015) Removal of ammonium from municipal landfill leachate using natural zeolites. Environ Technol 36:2919–2923
Zhang GS, Qu JH, Liu HJ, Cooper AT, Wu RC (2007) CuFe2O4/activated carbon composite: A novel magnetic adsorbent for the removal of acid orange II and catalytic regeneration. Chemosphere 68:1058–1066
Zhang J, Shao XT, Shi C, Yang SY (2013) Decolorization of Acid Orange 7 with peroxymonosulfate oxidation catalyzed by granular activated carbon. Chem Eng J 232:259–265
Acknowledgements
This study was supported by Natural Science Foundation of China (Grant No. 21547006), Wuhan Applied Basic Research Project (Grant No. 2016060101010074), and Shenzhen Basic Research Plan Project (Grant No. JCYJ20150508152951667). The generous help of Professor David H. Bremner in revising this manuscript is greatly appreciated.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Bingcai Pan
Rights and permissions
About this article
Cite this article
Li, J., Du, Y., Deng, B. et al. Activated carbon adsorptive removal of azo dye and peroxydisulfate regeneration: from a batch study to continuous column operation. Environ Sci Pollut Res 24, 4932–4941 (2017). https://doi.org/10.1007/s11356-016-8234-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-016-8234-4