Abstract
Purpose
The aim of this study was to evaluate the in vitro and in vivo characteristics of [89Zr]JRF/AβN/25, a radiolabeled monoclonal antibody directed against amyloid-β (Aβ).
Procedures
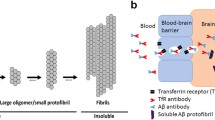
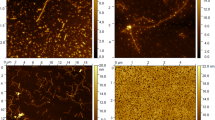
JRF/AβN/25 was labeled with 89Zr following modification with desferal. The affinity of the tracer for Aβ1–40 was determined in a saturation binding assay. In vitro stability was evaluated, and in vivo plasma stability and biodistribution of [89Zr]Df-Bz-JRF/AβN/25 were determined in wild-type mice. To evaluate whether the antibody can cross the blood-brain barrier, brain uptake in wild-type mice was additionally assessed by ex vivo autoradiography.
Results
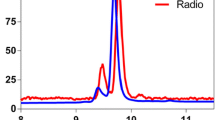
[89Zr]Df-Bz-JRF/AβN/25 was obtained in an average radiochemical yield of 50 % and a radiochemical purity of >97 %. A saturation binding assay demonstrated specific binding of [89Zr]Df-Bz-JRF/AβN/25 to Aβ1–40 with nanomolar affinity. The tracer was stable in buffer and proved to be stable in vivo with >92 % intact monoclonal antibody (mAb) remaining in the plasma at 48 h post injection. A biodistribution study showed a slow blood clearance with no significant accumulation of activity in any of the organs. Furthermore, [89Zr]Df-Bz-JRF/AβN/25 demonstrated modest brain penetration, which slowly decreased in time. This cerebral uptake was confirmed by ex vivo autoradiography.
Conclusions
[89Zr]Df-Bz-JRF/AβN/25 binds with high affinity to Aβ1–40. The tracer displays an acceptable in vivo stability and is able to cross the blood-brain barrier. [89Zr]Df-Bz-JRF/AβN/25 might therefore be a potential candidate for in vivo imaging of Aβ deposition in the brain.




Similar content being viewed by others
References
Nordberg A (2011) Molecular imaging in Alzheimer’s disease: new perspectives on biomarkers for early diagnosis and drug development. Alzheimers Res Ther 3:34
Nordberg A, Rinne J, Kadir A, Långström B (2010) The use of PET in Alzheimer disease. Nat Rev Neurol 6:78–87
Lemere C (2013) Immunotherapy for Alzheimer’s disease: hoops and hurdles. Mol Neurodegeneration 8:36
McLean D, Cooke MJ, Albay R et al (2013) Positron emission tomography imaging of fibrillar parenchymal and vascular amyloid-β in TgCRND8 mice. ACS Chem Neurosci 4:613–623
McLean D, Cooke MJ, Wang Y et al (2012) Anti-amyloid-β-mediated positron emission tomography imaging in Alzheimer’s disease mouse brains. PLoS One 7:e51958
Karran E, Mercken M, Strooper B (2011) The amyloid cascade hypothesis for Alzheimer’s disease: an appraisal for the development of therapeutics. Nat Rev Drug Discov 10:698–712
Forsberg A, Engler H, Almkvist O et al (2008) PET imaging of amyloid deposition in patients with mild cognitive impairment. Neurobiol Aging 29:1456–1465
Hickey JL, Lim S, Hayne DJ et al (2013) Diagnostic imaging agents for Alzheimer’s disease: copper radiopharmaceuticals that target Aβ plaques. J Am Chem Soc 135:16120–16132
Klunk W, Engler H, Nordberg A et al (2004) Imaging brain amyloid in Alzheimer’s disease with Pittsburgh compound-B. Ann Neurol 55:306–319
Ikonomovic M, Klunk W, Abrahamson E et al (2008) Post-mortem correlates of in vivo PiB-PET amyloid imaging in a typical case of Alzheimer’s disease. Brain 131:1630–1645
Magnusson K, Sehlin D, Syvänen S et al (2013) Specific uptake of an amyloid-β protofibril-binding antibody-tracer in AβPP transgenic mouse brain. J Alzheimers Dis 37:29–40
Yu YJ, Zhang Y, Kenrick M et al (2011) Boosting brain uptake of a therapeutic antibody by reducing its affinity for a transcytosis target. Sci Transl Med 3:84ra44
Vandermeeren M, Geraerts M, Pype S et al (2001) The functional γ-secretase inhibitor prevents production of amyloid β 1–34 in human and murine cell lines. Neurosci Lett 315:145–148
Mathews PM, Jiang Y, Schmidt SD et al (2002) Calpain activity regulates the cell surface distribution of amyloid precursor protein: inhibition of calpains enhances endosomal generation of β-cleaved C-terminal APP fragments. J Biol Chem 277(39):36415–36424
Holland JP, Caldas-Lopes E, Divilov V et al (2010) Measuring the pharmacodynamic effects of a novel Hsp90 inhibitor on HER2/neu expression in mice using Zr-DFO-trastuzumab. PLoS One 5:e8859
Vosjan MJ, Perk LR, Visser GW et al (2010) Conjugation and radiolabeling of monoclonal antibodies with zirconium-89 for PET imaging using the bifunctional chelate p-isothiocyanatobenzyl-desferrioxamine. Nat Protoc 5:739–743
Perk LR, Vosjan MJ, Visser GW et al (2010) p-Isothiocyanatobenzyl-desferrioxamine: a new bifunctional chelate for facile radiolabeling of monoclonal antibodies with zirconium-89 for immuno-PET imaging. Eur J Nucl Med Mol Imaging 37:250–259
Verel I, Visser GW, Boellaard R et al (2003) 89Zr immuno-PET: comprehensive procedures for the production of 89Zr-labeled monoclonal antibodies. J Nucl Med 44:1271–1281
Radde R, Bolmont T, Kaeser SA et al (2006) Aβ42-driven cerebral amyloidosis in transgenic mice reveals early and robust pathology. EMBO Rep 7:940–946
Abou DS, Ku T, Smith-Jones PM (2011) In vivo biodistribution and accumulation of 89Zr in mice. Nucl Med Biol 38:675–681
Fischer G, Seibold U, Schirrmacher R et al (2013) 89Zr, a radiometal nuclide with high potential for molecular imaging with PET: chemistry, applications and remaining challenges. Molecules 18:6469–6490
Zeglis BM, Davis CB, Aggeler R et al (2013) Enzyme-mediated methodology for the site-specific radiolabeling of antibodies based on catalyst-free click chemistry. Bioconjug Chem 24:1057–1067
Holland JP, Divilov V, Bander NH et al (2010) 89Zr-DFO-J591 for immunoPET of prostate-specific membrane antigen expression in vivo. J Nucl Med 51:1293–1300
Deri MA, Ponnala S, Zeglis BM et al (2014) Alternative chelator for 89Zr radiopharmaceuticals: radiolabeling and evaluation of 3,4,3-(LI-1,2-HOPO). J Med Chem 57:4849–4860
Zhai C, Summer D, Rangger C et al (2015) Novel bifunctional cyclic chelator for 89 Zr labeling–radiolabeling and targeting properties of RGD conjugates. Mol Pharm 12(6):2142–2150
Guérard F, Lee Y-S, Tripier R et al (2013) Investigation of Zr(IV) and 89Zr(IV) complexation with hydroxamates: progress towards designing a better chelator than desferrioxamine B for immuno-PET imaging. Chem Commun 49(10):1002–1004
Prins ND, Scheltens P (2013) Treating Alzheimer’s disease with monoclonal antibodies: current status and outlook for the future. Alzheimers Res Ther 5(6):56
Panza F, Logroscino G, Imbimbo BP, Solfrizzi V (2014) Is there still any hope for amyloid-based immunotherapy for Alzheimer’s disease? Current Opinion Psychiatry 27(2):128–137
Banks WA, Terrell B, Farr SA et al (2002) Passage of amyloid beta protein antibody across the blood-brain barrier in a mouse model of Alzheimer’s disease. Peptides 23:2223–2226
Acknowledgments
The authors are thankful to Joana Ramalho (Janssen Pharmaceutica) for her support with the in vitro binding assays and to Philippe Joye and Caroline Berghmans (Molecular Imaging Center Antwerp) for their support with the in vivo experiments. We also want to thank Annemie Van Eetveldt (Translational Neurosciences, University of Antwerp) for her help with the brain autoradiography studies. This work was supported in part by the Agency for Innovation by Science and Technology (IWT) grant 42/FA020000/5970.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest. All authors contributed to the conception of the study, coordinated the experimental design, and contributed to the writing and proofreading of the final manuscript.
Rights and permissions
About this article
Cite this article
Fissers, J., Waldron, AM., De Vijlder, T. et al. Synthesis and Evaluation of a Zr-89-Labeled Monoclonal Antibody for Immuno-PET Imaging of Amyloid-β Deposition in the Brain. Mol Imaging Biol 18, 598–605 (2016). https://doi.org/10.1007/s11307-016-0935-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-016-0935-z