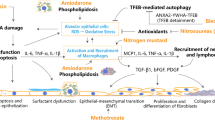
Abstract
Adenosine receptor (AR) suppresses inflammation and fibrosis by activating cyclic adenosine monophosphate (cAMP) signaling. We investigated whether altered AR expression contributes to the development of fibrotic diseases and whether A2AAR and A2BAR upregulation inhibits fibrotic responses. Primary human lung fibroblasts (HLFs) from normal (NHLFs) or patients with idiopathic pulmonary fibrosis (DHLF) were used for in vitro testing. Murine models of fibrotic liver or pulmonary disease were developed by injecting thioacetamide intraperitoneally, by feeding a high-fat diet, or by intratracheal instillation of bleomycin. Modafinil, which activates cAMP signaling via A2AAR and A2BAR, was administered orally. The protein amounts of A2AAR, A2BAR, and exchange protein directly activated by cAMP (Epac) were reduced, while collagen and α-smooth muscle actin (α-SMA) were elevated in DHLFs compared to NHLFs. In liver or lung tissue from murine models of fibrotic diseases, A2AAR and A2BAR were downregulated, but A1AR and A3AR were not. Epac amounts decreased, and amounts of collagen, α-SMA, KCa2.3, and KCa3.1 increased compared to the control. Modafinil restored the amounts of A2AAR, A2BAR, and Epac, and reduced collagen, α-SMA, KCa2.3, and KCa3.1 in murine models of fibrotic diseases. Transforming growth factor-β reduced the amounts of A2AAR, A2BAR, and Epac, and elevated collagen, α-SMA, KCa2.3, and KCa3.1 in NHLFs; however, these alterations were inhibited by modafinil. Our investigation revealed that A2AAR and A2BAR downregulation induced liver and lung fibrotic diseases while upregulation attenuated fibrotic responses, suggesting that A2AAR and A2BAR-upregulating agents, such as modafinil, may serve as novel therapies for fibrotic diseases.





Similar content being viewed by others
Data availability
The datasets and materials generated and analysed during the current study are available from the corresponding authors on reasonable request.
Abbreviations
- α-SMA:
-
α-smooth muscle actin
- AC:
-
Adenylyl cyclase
- AR:
-
Adenosine receptor
- Bleomycin:
-
BLM
- cAMP:
-
Cyclic adenosine monophosphate
- CDAHFD:
-
Choline-deficient, L-amino-acid-defined, high-fat diet with 0.1% methionine
- Col:
-
Collagen
- DMSO:
-
Dimethyl sulfoxide
- Epac:
-
Exchange protein directly activated by the cAMP
- HSC:
-
Hepatic stellate cell
- H&E:
-
Hematoxylin and eosin
- IL:
-
Interleukin
- IPF:
-
Idiopathic pulmonary fibrosis
- MF:
-
Modafinil
- MF-S:
-
(S)-Isomer of modafinil
- MMP9:
-
Matrix metalloproteinase-9
- MRS:
-
MRS 1754
- NECA:
-
5’-N-ethylcarboxamidoadenosine
- PCR:
-
Polymerase chain reaction
- P1NP:
-
Procollagen type1 N-terminal propeptide
- A2 :
-
AR-siRs siRNA against A2AAR + siRNA against A2BAR
- TAA:
-
Thioacetamide
- TIMP:
-
Tissue inhibitor of metalloproteinases
- TGFβ:
-
Transforming growth factor-β
- TNFα:
-
Tumor necrosis factor-α
- ZM:
-
ZM 241,385
References
Henderson NC, Rieder F, Wynn TA (2020) Fibrosis: from mechanisms to medicines. Nature 587(7835):555–566. https://doi.org/10.1038/s41586-020-2938-9
Murtha LA, Schuliga MJ, Mabotuwana NS, Hardy SA, Waters DW, Burgess JK, Knight DA, Boyle A (2017) The processes and mechanisms of Cardiac and Pulmonary Fibrosis. Front Physiol 8:777. https://doi.org/10.3389/fphys.2017.00777
Hecker L, Logsdon NJ, Kurundkar D, Kurundkar A, Bernard K, Hock T, Meldrum E, Sanders YY, Thannickal VJ (2014) Reversal of persistent fibrosis in aging by targeting Nox4-Nrf2 redox imbalance. Sci Transl Med 6(231):231ra47. https://doi.org/10.1126/scitranslmed.3008182
Duffield JS, Lupher M, Thannickal VJ, Wynn TA (2013) Host responses in tissue repair and fibrosis. Annu Rev Pathol 8:241–276. https://doi.org/10.1146/annurev-pathol-020712-163930
Mei Q, Liu Z, Zuo H, Yang Z, Qu J (2021) Idiopathic Pulmonary Fibrosis: an update on Pathogenesis. Front Pharmacol 12:797292. https://doi.org/10.3389/fphar.2021.797292
Maher TM, Bendstrup E, Dron L, Langley J, Smith G, Khalid JM, Patel H, Kreuter M (2021) Global incidence and prevalence of Idiopathic Pulmonary Fibrosis. Respir Res 22(1):197. https://doi.org/10.1186/s12931-021-01791-z
Cholankeril G, Perumpail RB, Pham EA, Ahmed A, Harrison SA (2016) Nonalcoholic fatty Liver Disease: Epidemiology, Natural History, and Diagnostic challenges. Hepatology 64(3):954. https://doi.org/10.1002/hep.28719
Raker VK, Becker C, Steinbrink K (2016) The cAMP pathway as therapeutic target in Autoimmune and Inflammatory Diseases. Front Immunol 7:123. https://doi.org/10.3389/fimmu.2016.00123
Insel PA, Murray F, Yokoyama U, Romano S, Yun H, Brown L, Snead A, Lu D, Aroonsakool N (2021) cAMP and Epac in the regulation of tissue fibrosis. Br J Pharmacol 166(2):447–456. https://doi.org/10.1111/j.1476-5381.2012.01847.x
Campo GM, Avenoso A, D’Ascola A, Prestipino V, Scuruchi M, Nastasi G, Calatroni A, Campo S (2012) Protein kinase a mediated anti-inflammatory effects exerted by adenosine treatment in mouse chondrocytes stimulated with IL-1beta. BioFactors 38(6):429–439. https://doi.org/10.1002/biof.1040
Hewer RC, Sala-Neby GB, Wu YJ, Newby AC, Bond M (2011) PKA and Epac synergistically inhibit smooth muscle cell proliferation. J Mol Cell Cardiol 50(1):87–98. https://doi.org/10.1016/j.yjmcc.2010.10.010
Choi S, Kim JA, Li H, Jo SE, Lee H, Kim TH, Kim M, Kim SJ, Suh SH (2021) Anti-inflammatory and anti-fibrotic effects of modafinil in nonalcoholic Liver Disease. Biomed Pharmacother 144:112372. https://doi.org/10.1016/j.biopha.2021.112372
Roach KM, Bradding P (2020) Ca(2+) signalling in fibroblasts and the therapeutic potential of K(ca)3.1 channel blockers in fibrotic Diseases. Br J Pharmacol 177(5):1003–1024. https://doi.org/10.1111/bph.14939
Choi S, Kim MY, Joo KY, Park S, Kim JA, Jung JC, Oh S, Suh SH (2012) Modafinil inhibits K(ca)3.1 currents and muscle contraction via a cAMP-dependent mechanism. Pharmacol Res 66(1):51–59. https://doi.org/10.1016/j.phrs.2012.02.009
Brandao WN, Andersen ML, Palermo-Neto J, Peron JP, Zager A (2019) Therapeutic treatment with Modafinil decreases the severity of experimental autoimmune encephalomyelitis in mice. Int Immunopharmacol 75:105809. https://doi.org/10.1016/j.intimp.2019.105809
Pal China S, Pal S, Chattopadhyay S, Porwal K, Mittal M, Sanyal S, Chattopadhyay N (2018) The wakefulness promoting drug Modafinil causes adenosine receptor-mediated upregulation of receptor activator of nuclear factor kappaB ligand in osteoblasts: negative impact of the drug on peak bone accrual in rats. Toxicol Appl Pharmacol 348:22–31. https://doi.org/10.1016/j.taap.2018.04.006
Effendi WI, Nagano T, Kobayashi K, Nishimura Y (2020) Focusing on Adenosine Receptors as a potential targeted therapy in Human Diseases. Cells 9(3):785. https://doi.org/10.3390/cells9030785
Scheibner KA, Boodoo S, Collins S, Black KE, Chan-Li Y, Zarek P, Powell JD, Horton MR (2009) The adenosine a2a receptor inhibits matrix-induced inflammation in a novel fashion. Am J Respir Cell Mol Biol 40(3):251–259. https://doi.org/10.1165/rcmb.2008-0168OC
Antonioli L, Csoka B, Fornai M, Colucci R, Kokai E, Blandizzi C, Blandizzi C, Hasko G (2014) Adenosine and inflammation: what’s new on the horizon? Drug Discov Today 19(8):1051–1068. https://doi.org/10.1016/j.drudis.2014.02.010
Linden J (2006) New insights into the regulation of inflammation by adenosine. J Clin Invest 116(7):1835–1837. https://doi.org/10.1172/JCI29125
Vecchio EA, White PJ, May LT (2017) Targeting Adenosine receptors for the treatment of Cardiac Fibrosis. Front Pharmacol 8:243. https://doi.org/10.3389/fphar.2017.00243
Prisinzano T, Podobinski J, Tidgewell K, Luo M, Swenson D (2004) Synthesis and determination of the absolute configuration of the enantiomers of modafinil. Tetrahedron-Asymmetry 15(6):1053–1058. https://doi.org/10.1016/j.tetasy.2004.01.039
Matsumoto M, Hada N, Sakamaki Y, Uno A, Shiga T, Tanaka C, Ito T, Katsume A, Sudoh M (2013) An improved mouse model that rapidly develops fibrosis in non-alcoholic steatohepatitis. Int J Exp Pathol 94(2):93–103. https://doi.org/10.1111/iep.12008
Salinthone S, Yadav V, Schillace RV, Bourdette DN, Carr DW (2010) Lipoic acid attenuates inflammation via cAMP and protein kinase A signaling. PLoS ONE 5(9):e13058. https://doi.org/10.1371/journal.pone.0013058
Choi S, Kim JA, Li HY, Lee SJ, Seok YS, Kim TH, Han KH, Park MH, Cho GJ, Suh SH (2019) Altered Redox State modulates endothelial K(ca)2.3 and K(ca)3.1 levels in normal pregnancy and Preeclampsia. Antioxid Redox Signal 30(4):505–519. https://doi.org/10.1089/ars.2017.7038
Organ L, Bacci B, Koumoundouros E, Kimpton WG, Samuel CS, Nowell CJ, Bradding P, Roach KM, Westall G, Jaffar J, Snibson KJ (2017) Inhibition of the K(ca)3.1 Channel alleviates established pulmonary fibrosis in a large animal model. Am J Respir Cell Mol Biol 56(4):539–550. https://doi.org/10.1165/rcmb.2016-0092OC
Lee WR, Kim kh, An HJ, Kim JY, Lee SJ, Han SM, Pak KK (2014) Apamin inhibits hepatic fibrosis through suppression of transforming growth factor beta1-induced hepatocyte epithelial-mesenchymal transition. Biochem Biophys Res Commun 450(1):195–201. https://doi.org/10.1016/j.bbrc.2014.05.089
Zhou Y, Murthy JN, Zeng D, Belardinelli L, Blackburn MR (2010) Alterations in adenosine metabolism and signaling in patients with Chronic Obstructive Pulmonary Disease and Idiopathic Pulmonary Fibrosis. PLoS ONE 5(2):e9224. https://doi.org/10.1371/journal.pone.0009224
Hasko G, Csoka B, Nemeth AH, Vizi ES, Pacher P (2009) A(2B) adenosine receptors in immunity and inflammation. Trends Immunol 30(6):263–270. https://doi.org/10.1016/j.it.2009.04.001
Frick JS, MacManus CF, Scully M, Glover LF, Eltzsching HK, Clogan SP (2009) Contribution of adenosine A2B receptors to inflammatory parameters of experimental Colitis. J Immunol 182(8):4957–4964. https://doi.org/10.4049/jimmunol.0801324
Hasko G, Linden J, Cronstein B, Pacher P (2008) Adenosine receptors: therapeutic aspects for inflammatory and immune Diseases. Nat Rev Drug Discov 7(9):759–770. https://doi.org/10.1038/nrd2638
Feng W, Song Y, Chen C, Lu ZZ, Zhang YY (2010) Stimulation of adenosine A(2B) receptors induces interleukin-6 secretion in cardiac fibroblasts via the PKC-delta-P38 signalling pathway. Br J Pharmacol 159(8):1598–1607. https://doi.org/10.1111/j.1476-5381.2009.00558.x
Cohen MV, Yang XL, Downey JM (2010) A(2b) adenosine receptors can change their spots. Br J Pharmacol 159(8):1595–1597. https://doi.org/10.1111/j.1476-5381.2010.00668.x
Eckle T, Grenz A, Laucher S, Eltzsching HK (2008) A2B adenosine receptor signaling attenuates acute lung injury by enhancing alveolar fluid clearance in mice. J Clin Invest 118(10):3301–3315. https://doi.org/10.1172/JCI34203
Coward WR, Saini G, Jenkins G (2010) The pathogenesis of Idiopathic Pulmonary Fibrosis. Ther Adv Respir Dis 4(6):367–388. https://doi.org/10.1177/1753465810379801
Funding
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (NRF-2022R1A2C1007823), NRF grant funded by the Korea government (MSIT) (NRF-2022R1A2C1092484), Ewha Womans University Research Grant of 2022 (1-2022-1108-001-1) and intramural research promotion grants from Ewha Womans University, School of Medicine (RP-2020).
Author information
Authors and Affiliations
Contributions
Haiyan Li, Jee Aee Kim, Seong-Eun Jo, Huisu Lee: performed the experiments; Haiyan Li and Suk Hyo Suh: wrote the manuscipt; Kwan-Chang Kim, Shinkyu Choi, and Suk Hyo Suh: conceived and designed the research, supervised project, edited manuscript, obtained funding.
Corresponding authors
Ethics declarations
Ethical approval
Experiments involving mice were approved by the Institutional Review Board for Human Research and Animal Care and Use Committee of the Ewha Womans University, Seoul, South Korea (EUM20-052). Experiments were conducted in accordance with the Declaration of Helsinki, Animal Care Guidelines of the Ewha Womans University Medical School, and Guide for the Care and Use of Laboratory Animals (NIH Publication no. 85 − 23, revised in 1996).
Competing interests
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, H., Kim, J.A., Jo, SE. et al. Modafinil exerts anti-inflammatory and anti-fibrotic effects by upregulating adenosine A2A and A2B receptors. Purinergic Signalling (2023). https://doi.org/10.1007/s11302-023-09973-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11302-023-09973-8