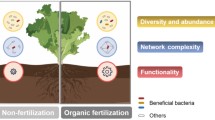
Abstract
Soil bacteria-fungi interactions are essential in the biogeochemical cycles of several nutrients, making these microbes major players in agroecosystems. While the impact of the farming system on microbial community composition has been extensively reported in the literature, whether sustainable farming approaches can promote associations between bacteria and fungi is still unclear. To study this, we employed 16S, ITS, and 18S DNA sequencing to uncover how microbial interactions were affected by conventional and organic farming systems on maize crops. The Bray–Curtis index revealed that bacterial, fungal, and arbuscular mycorrhizal fungi communities were significantly different between the two farming systems. Several taxa known to thrive in healthy soils, such as Nitrosophaerales, Orbiliales, and Glomus were more abundant in the organic farming system. Constrained ordination revealed that the organic farming system microbial community was significantly correlated with the β-glucosidase activity, whereas the conventional farming system microbial community significantly correlated with soil pH. Both conventional and organic co-occurrence interkingdom networks exhibited a parallel node count, however, the former had a higher number of edges, thus being denser than the latter. Despite the similar amount of fungal nodes in the co-occurrence networks, the organic farming system co-occurrence network exhibited more than 3-fold the proportion of fungal taxa as keystone nodes than the conventional co-occurrence network. The genera Bionectria, Cercophora, Geastrum, Penicillium, Preussia, Metarhizium, Myceliophthora, and Rhizophlyctis were among the fungal keystone nodes of the organic farming system network. Altogether, our results uncover that beyond differences in microbial community composition between the two farming systems, fungal keystone nodes are far more relevant in the organic farming system, thus suggesting that bacteria-fungi interactions are more frequent in organic farming systems, promoting a more functional microbial community.






Similar content being viewed by others
Data availability
Sequence data that support the findings of this study have been deposited in the National Center for Biotechnology Information—NCBI, and can be accessed by the Bioproject codes: PRJNA686771, PRJNA953004, and PRJNA953026.
References
Asghar W, Kataoka R (2021) Green manure incorporation accelerates enzyme activity, plant growth, and changes in the fungal community of soil. Arch Microbiol 204:7. https://doi.org/10.1007/s00203-021-02614-x
Banerjee S, Schlaeppi K, van der Heijden MGA (2018) Keystone taxa as drivers of microbiome structure and functioning. Nat Rev Microbiol 16:567–576. https://doi.org/10.1038/s41579-018-0024-1
Banerjee S, Walder F, Buchi L, Meyer M, Held AY, Gattinger A, Keller T, Charles R, van der Heijden MGA (2019) Agricultural intensification reduces microbial network complexity and the abundance of keystone taxa in roots. ISME J 13:1722–1736. https://doi.org/10.1038/s41396-019-0383-2
Banerjee S, Zhao C, Kirkby CA, Coggins S, Zhao S, Bissett A, van der Heijden MG, Kirkegaard JA, Richardson AE (2021) Microbial interkingdom associations across soil depths reveal network connectivity and keystone taxa linked to soil fine-fraction carbon content. Agric Ecosyst Environ 320:107559. https://doi.org/10.1016/j.agee.2021.107559
Berry D, Widder S (2014) Deciphering microbial interactions and detecting keystone species with co-occurrence networks. Front Microbiol 20:219. https://doi.org/10.3389/fmicb.2014.00219
Bolyen E et al (2019) Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat Biotechnol 37:852–857. https://doi.org/10.1038/s41587-019-0209-9
Bonanomi G, Lorito M, Vinale F, Woo SL (2018) Organic amendments, Beneficial Microbes, and Soil Microbiota: toward a Unified Framework for Disease suppression. Annu Rev Phytopathol 56:1–20. https://doi.org/10.1146/annurev-phyto-080615-100046
Callahan BJ, McMurdie PJ, Rosen MJ, Han AW, Johnson AJ, Holmes SP (2016) DADA2: high-resolution sample inference from Illumina amplicon data. Nat Methods 13:581–583. https://doi.org/10.1038/nmeth.3869
Cao Y, Dong Q, Wang D, Zhang P, Liu Y, Niu C (2022) microbiomeMarker: an R/Bioconductor package for microbiome marker identification and visualization. Bioinformatics 38:4027–4029. https://doi.org/10.1093/bioinformatics/btac438
Chaudhry V, Rehman A, Mishra A, Chauhan PS, Nautiyal CS (2012) Changes in bacterial community structure of agricultural land due to long-term organic and chemical amendments. Microb Ecol 64:450–460. https://doi.org/10.1007/s00248-012-0025-y
Chen J, Li J, Yang Y, Wang Y, Zhang Y, Wang P (2022) Effects of conventional and organic agriculture on soil arbuscular mycorrhizal fungal community in low-quality farmland. Front Microbiol 13:914627. https://doi.org/10.3389/fmicb.2022.914627
Csardi G, Nepusz T (2019) Package ‘igraph’. Network analysis and visualization. Repos CRAN Version 0(5):1–4
Culman SW et al (2012) Permanganate oxidizable carbon reflects a processed soil fraction that is sensitive to management. Soil Sci Soc Am J 76:494–504. https://doi.org/10.2136/sssaj2011.0286
De Oliveira-Pereira DC, Henriques-Longaresi R, do Valle Pereira G, Pelizari D, Homma SK, Demattê Filho L (2018) Organic maize: Changes in amino acid composition. Braz. J. Sustain. Agr 8:74–78
Deveau A et al (2018) Bacterial-fungal interactions: ecology, mechanisms and challenges. FEMS Microbiol Rev 42:335–352. https://doi.org/10.1093/femsre/fuy008
Durrer A, Gumiere T, Rumenos Guidetti Zagatto M, Petry Feiler H, Miranda Silva AM, Henriques Longaresi R, Homma SK, Cardoso E (2021) Organic farming practices change the soil bacteria community, improving soil quality and maize crop yields. PeerJ 9:e11985. https://doi.org/10.7717/peerj.11985
Geisseler D, Scow KM (2014) Long-term effects of mineral fertilizers on soil microorganisms—a review. Soil Biol Biochem 75:54–63. https://doi.org/10.1016/j.soilbio.2014.03.023
Gu Y, Banerjee S, Dini-Andreote F, Xu Y, Shen Q, Jousset A, Wei Z (2022) Small changes in rhizosphere microbiome composition predict disease outcomes earlier than pathogen density variations. ISME J 16:2448–2456. https://doi.org/10.1038/s41396-022-01290-z
Hartman K, van der Heijden MGA, Wittwer RA, Banerjee S, Walser JC, Schlaeppi K (2018) Cropping practices manipulate abundance patterns of root and soil microbiome members paving the way to smart farming. Microbiome 6:14. https://doi.org/10.1186/s40168-017-0389-9
Hirano H, Takemoto K (2019) Difficulty in inferring microbial community structure based on co-occurrence network approaches. BMC Bioinformatics 20:329. https://doi.org/10.1186/s12859-019-2915-1
Hurlbert SH (1984) Pseudoreplication and the design of ecological field experiments. Ecol Monogr 54:187–211. https://doi.org/10.2307/1942661
Jiang F, Zhang L, Zhou J, George TS, Feng G (2021) Arbuscular mycorrhizal fungi enhance mineralisation of organic phosphorus by carrying bacteria along their extraradical hyphae. New Phytol 230:304–315. https://doi.org/10.1111/nph.17081
Jupke JF, Schafer RB (2020) Should ecologists prefer model- over distance-based multivariate. Methods? Ecol Evol 10:2417–2435. https://doi.org/10.1002/ece3.6059
Kalam S, Basu A, Ahmad I, Sayyed RZ, El-Enshasy HA, Dailin DJ, Suriani NL (2020) Recent understanding of soil acidobacteria and their ecological significance: a critical review. Front Microbiol 11:580024. https://doi.org/10.3389/fmicb.2020.580024
Karanja EN, Fliessbach A, Adamtey N, Kambura AK, Musyoka M, Fiaboe K, Mwirichia R (2020) Diversity and structure of prokaryotic communities within organic and conventional farming systems in central highlands of Kenya. PLoS ONE 15:e0236574. https://doi.org/10.1371/journal.pone.0236574
Karnaouri A, Topakas E, Antonopoulou I, Christakopoulos P (2014) Genomic insights into the fungal lignocellulolytic system of Myceliophthora thermophila. Front Microbiol 5:281. https://doi.org/10.3389/fmicb.2014.00281
Khatri S, Sharma S (2021) How does organic farming shape the soil-and plant-associated microbiota? Symbiosis 84:391–398. https://doi.org/10.1007/s13199-021-00767-3
Kielak AM, Barreto CC, Kowalchuk GA, van Veen JA, Kuramae EE (2016) The ecology of acidobacteria: moving beyond genes and genomes. Front Microbiol 7:744. https://doi.org/10.3389/fmicb.2016.00744
Kwiatkowski CA, Harasim E, Feledyn-Szewczyk B, Antonkiewicz J (2020) Enzymatic activity of loess soil in organic and conventional farming systems. Agriculture 10:135. https://doi.org/10.3390/agriculture10040135
LeBlanc N, Crouch JA (2019) Prokaryotic taxa play keystone roles in the soil microbiome associated with woody perennial plants in the genus Buxus. Ecol Evol 9:11102–11111. https://doi.org/10.1002/ece3.5614
Lee J, Lee S, Young JP (2008) Improved PCR primers for the detection and identification of arbuscular mycorrhizal fungi. FEMS Microbiol Ecol 65:339–349. https://doi.org/10.1111/j.1574-6941.2008.00531.x
Leger RJS, Wang JB (2020) Metarhizium: jack of all trades, master of many. Open Biol 10:200307. https://doi.org/10.1098/rsob.200307
Li J, Zou C, Xu J, Ji X, Niu X, Yang J, Huang X, Zhang KQ (2015) Molecular mechanisms of nematode-nematophagous microbe interactions: basis for biological control of plant-parasitic nematodes. Annu Rev Phytopathol 53:67–95. https://doi.org/10.1146/annurev-phyto-080614-120336
Liu J, Zhang J, Li D, Xu C, Xiang X (2020) Differential responses of arbuscular mycorrhizal fungal communities to mineral and organic fertilization. Microbiologyopen 9:e00920. https://doi.org/10.1002/mbo3.920
Lori M, Symnaczik S, Mader P, De Deyn G, Gattinger A (2017) Organic farming enhances soil microbial abundance and activity-A meta-analysis and meta-regression. PLoS ONE 12:e0180442. https://doi.org/10.1371/journal.pone.0180442
Lupatini M, Korthals GW, de Hollander M, Janssens TK, Kuramae EE (2016) Soil microbiome is more heterogeneous in organic than in conventional farming system. Front Microbiol 7:2064. https://doi.org/10.3389/fmicb.2016.02064
Matteoli FP, Silva AMM, Feiler HP, de Araújo VLVP, Cardoso EJBN (2022) Predicting soil farming system and attributes based on soil bacterial community. Appl Soil Ecol 171:104335. https://doi.org/10.1016/j.apsoil.2021.104335
McMurdie PJ, Holmes S (2013) Phyloseq: an R package for reproducible interactive analysis and graphics of microbiome census data. PLoS ONE 8:e61217. https://doi.org/10.1371/journal.pone.0061217
Merloti LF, Mendes LW, Pedrinho A, de Souza LF, Ferrari BM, Tsai SM (2019) Forest-to-agriculture conversion in Amazon drives soil microbial communities and N-cycle. Soil Biol Biochem 137:107567. https://doi.org/10.1016/j.soilbio.2019.107567
Monaci E, Casucci C, De Bernardi A, Marini E, Landi L, Toscano G, Romanazzi G, Vischetti C (2022) Brassica carinata seed meal as soil amendment and potential biofumigant. Crops 2:233–246. https://doi.org/10.3390/crops2030017
Nagpal S, Singh R, Yadav D, Mande SS (2020) MetagenoNets: comprehensive inference and meta-insights for microbial correlation networks. Nucleic Acids Res 48:W572–W579. https://doi.org/10.1093/nar/gkaa254
Nguyen TBA, Chen QL, Yan ZZ, Li C, He JZ, Hu HW (2023) Trophic interrelationships of bacteria are important for shaping soil protist communities. Environ Microbiol Rep 15:298–307. https://doi.org/10.1111/1758-2229.13143
Nilsson RH et al (2019) The UNITE database for molecular identification of fungi: handling dark taxa and parallel taxonomic classifications. Nucleic Acids Res 47:D259–D264. https://doi.org/10.1093/nar/gky1022
Odom AR, Faits T, Castro-Nallar E, Crandall KA, Johnson WE (2023) Metagenomic profiling pipelines improve taxonomic classification for 16S amplicon sequencing data. Sci Rep 13:13957. https://doi.org/10.1038/s41598-023-40799-x
Oksanen J, Kindt R, Legendre P, O’Hara B, Stevens MH, Oksanen MJ (2007) The vegan package, Community ecology package, p. 719
Op De Beeck M, Lievens B, Busschaert P, Declerck S, Vangronsveld J, Colpaert JV (2014) Comparison and validation of some ITS primer pairs useful for fungal metabarcoding studies. PLoS ONE 9:e97629. https://doi.org/10.1371/journal.pone.0097629
Opik M, Vanatoa A, Vanatoa E, Moora M, Davison J, Kalwij JM, Reier U, Zobel M (2010) The online database MaarjAM reveals global and ecosystemic distribution patterns in arbuscular mycorrhizal fungi (Glomeromycota). New Phytol 188:223–241. https://doi.org/10.1111/j.1469-8137.2010.03334.x
Peltoniemi K, Velmala S, Fritze H, Lemola R, Pennanen T (2021) Long-term impacts of organic and conventional farming on the soil microbiome in boreal arable soil. Eur J Soil Biol 104:103314. https://doi.org/10.1016/j.ejsobi.2021.103314
Pichler M, Coskun OK, Ortega-Arbulu AS, Conci N, Worheide G, Vargas S, Orsi WD (2018) A 16S rRNA gene sequencing and analysis protocol for the Illumina MiniSeq platform. Microbiologyopen 7:e00611. https://doi.org/10.1002/mbo3.611
Purahong W, Wubet T, Lentendu G, Schloter M, Pecyna MJ, Kapturska D, Hofrichter M, Kruger D, Buscot F (2016) Life in leaf litter: novel insights into community dynamics of bacteria and fungi during litter decomposition. Mol Ecol 25:4059–4074. https://doi.org/10.1111/mec.13739
R Core Team (2021) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Sato K, Suyama Y, Saito M, Sugawara K (2005) A new primer for discrimination of arbuscular mycorrhizal fungi with polymerase chain reaction-denature gradient gel electrophoresis. Grassl Sci 51:179–181
Schmidt JE, Kent AD, Brisson VL, Gaudin ACM (2019) Agricultural management and plant selection interactively affect rhizosphere microbial community structure and nitrogen cycling. Microbiome 7:146. https://doi.org/10.1186/s40168-019-0756-9
Skinner C, Gattinger A, Krauss M, Krause HM, Mayer J, van der Heijden MGA, Mader P (2019) The impact of long-term organic farming on soil-derived greenhouse gas emissions. Sci Rep 9:1702. https://doi.org/10.1038/s41598-018-38207-w
Sudhakaran M, Ramamoorthy D, Savitha V, Kirubakaran N (2019) Soil enzyme activities and their relationship with soil physico-chemical properties and oxide minerals in coastal agroecosystem of Puducherry. Geomicrobiol J 36:452–459. https://doi.org/10.1080/01490451.2019.1570396
Suleiman AKA, Harkes P, van den Elsen S, Holterman M, Korthals GW, Helder J, Kuramae EE (2019) Organic amendment strengthens interkingdom associations in the soil and rhizosphere of barley (Hordeum vulgare). Sci Total Environ 695:133885. https://doi.org/10.1016/j.scitotenv.2019.133885
Tabatabai MA (1994) Soil enzymes. In: Weaver RW (ed) Methods of soil analysis: part 2 microbiological and biochemical properties. SSSA, Madison, pp 775–833
Tabatabai MA, Bremner JM (1969) Use of p-nitrofenol phosphate for assay of soil phosphatase activity. Soil Biol Biochem 1:301–307. https://doi.org/10.1016/0038-0717(69)90012-1
van Raij B, de Andrade JC, Cantarella H, Quaggio JA (2001) Análise química para avaliação da fertilidade de solos tropicais. IAC, São Paulo
Wagg C, Schlaeppi K, Banerjee S, Kuramae EE, van der Heijden MGA (2019) Fungal-bacterial diversity and microbiome complexity predict ecosystem functioning. Nat Commun 10:4841. https://doi.org/10.1038/s41467-019-12798-y
Walkley A, Black IA (1934) An examination of the Degtjareff method for determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Sci 37(1):29–38
Wang J, Song Y, Ma T, Raza W, Li J, Howland JG, Huang Q, Shen Q (2017) Impacts of inorganic and organic fertilization treatments on bacterial and fungal communities in a paddy soil. Appl Soil Ecol 112:42–50. https://doi.org/10.1016/j.apsoil.2017.01.005
Wang JL, Liu KL, Zhao XQ, Zhang HQ, Li D, Li JJ, Shen RF (2021) Balanced fertilization over four decades has sustained soil microbial communities and improved soil fertility and rice productivity in red paddy soil. Sci Total Environ 793:148664. https://doi.org/10.1016/j.scitotenv.2021.148664
Yilmaz P et al (2014) The SILVA and all-species living Tree Project (LTP) taxonomic frameworks. Nucleic Acids Res 42:D643–648. https://doi.org/10.1093/nar/gkt1209
Zumpf C, Cacho J, Grasse N, Quinn J, Hampton-Marcell J, Armstrong A, Campbell P, Negri MC, Lee DK (2021) Influence of shrub willow buffers strategically integrated in an Illinois corn-soybean field on soil health and microbial community composition. Sci Total Environ 772:145674. https://doi.org/10.1016/j.scitotenv.2021.145674
Acknowledgements
We thank Dr. Ademir Durrer, Dr. Sérgio Kenji Homma and Mr. Rodrigo Henriques Longaresi for their aid during the samples acquisition.
Funding
This research was funded by the São Paulo Research Foundation (FAPESP) (#2019/13436-8, #2017/24785-8, and #2016/18944-3) and the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brazil (CAPES)—Finance Code 001.
Author information
Authors and Affiliations
Contributions
F.P.M. Wrote the main manuscript text and performed the formal analyses. A.M.M.S. and V.L.V.P. performed the experimental setting, sample acquisition, and figures edition. H.P.F. performed the experimental setting and sample acquisition. E.J.B.N. obtained funding, performed the supervision, and project administration. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Matteoli, F.P., Silva, A.M.M., de Araújo, V.L.V.P. et al. Organic farming promotes the abundance of fungi keystone taxa in bacteria-fungi interkingdom networks. World J Microbiol Biotechnol 40, 119 (2024). https://doi.org/10.1007/s11274-024-03926-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-024-03926-y