Abstract
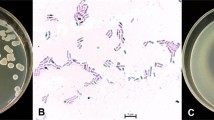
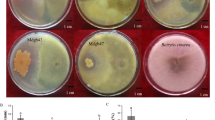
Isolate TEB1 an antagonistic endophytic bacterium, obtained from citrus leaves and identified as Bacillus amyloliquefaciens by 16S rDNA sequencing, was used for the biological control of mal secco disease of Citrus aurantium seedlings caused by the mitosporic fungus Phoma tracheiphila. The isolate TEB1 exhibited a good in vitro activity against P. tracheiphila in dual cultures as well as with the well diffusion method. C. aurantium seedlings watered with a suspension of TEB1 cells showed a reduction of 53.61 and 48.63 % in disease severity and incidence, respectively. A PCR test with specific primers was performed 365 days after inoculation and P. tracheiphila was detected along the whole stem in inoculated control plant while no amplification product was obtained in TEB1 treated seedlings. Molecular analysis of TEB1 revealed a positive amplification of fenD and ituC genes responsible of the biosynthesis of fengycin and iturin lipopeptides, respectively. Moreover, observations by optical microscope showed that TEB1 reduced by 55 % the germination of P. tracheiphila conidia and exhibited a marked effect on mycelia structure. Data suggest that lipopeptides produced by the bacterium interact with the cytoplasmic membrane of the fungus causing pore formation. TEB1 appears a potential candidate for the biological control of citrus mal secco disease.





Similar content being viewed by others
References
Avis TJ, Bélanger RR (2002) Mechanisms and means of detection of biocontrol activity of Pseudozima yeast against plant-pathogenic fungi. FEMS Yeast Res 2:5–8
Balmas V, Scherm B, Ghignone S, Salem AOM, Cacciola S, Migheli Q (2005) Characterisation of Phoma tracheiphila by RAPD, microsatellite-primed PCR and ITS rDNA sequencing and development of specific primers for in planta PCR detection. Eur J Plant Pathol 111:235–247
Chen XH, Koumoutsi A, Scholz R, Eisenreich A, Schneider K, Heinemeyer I, Morgenstern B, Voss B, Hess WR, Reva O, Junge H, Voigt B, Jungblut PR, Vater J, Süssmuth R, Liesegang H, Strittmatter A, Gottschalk G, Borriss R (2007) Comparative analysis of the complete genome sequence of the plant growth-promoting bacterium Bacillus amyloliquefaciens FZB42. Nat Biotechnol 25:1007–1014
Cho KM, Hong SY, Lee SM, Kim YH, Kahng GG, Lim YP, Kim H, Yun HD (2007) Endophytic bacterial communities in ginseng and their antifungal activity against pathogens. Microb Ecol 54:341–351
Chung S, Kong H, Buyer JS, Lakshman DK, Lydon J, Kim SD, Roberts DP (2008) Isolation and partial characterization of Bacillus subtilis ME488 for suppression of soilborne pathogens of cucumber and pepper. Appl Microbiol Biotechnol 80:115–123
Coco V, Grimaldi V, Licciardello G, Cirvilleri G, Grasso S, Catara A (2004) Inhibition of Phoma tracheiphila by Pseudomonas in citrus seedlings. Proc Int Soc Citric 2:729–732
Compant S, Clément C, Sessitsch A (2010) Plant growth-promoting bacteria in the rhizo-and endosphere of plants: their role, colonization, mechanisms involved and prospects for utilization. Soil Biol Biochem 42:669–678
Cutuli G, Laviola C, Perrotta G, Salerno M, Spina P (1984) Il mal secco degli agrumi. Seminario Internazionale di Studio organizzato nell’ambito del programma di ricerche Agrimed. Museo Villa Piccolo. Capo d’Orlando (Messina). (In Italian.) Fondazione Piccolo di Calanovella, Capo d’Orlando (ME), Italy
Demontis MA, Cacciola SO, Balmas V, Chessa V, Orrù M, Maserti BE, Mascia L, Raudino F, di San Lio GM, Migheli Q (2008) Development of real-time PCR systems based on SYBR® Green I and TaqMan® technologies for specific quantitative detection of Phoma tracheiphila in infected citrus. Eur J Plant Pathol 120:339–351
El Baltagy A, Nishioka K, Suzuki H, Sato T, Sato YI, Morisaki H, Mitsui H, Minamisawa K (2000) Isolation of endophytic bacteria from wild and traditionally cultivated rice varieties. Soil Sci Plant Nutr 46:617–629
Ezra D, Kroiter T, Sadowsky A (2007) Molecular characterization of Phoma tracheiphila, causal agent of mal secco disease of citrus, in Israel. Eur J Plant Pathol 118:183–191
Gagné S, Richard C, Rousseau H, Antoun H (1987) Xylem-residing bacteria in alfalfa roots. Can J Microbiol 33:996–1000
Hsieh FC, Li MC, Lin TC, Kao SS (2004) Rapid detection and characterization of surfactin-producing Bacillus subtilis and closely related species based on PCR. Curr Microbiol 49:186–191
Hudson O, Ahmad NH, Van Buren R, Savka MA (2010) Sugarcane and grapevine endophytic bacteria: isolation, detection of quorum sensing signals and identification by 16S v3 rDNA sequence analysis. In: Mendez-Vilas A (ed) Current research technology and education topics in applied microbiology and microbial biotechnology. Badajoz, Spain, pp 801–806
Kalai L, Mnari-Hattab M, Hajlaoui MR (2010) Molecular diagnostic to assess the progression of Phoma tracheiphila in Citrus aurantium seedlings and analysis of genetic diversity of isolates recovered from different citrus species in Tunisia. J plant Pathol 92:629–636
Kim PI, Bai H, Bai D, Chae H, Chung S, Kim Y, Park R, Chi YT (2004) Purification and characterization of a lipopeptide produced by Bacillus thuringiensis CMB26. J Appl Microbiol 97:942–949
Korsten L, De Jager ES, De Villurs EE, Lourens A, Wehner FC (1995) Evaluation of bacterial epiphytes isolated from avocado leaf and fruit surfaces for biocontrol of avocado postharvest diseases. Plant Dis 79:1149–1156
Leeman M, Den Ouden EM, Van Pelt JA, Cornelissen C, Matamala-Garros A, Bakker P, Schippers B (1996) Suppression of fusarium wilt of radish by co-inoculation of fluorescent Pseudomonas spp. and root-colonizing fungi. Eur J Plant Pathol 102:21–31
Lima G, Ippolito A, Nigro F, Salerno M (1994) tentativi di lotta biologica contro il mal secco degli agrumi (Phoma tracheiphila) a mezzo di batteri endofiti. La Difesa delle Piante 17:43–49 (In Italian)
Luisi N, De Cicco V, Cutuli G, Salerno M (1978) Analisi della patogenicită di Phoma tracheiphila (Petri) Kanc. et Ghik. Su alcune specie e cultivar di Agrumi. Phytopath Medit 18:162–165
Maget-Dana R, Marius Ptak (1995) Interactions of surfactin with membrane models. Biophys J 68:1937–1943
Magnet-Dana R, Peypoux F (1994) Iturins, a special class of pore forming lipopeptides: biological and physiological properties. Toxicol 87:151–174
Mahaffee WF, Kloepper JW (1997) Temporal changes in the bacterial communities of soil, rhizosphere and endorhiza associated with field-grown cucumber (Cucumis sativus L.). Microbial Ecol 34:210–223
McKinney HH (1923) Influence of soil temperature and moisture on infection of wheat seedlings by Helminthosporium sativum. J Agr Res 26:195–218
Migheli Q, Cacciola SA, Balmas V, Pane A, Ezra D, Di San Lio GM (2009) Mal secco disease caused by Phoma tracheiphila: a potential threat to lemon production worldwide. Plant Dis 93:852–867
Narisawa K, Tokumasu S, Hashiba T (1998) Suppression of clubroot formation in Chinese cabbage by the root endophytic fungus, Heteroconium chaetospira. Plant Pathol 47:206–210
Ongena M, Jacques P, Touré Y, Destain J, Jabrane A, Thonart P (2005) Involvement of fengycin-type lipopeptides in the multifaceted biocontrol potential of Bacillus subtilis. Appl Microbiol Biotechnol 69:29–38
Punithalingam E, Holliday P (1973) Deuterophoma tracheiphila. CMI Descriptions of Pathogenic Fungi and Bacteria No. 399. CAB International, Wallingford, UK
Ramarathnam R, Bo S, Chen Y, Fernando WGD, Xuewen G, de Kievit T (2007) Molecular and biochemical detection of fengycin- and bacillomycin D-producing Bacillus spp., antagonistic to fungal pathogens of canola and wheat. Can J Microbiol 53:901–911
Romero D, de Vicente A, Rakotoaly RH, Dufour SE, Veening JW, Arrebola E, Cazorla FM, Kuipers OP, Paquot M, Pérez-García A (2006) The iturin and fengycin families of lipopeptides are Key factors in antagonism of Bacillus subtilis toward Podosphaera fusca. Mol Plant Microbe Interact 20:430–440
Romero D, de Vicente A, Olmos JL, Davila JC, Perez-Garcia A (2007) Effect of lipopeptides of antagonistic strains of Bacillus subtilis on the morphology and ultrastructure of the cucurbit fungal pathogen Podosphaera fusca. J Appl Microbiol 103:969–976
Sadfi N, Chérif M, Fliss I, Boudabbous A, Antoun H (2001) Evaluation of Bacillus isolates from salty soils and Bacillus thuringiensis strains for the biocontrol of Fusarium dry rot of potato tubers. J. Plant Pathol 83:101–118
Shishido M, Loeb BM, Chanway CP (1995) External and internal root colonization of lodgepole pine seedlings by two growth-promoting Bacillus strains originated from different root microsites. Can J Microbiol 41:707–713
Soad A, GlX Algam, Coosemans J (2005) Delivery methods for introducing endophytic Bacillus into tomato and their effect on growth promotion and suppression of tomato wilt. Plant Pathol J 4:69–74
Timmer LW, Garnsey SM, Graham JH (2000) Compendium of citrus diseases. American Phytopathological Society, St Paul, MN, pp 33–35
Walker TS, Bais HP, Grotewold E, Vivanco JM (2003) Root exudation and rhizosphere biology. Plant Physiol 132:44–51
Wang H, Wen K, Zhao X, Wang X, Li A, Hong H (2009) The inhibitory activity of endophytic Bacillus sp. strain CHM1 against plant pathogenic fungi and its plant growth promoting effect. Crop Prot 28:634–639
Weisburg WG, Barns SM, Pelletier DA, Lane DJ (1991) 16S ribosomal DNA amplification for phylogenetic study. J Bacteriol 173:697–703
Whipps JM (1987) Effect of media on growth and interactions between a range of soil-borne glasshouse pathogens and antagonistic fungi. New Phytol 107:127–142
Yu GY, Sinclair JB, Hartman GL, Bertagnolli BL (2002) Production of iturin A by Bacillus amylolequefaciens suppressing Rhizoctonia solani. Soil Biol Biochem 34:955–963
Acknowledgments
This work was financially supported by grants from the Tunisian Ministry of Higher Education and Scientific Research. We would like to thank Prof. Ezzedine Aouani for critically reading the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kalai-Grami, L., Ben Slimane, I., Mnari-Hattab, M. et al. Protective effect of Bacillus amyloliquefaciens against infections of Citrus aurantium seedlings by Phoma tracheiphila . World J Microbiol Biotechnol 30, 529–538 (2014). https://doi.org/10.1007/s11274-013-1471-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-013-1471-5