Abstract
Purpose
Optimal management strategies for patients with high-risk prostate cancer (PCa) have not been established. This study aimed to estimate the impact of surgical procedures on perioperative and oncological outcomes in patients with high-risk PCa who received neoadjuvant chemohormonal therapy (CHT) prior to radical prostatectomy (RP).
Methods
In this retrospective study, we focused on patients with high-risk PCa who received neoadjuvant CHT followed by RP. The enrolled patients were divided into the following two groups according to surgical procedure: the robot-assisted RP (RARP) group and minimum incision endoscopic RP (MIE-RP) group. The primary endpoint was biochemical recurrence-free survival (BRFS).
Results
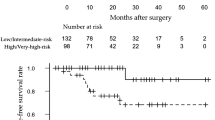
A total of 522 high-risk PCa patients were enrolled in this study. The median operating time was significantly shorter in the MIE-RP group than in the RARP group. The median estimated blood loss was significantly lower in the RARP group than in the MIE-RP group. The rates of positive surgical margins (PSMs) were not statistically significant in either group. During the follow-up period, biochemical recurrence (BCR) without clinical recurrence occurred in 60 (23.9%) patients in the MIE-RP group and 5 (1.8%) in the RARP group. The 5-year BRFS rate was 76.5% in the MIE-RP group and 97.6% in the RARP group (P < 0.001). On multivariate analysis, RARP, PSM, pathological T stage, and initial prostate-specific antigen were significantly associated with BCR.
Conclusions
Neoadjuvant CHT with subsequent RARP may decrease the risk of BCR when compared to MIE-RP.

Similar content being viewed by others
References
D’Amico AV, Whittington R, Malkowicz SB et al (1998) Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA 280:969–974
Heidenreich A, Bastian PJ, Bellmunt J et al (2014) European Association of Urology. EAU guidelines on prostate cancer. part 1: screening, diagnosis, and local treatment with curative intent-update 2013. Eur Urol 65:124–137
Yuh B, Artibani W, Heidenreich A et al (2014) The role of robot-assisted radical prostatectomy and pelvic lymph node dissection in the management of high-risk prostate cancer: a systematic review. Eur Urol 65:918–927
Koie T, Yamamoto H, Hatakeyama S et al (2011) Minimum incision endoscopic radical prostatectomy: clinical and oncological outcomes at a single institute. Eur J Surg Oncol 37:805–810
Hu JC, Gu X, Lipsitz SR, Barry MJ, D’Amico AV, Weinberg AC, Keating NL (2009) Comparative effectiveness of minimally invasive vs open radical prostatectomy. JAMA 302:1557–1564
Punnen S, Meng MV, Cooperberg MR, Greene KL, Cowan JE, Carroll PR (2013) How does robot-assisted radical prostatectomy (RARP) compare with open surgery in men with high-risk prostate cancer? BJU Int 112:E314–E320
Pierorazio PM, Mullins JK, Eifler JB et al (2013) Contemporaneous comparison of open vs minimally-invasive radical prostatectomy for high-risk prostate cancer. BJU Int 112:751–757
Koie T, Ohyama C, Yamamoto H, Hatakeyama S, Yoneyama T, Hashimoto Y, Kamimura N (2012) Safety and effectiveness of neoadjuvant luteinizing hormone-releasing hormone agonist plus low-dose estramustine phosphate in high-risk prostate cancer: a prospective single-arm study. Prostate Cancer Prostatic Dis 15:397–401
Koie T, Mitsuzuka K, Yoneyama T et al (2015) Neoadjuvant luteinizing-hormone-releasing hormone agonist plus low-dose estramustine phosphate improves prostate-specific antigen-free survival in high-risk prostate cancer patients: a propensity score-matched analysis. Int J Clin Oncol 20:1018–1025
Hagiwara K, Koie T, Ohyama C et al (2017) Efficacy of a neoadjuvant gonadotropin-releasing hormone antagonist plus low-dose estramustine phosphate in high-risk prostate cancer: a single-center study. Int Urol Nephrol 49:811–816
Fujita N, Koie T, Ohyama C et al (2017) Overall survival of high-risk prostate cancer patients who received neoadjuvant chemohormonal therapy followed by radical prostatectomy at a single institution. Int J Clin Oncol 22:1087–1093
Edge SB (2010) American Joint Committee on Cancer (AJC) Prostate. In: Edge SB, Byrd DR, Compton CC et al (eds) AJCC cancer staging manual, 7th edn. Springer, New York, pp 457–468
Epstein JI, Allsbrook WC Jr, Amin MB, Egevad LL, ISUP Grading Committee (2005) The 2005 International Society of Urological Pathology (ISUP) consensus conference on Gleason grading of prostatic carcinoma. Am J Surg Pathol 29:1228–1242
Marshall FF, Chan D, Partin AW, Gurganus R, Hortopan SC (1998) Minilaparotomy radical retropubic prostatectomy: technique and results. J Urol 160:2440–2445
Menon M, Shrivastava A, Kaul S, Badani KK, Fumo M, Bhandari M, Peabody JO (2007) Vattikuti institute prostatectomy: comtemporary technique and analysis of results. Eur Urol 51:648–657
Narita T, Koie T, Ookubo T et al (2017) The impact of extended lymph node dissection versus neoadjuvant therapy with limited lymph node dissection on biochemical recurrence in high-risk prostate cancer patients treated with radical prostatectomy: a multi-institutional analysis. Med Oncol 34:1
Clavien PA, Barkun J, de Oliveira ML et al (2009) The Clavien–Dindo classification of surgical complications: five-year experience. Ann Surg 250:187–196
Loeb S, Schaeffer EM, Trock BJ, Epstein JI, Humphreys EB, Walsh PC (2010) What are the outcomes of radical prostatectomy for high-risk prostate cancer? Urology 76:710–714
Sooriakumaran P, Nyberg T, Akre O et al (2014) Comparative effectiveness of radical prostatectomy and radiotherapy in prostate cancer: observational study of mortality outcomes. BMJ 348:g1502
Abdollah F, Sun M, Schmitges J et al (2012) Survival benefit of radical prostatectomy in patients with localized prostate cancer: estimations of the number needed to treat according to tumor and patient characteristics. J Urol 88:73–83
Yaxley JW, Coughlin GD, Chambers SK et al (2016) Robot-assisted laparoscopic prostatectomy versus open radical retropubic prostatectomy: early outcomes from a randomised controlled phase 3 study. Lancet 388:1057–1066
Ritch CR, You C, May AT et al (2014) Biochemical recurrence-free survival after robotic-assisted laparoscopic vs open radical prostatectomy for intermediate- and high-risk prostate cancer. Urology 83:1309–1315
Albadine R, Hyndman ME, Chaux A et al (2012) Characteristics of positive surgical margins in robotic-assisted radical prostatectomy, open retropubic radical prostatectomy, and laparoscopic radical prostatectomy: a comparative histopathologic study from a single academic center. Hum Pathol 43:254–260
Suardi N, Dell’Oglio P, Gallina A et al (2016) Evaluation of positive surgical margins in patients undergoing robot-assisted and open radical prostatectomy according to preoperative risk group. Urol Oncol 34:57, e1–e7
Jayram G, Decastro GJ, Large MC, Razmaria A, Zagaja GP, Shalhav AL, Brendler CB (2011) Robotic radical prostatectomy in patients with high-risk disease: a review of short-term outcomes from a high-volume center. J Endourol 25:455–457
Engel JD, Kao WW, Williams SB, Hong YM (2010) Oncologic outcome of robot-assisted laparoscopic prostatectomy in the high-risk setting. J Endourol 24:1963–1966
Acknowledgements
This study was supported by the following grants-in-aid for Scientific Research from the Japan Society for the Promotion of Science: 15H02563 (to CO), 15K15579 (to CO), and 17K11118 (to TK).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Informed consent
For this type of study, formal consent is not required. Pursuant to the provisions of the ethics committee and the ethic guideline in Japan, written consent was not required in exchange for public disclosure of study information in the case of retrospective and/or observational study using a material such as the existing documentation. The study information was open for the public consumption at http://www.med.hirosaki-u.ac.jp/~uro/html/IRB/IRBdoc.html.
Research involving human participants and/or animals
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Rights and permissions
About this article
Cite this article
Fujita, N., Koie, T., Hashimoto, Y. et al. Neoadjuvant chemohormonal therapy followed by robot-assisted and minimum incision endoscopic radical prostatectomy in patients with high-risk prostate cancer: comparison of perioperative and oncological outcomes at single institution. Int Urol Nephrol 50, 1999–2005 (2018). https://doi.org/10.1007/s11255-018-1985-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-018-1985-8