Abstract
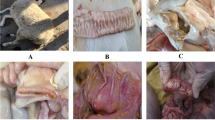
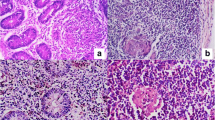
Camels are the prime source of meat and milk in many desert regions of the world including Saudi Arabia. Paratuberculosis of camels, locally called Silag, is a serious and invariably fatal disease in the Arabian camel. Six camels were used in this study. Five camels with clinical paratuberculosis were used to study the pathology of the disease and confirm its aetiology. The sixth camel was clinically healthy and used as a control. The camels were examined clinically and bled for haematological and blood chemistry analysis. They were then humanely killed with a high intravenous dose of thiopental sodium (10 mg/kg) for pathological studies as well as obtaining tissues for microbiological and molecular studies. The clinical signs of the disease were emaciation, diarrhoea, alopecia, wry neck and pale mucous membranes. Laboratory diagnosis showed reduced haemoglobin concentration, low haematocrit and high activity of the serum enzyme alanine aminotransferase. Serum creatinine concentration was normal. These results indicated the infected camels were anaemic and the function of their livers was affected. Postmortem examination showed thickened and corrugated intestinal mucosa, enlarged granulomatous mesenteric lymph nodes, miliary and diffuse granulomas in the liver (in four camels), generalized lymph node granulomas (in one camel), splenic granuloma (in one camel) and mediastinal lymph node granuloma (in two camels). Histopathological examination showed diffuse infiltration of macrophages in all organs showing lesions. Ziehl–Neelsen staining of tissue scraping and tissue sections showed masses of acid fast bacilli, except for the spleen. Infection with Mycobacterium avium subsp. paratuberculosis was confirmed by PCR by targeting the IS900 gene.


Similar content being viewed by others
References
Al-Dubaib, M. A. and Mahmoud, O. M., 2008. Paratuberculosis of goats at Qassim region of central Saudi Arabia. Bulgarian Journal of Veterinary Medicine 11 (1), 65 – 69.
Al-Hajr, S. M. and Altuwaimi, A. M., 2007. Elisa and PBR for evaluation of subclinical paratuberculosis in the Saudi diary herds. Veterinary Microbiology 121, 384 – 385.
Al-Shami, S. A., 2009. Comparative determination of serobiochemical constituents in-door and free grazing camels. Journal of Animal and Veterinary Advances 8, 896 – 898.
Beaudeau, F., Belliard, M., Joly, A. and Seegers, H., 2007. Reduction in milk yield associated with Mycibacterium avium subsp. paratuberculosis infection in dairy cows. Veterinary Research 38 (4), 625 – 634.
Bosshard, C., 2006. Mycobacterium avium subsp. paratuberculosis prevalence studies in bulk tank raw milk and slaughtered healthy dairy cows in Switzerland using an F57 sequence-based real-time PCR assay. Ph. D. thesis, University of Zurich.
Clarke, C. J., 1997. The pathology of paratuberculosis in ruminants and other species. Journal of Comparative Pathology 116, 217 – 261.
Cocito C., Gilot P., Coene, M., Kesel, M., Poupart, P. and Vannuffel, P., 1994. Paratuberculosis. Clinical Microbiology Reviews 7, 328–345.
Ilhan, Z., Solmaz, H., Aksakal, A., Gulhan, T., Ekin, I. H. and Boynukara, B., 2008. Detection of Brucella melitensis DNA in the milk of sheep after abortion by PCR assay. Archivos Medicina Veterinaria 40, 141–146.
Jones, T. C., Hunt, R. D. and King, N. W., 1997. Paratuberculosis. In: Veterinary Pathology, 6th Edition. Williams and Wilkins. Baltimore, Philadelphia, 498–501.
Kudahl, A. B. and Nielsen, S. S., 2009. Effect of paratuberculosis on slaughter weight and slaughter value of dairy cows. Journal of Dairy Science 92 (9), 4340 – 4346.
Mahmoud, O. M., Haroun, E. M. and Elfaki, M. G. and Abbas, B., 2002. Pigmented paratuberculosis granulomata in the liver of sheep. Small Ruminant Research 43, 211 – 217.
Mohamed, H. A. and Hussein, N. A. 1999. Studies on normal haematological and serum values of Hijin racing camels (Camelus dromdearius) in Kuwait. Veterinary Research Communications 24, 241 – 248.
Perez, V., Tellechea, J., Copra, J. M., Gutierrez, M. and Garcia Marin, J. F., 1999. Relation between pathologic findings and cellular immune response in sheep with naturally acquired paratuberculosis. American Journal of Veterinary Research 60, 123 – 127.
Sigurdardottir, O. G., Press, C. M., Saxegaard, F. and Evensen, O., 1999. Bacterial isolation, immunological response and histopathological lesions during the early subclinical phase of experimental infection of goat kids with Mycobacterium avium subsp. paratuberculosis. Veterinary Pathology 36, 542 – 550.
Stabel, J. R., 2000 Transitions in immune responses to Mycobacterium paratuberculosis. Veterinary Microbiology 77, 465–473.
Stabel, J. R., Bosworth, T. L., Kirkbride, T. A., Forde, R. L. and Whitlock, R. H., 2004. A simple, rapid, and effective method for the extraction of Mycobacterium paratuberculosis DNA from fecal samples for polymerase chain reaction. Journal of Veterinary Diagnostic Investigation 16, 22–30.
Tanaka, S., Itohara, S., Sato, M., Taniguchi, T. and Yokomizo, Y., 2000. Reduced formation of granulomatain gamma (delta) T cell and BALAB/c mice inoculated with Mycobacterium avium subsp. paratuberculosis. Veterinary Pathology 37, 415 – 421.
Wernery, U. and Kaaden, O. R., 2002. Paratuberculosis. In: Infectious Diseases of Camelids, 2nd edition. Blackwell Scientific Publication, UK, 83 – 90.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Alharbi, K.B., Al-Swailem, A., Al-Dubaib, M.A. et al. Pathology and molecular diagnosis of paratuberculosis of camels. Trop Anim Health Prod 44, 173–177 (2012). https://doi.org/10.1007/s11250-011-9905-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11250-011-9905-2