Abstract
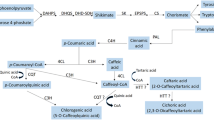
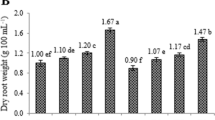
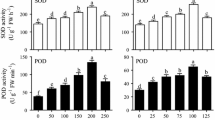
Madder (Rubia tinctorum L.) is a perennial plant that its roots and rhizomes have rich anthraquinone (AQ) derivatives including alizarin and purpurin. This study was carried out to determine the effects of salicylic acid (SA) and L-phenylalanine (L-Phe) applications on the root growth parameters and secondary metabolite accumulation in madder adventitious roots derived from internode parts without needing to be collected from nature. For this aim, two different L-Phe (50 and 100 µM) and SA (20 and 40 µM) were added separately and together to the liquid Murashige and Skoog (MS) medium cultured adventitious roots for 7 days. Then roots were evaluated in terms of fresh root weight, root growth index, dry root weight, and contents of total AQ, alizarin, purpurin, total phenolic contents (TPC), and some important phenolic compounds. According to the results, L-Phe stimulated the root growth of madder while the effects of SA on root growth parameters varied depending upon its concentrations. L-Phe had no significant influence on the total AQ, alizarin, and purpurin. Conversely, SA increased the AQs, and 20 µM SA was the most suitable application providing the greatest total AQ, alizarin, and purpurin. TPC and individual phenolic compounds changed according to the applications. Not only L-Phe but also SA had positive effects on the phenolic accumulation in adventitious roots. It was determined that the combinations of 40 µM SA and 100 µM L-Phe were the most effective applications in terms of phenolic accumulation.
Key message
Madder is a plant whose popularity has increased in food, cosmetics, pharmacy and recently medicine. This research was designed in order to produce high amounts of important secondary metabolites in adventitious root cultures of madder without the need to collect plants from nature. In this study, it has been shown that L-phenylalanine and salicylic acid applied to adventitious roots remarkably increase the production of anthraquinones and phenolics when used in appropriate concentrations and combinations.


Similar content being viewed by others
Abbreviations
- AQ:
-
Anthraquinone
- SA:
-
Salicylic acid
- L-Phe:
-
L-Phenylalanine
- IAA:
-
3-Indole acetic acid
- DW:
-
Dry weight
- TPC:
-
Total phenolic content
References
Açıkgöz MA, Kara ŞM, Aygün A, Özcan MM, Ay EB (2019) Effects of methyl jasmonate and salicylic acid on the production of camphor and phenolic compounds in cell suspension culture of endemic Turkish yarrow (Achillea gypsicola) species. TJAF 43:351–359. https://doi.org/10.3906/tar-1809-54
Al-Gendy AA, Bakr RO, El-Gindi OD (2015) Production of flavonoids and phenolic compounds by elicitation of Iphiona mucronata (Forssk.) Asch. & Schweinf (Asteraceae) callus and suspension cultures. Int J Pharmacogn Phytochem 30:1293–1300
Ali MB, Hahn EJ, Paek KY (2007) Methyl jasmonate and salicylic acid induced oxidative stress and accumulation of phenolics in Panax ginseng bioreactor root suspension cultures. Molecules 12:607–621. https://doi.org/10.3390/12030607
Angelini LG, Pistelli L, Belloni P, Bertoli A, Panconesi S (1997) Rubia tinctorum a source of natural dyes: agronomic evaluation, quantitative analysis of alizarin and industrial assays. Ind Crops Prod 6:303–311. https://doi.org/10.1016/S0926-6690(97)00021-6
Bányai P, Kuzovkina IN, Kursinszki L, Szőke É (2006) HPLC analysis of alizarin and purpurin produced by Rubia tinctorum L. hairy root cultures. Chromatographia 63:111–114. https://doi.org/10.1365/s10337-006-0792-z
Boudet AM (2007) Evolution and current status of research in phenolic compounds. Phytochem 68:2722–2735. https://doi.org/10.1016/j.phytochem.2007.06.012
Bozan B, Koşar M, Akyürek C, Ertuğrul K, Başer KHC (1999) Alizarin and purpurin contents of Rubia tinctorum L. roots collected from various regions of Turkey. Acta Pharm Sci 41:187–190
Bulgakov VP, Tchernoded GK, Mischenko NP, Khodakovskaya MV, Glazunov VP, Radchenko SV, Zhuravlev YN (2002) Effect of salicylic acid, methyl jasmonate, ethephon and cantharidin on AQ production by Rubia cordifolia callus cultures transformed with the rolB and rolC genes. J Biotechnol 97:213–221. https://doi.org/10.1016/s0168-1656(02)00067-6
Chaichana N, Dheeranupattana S (2012) Effects of methyl jasmonate and salicylic acid on alkaloid production from in vitro culture of Stemona sp. Int J Biosci Biochem Bioinforma 2:146–150. https://doi.org/10.7763/IJBBB.2012.V2.89
Chen Z, Silva S, Klessig D (1993) Active oxygen species in the induction of plant systemic acquired resistance by salicylic acid. Science 262:1883–1886. https://doi.org/10.1126/science.8266079
Cuoco G, Math C, Archier P, Chemat F, Vieillescazes C (2009) A multivariate study of the performance of an ultrasound-assisted madder dyes extraction and characterization by liquid chromatography-photodiode array detection. Ultrason Sonochem 16:75–82. https://doi.org/10.1016/j.ultsonch.2008.05.014
Demirci T, Çelikkol Akçay U, Göktürk Baydar N (2020) Effects of 24-epibrassinolide and L-phenylalanine on growth and caffeic acid derivatives production in hairy root culture of Echinacea purpurea L. MoenchActa Physiol Plant. https://doi.org/10.1007/s11738-020-03055-7
De Santis D, Moresi M (2007) Production of alizarin extracts from Rubia tinctorum and assessment of their dyeing properties. Ind Crops Prod 26:151–162. https://doi.org/10.1016/j.indcrop.2007.02.002
Derksen GC, Lelyveld GP, van Beek TA, Capelle A, de Groot Æ (2004) Two validated HPLC methods for the quantification of alizarin and other anthraquinones in Rubia tinctorum cultivars. Phytochem Anal 15:397–406. https://doi.org/10.1002/pca.800
Derksen GC, Van Beek TA (2002) Rubia tinctorum L. Stud Nat Prod Chem 26:629–684. https://doi.org/10.1016/S1572-5995(02)80016-3
Derksen GC, van Beek TA, de Groot Æ, Capelle A (1998) High-performance liquid chromatographic method for the analysis of anthraquinone glycosides and aglycones in madder root (Rubia tinctorum L.). J Chromatogr A 816:277–281. https://doi.org/10.1016/S0021-9673(98)00492-0
Desikan R, Reynolds A, Hancock JT, Neill SJ (1998) Harpin and hydrogen peroxide both initiate programmed cell death but have differential effects on defense gene expression in Arabidopsis suspension cultures. Biochem J 330:15–120. https://doi.org/10.1042/bj3300115
Dhingra V, Rao KV, Narasu ML (2000) Current status of artemisinin and its derivatives as antimalarial drugs. Life Sci 66:279–300. https://doi.org/10.1016/s0024-3205(99)00356-2
Dihazi A, Jaiti F, Zouine J, El Hassni M, El Hadrami I (2003) Effect of salicylic acid on phenolic compounds related to date palm resistance to Fusarium oxysporum f. sp. albedinis. Phytopath Medit 42:9–16. https://doi.org/10.14601/Phytopathol_Mediterr-1686
Ding CK, Wang CY, Gross KC (2002) Jasmonate and salicylate induce the expression of pathogenesis-related-protein genes and increase resistance to chilling injury in tomato fruit. Planta 21:895–901. https://doi.org/10.1007/s00425-001-0698-9
Dong J, Wan G, Liang Z (2010) Accumulation of salicylic acid-induced phenolic compounds and raised activities of secondary metabolic and anti-oxidative enzymes in Salvia miltiorrhiza cell culture. J Biotechnol 148:99–104. https://doi.org/10.1016/j.jbiotec.2010.05.009
Edahiro JI, Nakamura M, Seki M, Furusaki S (2005) Enhanced accumulation of anthocyanin in cultured strawberry cells by repetitive feeding of l-phenylalanine into the medium. J Biosci Bioeng 99:43–47. https://doi.org/10.1263/jbb.99.43
El Houssine Bouiamrine LB, Ibijbijen J, Nassiri L (2017) Fresh medicinal plants in middle atlas of Morocco: trade and threats to the sustainable harvesting. J Med Plants 5:123–128
El-Mawla AMAA (2012) Influence of certain abiotic elicitors on production of anthraquinones in cell cultures of Rubia tinctorum. Spatula DD 2:89–94. https://doi.org/10.5455/SPATULA.20120528021745
Giri A, Narasu ML (2000) Transgenic hairy roots. recent trends and applications. Biotechnol Adv 18:1–22. https://doi.org/10.1016/s0734-9750(99)00016-6
Govindaraju S, Arulselvi PI (2018) Effect of cytokinin combined elicitors (L-phenylalanine, salicylic acid and chitosan) on in vitro propagation, secondary metabolites and molecular characterization of medicinal herb–Coleus aromaticus Benth (L). J Saudi Soc Agric Sci 17:435–444. https://doi.org/10.1016/j.jssas.2016.11.001
Gaudeul M, Till-Bottraud I (2004) Reproductive ecology of the endangered Alpine species Eryngium alpinum L. (Apiaceae): phenology, gene dispersal and reproductive success. Ann Bot 93:711–721. https://doi.org/10.1093/aob/mch098
Hahlbrock K, Scheel D (1989) Physiology and molecular biology of phenylpropanoid metabolism. Ann Rev Plant Biol 40:347–369. https://doi.org/10.1146/annurev.pp.40.060189.002023
Han YS, Van der Heijden R, Verpoorte R (2001) Biosynthesis of anthraquinones in cell cultures of the Rubiaceae. Plant Cell Tiss Org 67:201–220. https://doi.org/10.1016/s0031-9422(01)00296-5
Hao W, Guo H, Zhang J, Hu G, Yao Y, Dong J (2014) Hydrogen peroxide is involved in salicylic acid-elicited rosmarinic acid production in Salvia miltiorrhiza cell cultures. The Science World J DOI. https://doi.org/10.1155/2014/843764
Jacob A, Malpathak N (2005) Manipulation of MS and B5 components for enhancement of growth and salsodine production in hairy root cultures of Solanum khasianum Clarke. Plant Cell Tiss Org 80:247–257. https://doi.org/10.1007/s11240-004-0740-2
Kalyoncu F, Cetin B, Saglam H (2006) Antimicrobial activity of common madder (Rubia tinctorum L.). Phytother Res 20:490–492. https://doi.org/10.1002/ptr.1884
Karuppusamy S (2009) A review on trends in production of secondary metabolites from higher plants by in vitro tissue, organ and cell cultures. J Med Plants Res 3:1222–1239. https://doi.org/10.5897/JMPR.9000026
Kawasaki Y, Goda Y, Yoshihira K (1992) The mutagenic constituents of Rubia tinctorum. Chem Pharm Bull 40:1504–1509. https://doi.org/10.1248/cpb.40.1504
Khan MIR, Fatma M, Per TS, Anjum NA, Khan NA (2015) Salicylic acid-induced abiotic stress tolerance and underlying mechanisms in plants. Front Plant Sci. https://doi.org/10.3389/fpls.2015.00462
Kinnosuko O, Takahito I, Keiko K (1991) Madders Dye in Plant Cell Culture in Japan (Atsuchi K (ed)), pp 138–142. CMC, Japan
Koca N, Karaman Ş (2015) The effects of plant growth regulators and l-phenylalanine on phenolic compounds of sweet basil. Food Chem 166:515–521. https://doi.org/10.1016/j.foodchem.2014.06.065
Krantev A, Yordanova R, Janda T, Szala G, Popova L (2008) Treatment with salicylic acid decreases the effect of cadmium on photosynthesis in maize plants. J Plant Physiol 165:920–931. https://doi.org/10.1016/j.jplph.2006.11.014
Kubota H, Sato K, Yamada T, Maitani T (1995) Phytochelatins (class lll metallothioneins) and their desglycyl peptides induced cadmium in normal root cultures of Rubia tinctorum. Plant Sci 110:157–166. https://doi.org/10.1016/0168-9452(95)04020-U
Lajkó E, Bányai P, Zámbó Z, Kursinszki L, Szőke E, Kőhidai L (2015) Targeted tumor therapy by Rubia tinctorum L.: Analytical characterization of hydroxyanthraquinones and investigation of their selective cytotoxic, adhesion and migration modulator effects on melanoma cell lines (A2058 and HT168-M1). Cancer Cell Int. https://doi.org/10.1186/s12935-015-0271-4
Largia MJV, Pothiraj G, Shilpha J, Ramesh M (2015) Methyl jasmonate and salicylic acid synergism enhances bacoside a content in shoot cultures of Bacopa monnieri L. Plant Cell Tiss Org 122:9–20. https://doi.org/10.1007/s11240-015-0745-z
Mahalakshmi R, Eganathan P, Parida AK (2013) Salicylic acid elicitation on production of secondary metabolite by cell cultures of Jatropha curcas L. J Pharm Pharmaceut Sci 5:655–659
Mathur L, Yadav RK (2011) Effect of salicylic acid on trigonelline production in Trigonella foenum-graecum L. cell suspension culture. Inter Referred Res J 1:137–138
Mobin M, Wu CH, Tewari RK, Paek KY (2015) Studies on the glyphosate induced amino acid starvation and addition of precursors on caffeic acid accumulation and profiles in adventitious roots of Echinacea purpurea (L.) Moench. Plant Cell Tiss Org 120:291–301. https://doi.org/10.1007/s11240-014-0606-1
Mora-Herrera ME, Peralta-Velázquez J, López-Delgado HA, García-Velasco R, González-Díaz JG (2011) Efecto del ácido ascórbico sobre crecimiento, pigmentos fotosintéticos y actividad peroxidasa en plantas de crisantemo. Rev Chapingo Ser Hortic 17:73–81
Mulabagal V, Tsay HS (2004) Plant cell cultures-an alternative and efficient source for the production of biologically important secondary metabolites. Int J Appl Sci Eng 2:29–48
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:472–497
Orban N, Boldizar I, Szücs Z, Bela D (2008) Influence of different elicitors on the synthesis of anthraquinone derivatives in Rubia tinctorum L. cell suspension cultures. Dyes Pigm 77:249–257. https://doi.org/10.1016/j.dyepig.2007.03.015
Palacio L, Cantero JJ, Cusidó R, Goleniowski M (2011) Phenolic compound production by Larrea divaricata Cav. plant cell cultures and effect of precursor feeding. Process Biochem 46:418–422. https://doi.org/10.1016/j.procbio.2010.08.029
Raskin I (1992) Role of salicylic acid in plants. Ann Rev Plant Biol 43:439–463. https://doi.org/10.1146/annurev.pp.43.060192.002255
Rivas-San Vicente M, Plasencia J (2011) Salicylic acid beyond defense: Its role in plant growth and development. J Exp Bot 62:3321–3338. https://doi.org/10.1093/jxb/err031
Sánchez-Chávez E, Barrera-Tovar R, Muñoz-Márquez E, Ojeda-Barrios DL (2011) Efecto del ácido salicílico sobre biomasa, actividad fotosintética, contenido nutricional y productividad del chile jalapeño. Rev Chap Ser Hortic 17:63–68
Schulte U, El-Shagi H, Zenk MN (1984) Optimization of 19 Rubiaceae species in cell culture for the production of anthraquinones. Plant Cell Rep 3:51–54. https://doi.org/10.1007/BF00270970
Senaratna T, Touchell D, Bunn E, Dixon K (2000) Acetylsalicylic acid (aspirin) and salicylic acid induce multiple stress tolerance in bean and tomato plant. Plant Growth Regul 30:157–161. https://doi.org/10.1186/s40529-018-0222-1
Singleton VL, Rossi JR (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid. Am J Enol Vitic 16:144–158
Sudha G, Ravishankar GA (2003) Elicitation of anthocyanin production in callus cultures of Daucus carota and the involvement of methyl jasmonate and salicylic acid. Acta Physiol Plant 25:249–256. https://doi.org/10.1007/s11738-003-0005-4
Thiruvengadam M, Rekha K, Rajakumar G, Lee TJ, Kim SH, Chung IM (2016) Enhanced production of Anthraquinones and Phenolic Compounds and biological activities in the cell suspension cultures of Polygonum multiflorum. Int J Mol Sci 17:1912. https://doi.org/10.3390/ijms17111912
Vázquez-Díaz DA, Salas-Pérez L, Preciado-Rangel P, Segura-Castruita MA, González-Fuentes JA, Valenzuela-García JR (2016) Efecto del ácido salicílico en la producción y calidad nutracéutica de frutos de tomate. Rev Mex Cienc Agríc 17:3405–3414
Westendorf J, Marquardt H, Poginsky B, Dominiak M, Schmidt J, Marquardt H (1990) Genotoxicity of naturally occurring hydroxyanthraquinones. Mutat Res 240:1–12. https://doi.org/10.1016/0165-1218(90)90002-j
Zhao J, Davis LC, Verpoorte R (2004) Elicitor signal transduction leading to production of plant secondary metabolites. Biotech Advanced 23:283–333. https://doi.org/10.1016/j.biotechadv.2005.01.003
Funding
The authors are thankful to TUBITAK (Scientific and Technological Research Council of Turkey) for the financial support for this research Project (TOVAG, Project No: 215O057).
Author information
Authors and Affiliations
Contributions
The authors have made the following declarations regarding their contributions: TD and NGB conceived the design of the experiments. TD and ÖAA monitored the research work. TD collected and analyses sample data. TD and NGB contributed to writing the manuscript. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Ali R. Alan.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Demirci, T., Aras Ascı, Ö. & Göktürk Baydar, N. Influence of salicylic acid and L-phenylalanine on the accumulation of anthraquinone and phenolic compounds in adventitious root cultures of madder (Rubia tinctorum L.). Plant Cell Tiss Organ Cult 144, 313–324 (2021). https://doi.org/10.1007/s11240-020-01952-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-020-01952-w