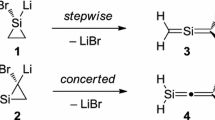
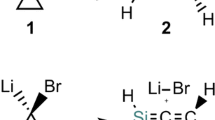
Abstract
Computational analysis has been carried out to understand the electronic effect of substituents on the reactivity of silenes. Three types of reactions, viz., (i) dimerization of silenes, (ii) small molecule activation (NH3 and NO), and (iii) isomerization of silene to silylene, were taken for the analysis using monosubstituted silenes (RHC = SiH2 and H2C = SiRH) and disubstituted silene (HRC = SiRH) with substituents –CH3, –SiH3, –OH, –CN, and –F. It is found that the position of the substituent is decisive in C-Si bond polarity as well as the reactivity of silenes. The dimerization of silenes prefers the diradical pathway, and the presence of π-donating substituents gives a better stabilization of the free energy profile. The N–H and N–O activation by silenes forms thermodynamically stable adducts, hinting at the unexplored potential of silenes in small molecule activation. Thermodynamic and kinetic feasibility of the isomerization of silenes to silylenes can also be achieved by the introduction of π-donating substituents. The present analysis suggests that the chemical reactivity of silenes can be significantly controlled by the fine tuning of the electronic effects of substituents.











Similar content being viewed by others
Data availability
Cartesian coordinates of all structures, isodesmic reaction schemes, and the results at the PW6B95D3/6–311 + + G(d,p) calculations are available in the Supplementary Materials section.
Code availability
Not applicable.
References
Gusel’Nikov LE, Flowers MC (1967) The thermal decomposition of 1,1-dimethyl-1-silacyclobutane and some reactions of an unstable intermediate containing a silicon–carbon double bond. Chem Commun (London) 864–865. https://doi.org/10.1039/C19670000864
Brook AG, Nyburg SC, Abdesaken F et al (1982) Stable solid silaethylenes. J Am Chem Soc 104:5667–5672. https://doi.org/10.1021/ja00385a019
Brook AG, Krishna R, Kallury MR, Poon YC (1982) Attempted stabilization of silaethylenes with aryl or trifluoromethyl groups. Organometallics 1:987–994. https://doi.org/10.1021/om00067a017
Brook AG, Abdesaken F, Gutekunst B et al (1981) A solid silaethene: isolation and characterization. J Chem Soc Chem Commun 191. https://doi.org/10.1039/c39810000191
Brook AG, Harris JW, Lennon J, El Sheikh M (1979) Relatively stable silaethylenes. Photolysis of acylpolysilanes. J Am Chem Soc 101:83–95. https://doi.org/10.1021/ja00495a015
Apeloig Y, Karni M (1984) Substituent effects of the carbon-silicon double bond. monosubstituted silenes. J Am Chem Soc 106:6676–6682. https://doi.org/10.1021/ja00334a036
Bendikov M, Quadt SR, Rabin O, Apeloig Y (2002) Addition of nucleophiles to silenes. A theoretical study of the effect of substituents on their kinetic stability. Organometallics 21:3930–3939. https://doi.org/10.1021/om0202571
El-Sayed I, Guliashvili T, Hazell R et al (2002) Evidence for formation of silenes strongly influenced by reversed Si=C bond polarity. Org Lett 4:1915–1918. https://doi.org/10.1021/ol025920w
El-Nahas AM, Johansson M, Ottosson H (2003) Reverse Si=C bond polarization as a means for stabilization of silabenzenes: a computational investigation. Organometallics 22:5556–5566. https://doi.org/10.1021/om030417o
Leigh WJ, Boukherroub R, Kerst C (1998) Substituent effects on the reactivity of the silicon−carbon double bond. resonance, inductive, and steric effects of substituents at silicon on the reactivity of simple 1-methylsilenes. J Am Chem Soc 120:9504–9512. https://doi.org/10.1021/ja981435d
Morkin TL, Leigh WJ (2001) Substituent effects on the reactivity of the silicon−carbon double bond. Acc Chem Res 34:129–136. https://doi.org/10.1021/ar960252y
Bradaric CJ, Leigh WJ (1997) Substituent effects on the reactivity of the silicon–carbon double bond. Arrhenius parameters for the reaction of 1,1-diarylsilenes with alcohols and acetic acid. Can J Chem 75:1393–1402. https://doi.org/10.1139/v97-167
Leigh WJ, Moiseev AG, Coulais E et al (2008) Substituent effects on silene reactivity - reactive silenes from photolysis of phenylated tri- and tetrasilanes. Can J Chem 86:1105–1117. https://doi.org/10.1139/V08-165
Pavelka LC, Hanson MA, Staroverov VN, Baines KM (2014) Mechanism of the addition of alkynes to silenes and germenes: a density functional study. Can J Chem 93:134–142. https://doi.org/10.1139/cjc-2014-0256
Ottosson H (2003) Zwitterionic silenes: interesting goals for synthesis? Chem - A Eur J 9:4144–4155. https://doi.org/10.1002/chem.200204583
Ottosson H, Eklöf AM (2008) Silenes: connectors between classical alkenes and nonclassical heavy alkenes. Coord Chem Rev 252:1287–1314. https://doi.org/10.1016/j.ccr.2007.07.005
Müller T, Ziche W, Auner N (1998) Silicon–carbon and silicon–nitrogen multiply bonded compounds. In: Rappoport Z, Apeloig Y (eds) 2nd ed. John Wiley & Sons, Ltd, pp 857–1062
Baines KM (2013) Brook silenes: inspiration for a generation. Chem Commun 49:6366–6369. https://doi.org/10.1039/c3cc42595a
Hardwick JA, Baines KM (2011) Addition of nitriles to two brook silenes. Organometallics 30:2831–2837. https://doi.org/10.1021/om200182q
Tanaka H, Shiota Y, Hori K et al (2012) Substituent effects in thermal reactions of a silene with silyl-substituted alkynes: a theoretical study. Organometallics 31:4737–4747. https://doi.org/10.1021/om300310g
Takahashi M, Veszprémi T, Kira M (2004) 1,2-Addition reaction of monosubstituted disilenes: an ab initio study. Organometallics 23:5768–5778. https://doi.org/10.1021/om049418m
Veszprémi T, Takahashi M, Hajgató B, Kira M (2001) The mechanism of 1,2-addition of disilene and silene. 1. Water and alcohol addition. J Am Chem Soc 123:6629–6638. https://doi.org/10.1021/ja0040823
Hajgató B, Takahashi M, Kira M, Veszprémi T (2002) The mechanism of 1,2-addition of disilene and silene: hydrogen halide addition. Chem - A Eur J 8:2126–2133. https://doi.org/10.1002/1521-3765(20020503)8:9%3c2126::AID-CHEM2126%3e3.0.CO;2-2
Lee PTK, Rosenberg L (2016) Scope and selectivity of B(C6F5)3-catalyzed reactions of the disilane (Ph2SiH)2. J Organomet Chem 809:86–93. https://doi.org/10.1016/j.jorganchem.2016.02.035
Sen SS, Hey J, Herbst-Irmer R et al (2011) Striking stability of a substituted silicon(II) bis(trimethylsilyl)amide and the facile Si–Me bond cleavage without a transition metal catalyst. J Am Chem Soc 133:12311–12316. https://doi.org/10.1021/ja205369h
Su B, Kostenko A, Yao S, Driess M (2020) Isolable dibenzo[a, e ]disilapentalene with a dichotomic reactivity toward CO 2. J Am Chem Soc 142:16935–16941. https://doi.org/10.1021/jacs.0c09040
Poitiers NE, Huch V, Zimmer M, Scheschkewitz D (2020) Nickel-assisted complete cleavage of CO by a silylene/siliconoid hybrid under formation of an Si-C enol ether bridge. Chem Commun 56:10898–10901. https://doi.org/10.1039/D0CC04922K
Sun X, Hinz A, Kucher H et al (2022) Stereoselective Activation of Small Molecules by a Stable Chiral Silene. Chem A Eur J. https://doi.org/10.1002/chem.202201963
Zborovsky L, Kostenko A, Bravo-Zhivotovskii D, Apeloig Y (2019) Mechanism of the thermal Z⇌E isomerization of a stable silene; experiment and theory. Angew Chemie 131:14666–14670. https://doi.org/10.1002/ange.201907864
Jabłoński M, Krygowski TM (2020) Changes in electron structure of the triple bond in substituted acetylene and diacetylene derivatives. ChemPhysChem 21:1847–1857. https://doi.org/10.1002/cphc.202000378
Campos-Fernández L, Barrientos-Salcedo C, Herrera Valencia EE et al (2019) Substituent effects on the stability, physicochemical properties and chemical reactivity of nitroimidazole derivatives with potential antiparasitic effect: a computational study. New J Chem 43:11125–11134. https://doi.org/10.1039/C9NJ02207D
Exner O, Bohm S (2006) Theory of substituent effects: recent advances. Curr Org Chem 10:763–778. https://doi.org/10.2174/138527206776818892
Becke AD (1993) Density-functional thermochemistry. III. The role of exact exchange. J Chem Phys 98:5648–5652. https://doi.org/10.1063/1.464913
Lee C, Yang W, Parr RG (1988) Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys Rev B 37:785–789. https://doi.org/10.1103/PhysRevB.37.785
Frisch MJ, Trucks GW, Schlegel HB et al (2016) G16_C01. Gaussian 16, Revision C.01, Gaussian, Inc., Wallin
Dimitrova V, Ilieva S, Galabov B (2003) Electrostatic potential at nuclei as a reactivity index in hydrogen bond formation. Complexes of ammonia with C-H, N–H and O–H proton donor molecules. J Mol Struct THEOCHEM 637:73–80. https://doi.org/10.1016/S0166-1280(03)00402-0
Mathew J, Suresh CH (2010) Use of molecular electrostatic potential at the carbene carbon as a simple and efficient electronic parameter of N-heterocyclic carbenes. Inorg Chem 49. https://doi.org/10.1021/ic1004243
Suresh CH, Remya GS, Anjalikrishna PK (2022) Molecular electrostatic potential analysis: a powerful tool to interpret and predict chemical reactivity. WIREs Comput Mol Sci. https://doi.org/10.1002/wcms.1601
Gusel’nikov LE, Avakyan VG, Guselnikov SL, (2002) Effect of geminal substitution at silicon on 1-sila- and 1,3-disilacyclobutanes’ strain energies, their 2+2 cycloreversion enthalpies, and Si=C π-bond energies in silenes. J Am Chem Soc 124:662–671. https://doi.org/10.1021/ja011287i
Ishikawa M (1978) Photolysis of organopolysilanes. Generation and reactions of silicon-carbon double-bonded intermediates. Pure Appl Chem 50:11–18. https://doi.org/10.1351/pac197850010011
Leigh WJ (1999) Kinetics and mechanisms of the reactions of Si=C and Ge=C double bonds. Pure Appl Chem 71:453–462. https://doi.org/10.1351/pac199971030453
Bernardi F, Bottoni A, Olivucci M et al (1993) Does a concerted path exist for the head-to-tail [2.pi.S + 2.pi.S] cycloaddition of silaethylene? J Am Chem Soc 115:3322–3323. https://doi.org/10.1021/ja00061a038
Venturini A, Bernardi F, Olivucci M et al (1998) Dimerization of silaethylene: computational evidence for a novel mechanism for the formation of 1,3-disilacyclobutane via a 1,2 approach. J Am Chem Soc 120:1912–1913. https://doi.org/10.1021/ja973472v
Seidl ET, Grev RS, Schaefer HF (1992) Mechanistic, structural, and vibrational aspects of the dimerization of silaethylene. J Am Chem Soc 114:3643–3650. https://doi.org/10.1021/ja00036a011
Zhang S, Conlin RT, McGarry PF, Scaiano JC (1992) Reaction kinetics, quantum yields, and product studies for the dimerization of a stabilized silene. Organometallics 11:2317–2319. https://doi.org/10.1021/om00042a058
Acknowledgements
The authors are grateful to Rashtriya Uchchatar Shiksha Abhiyan (RUSA) and University Grants Commission (UGC) for the financial support. AK thanks Kerala State Council for Science, Technology and Environment (KSCSTE) for a fellowship.
Author information
Authors and Affiliations
Contributions
Major part of the work has been done by A. K. and J. J. M. J. M. wrote the main manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kizhuvedath, A., Mallikasseri, J.J. & Mathew, J. Electronic effects of substituents on the reactivity of silenes: a computational analysis. Struct Chem 35, 119–133 (2024). https://doi.org/10.1007/s11224-023-02169-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-023-02169-1