Abstract
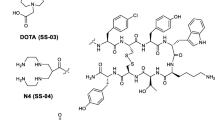
The complex of the short somatostatin analog, DOTA—Phe—D-Trp—Lys—Thr—OMe (DOTA is 1,4,7,10-tetrraazacyclododecane-1,4,7,10-tetraacetic acid), with the theragnostic 155Tb radionuclide was obtained for the first time. The radionuclide was generated in a cyclotron according to the reaction natGd(α,x)155Dy⟶155Tb. The complex was characterized by thin layer chromatography. The in vitro stability was assessed in saline and in the presence of bovine serum. The complex showed a satisfactory stability under these conditions for 24 h.
Similar content being viewed by others
References
S. W. J. Lamberts, E. P. Krenning, J. C. Reubi, Endocr. Rev., 1991, 12, 450–482; DOI: https://doi.org/10.1210/edrv-12-4-450.
R. Garcia-Carbonero, R. Garcia-Figueiras, A. Carmona-Bayonas, I. Sevilla, A. Teule, M. Quindos, E. Grande, J. Capdevila, J. Aller, J. Arbizu, P. Jimenez-Fonseca, Cancer Metastasis Rev., 2015, 34, 823–842; DOI: https://doi.org/10.1007/s10555-015-9598-5.
A. Dash, S. Chakraborty, M. R. A. Pillai, F. F. R. Knapp, Cancer Biother. Radiopharm., 2015, 30, 47–71; DOI: https://doi.org/10.1089/cbr.2014.1741.
D. F. Veber, R. M. Freidinger, D. S. Perlow, W. J. Paleveda, F. W. Holly, R. G. Strachan, R. F. Nutt, B. H. Arison, C. Homnick, W. C. Randall, M. S. Glitzer, R. Saperstein, R. Hirschmann, Nature, 1981, 292, 55–58; DOI: https://doi.org/10.1038/292055a0.
A. N. Balaev, V. N. Osipov, D. S. Khachatryan, Pharm. Chem. J., 2015, 49, 345–351; DOI: https://doi.org/10.1007/s11094-015-1284-y.
A. N. Balaev, V. N. Osipov, K. A. Okhmanovich, E. A. Ruchko, A. V. Kolotaev, D. S. Khachatryan, Russ. Chem. Bull., 2016, 65, 2766–2769; DOI: https://doi.org/10.1007/s11172-016-1651-1.
A. Yakusheva, N. Titchenko, B. Egorova, E. Matazova, N. Podkhalyuzina, V. Osipov, D. Khachatryan, D. Av-deev, G. Posypanova, S. Kalmykov, J. Label. Compd. Radiopharm., 2019, 62, 718–728; DOI: https://doi.org/10.1002/jlcr.3799.
G. Y. Aleshin, S. Y. Khabirova, V. N. Osipov, D. S. Khachatryan, INEOS OPEN, 2020, 2, 200–204; DOI: https://doi.org/10.32931/io1929a.
M. de Jong, W. A. P. Breeman, B. F. Bernard, E. J. Rolleman, L. J. Hoflande, T. J. Visser, B. Setyono-Han, W. H. Bakker, M. E. van der Pluijm, E. P. Krenning, Eur. J. Nucl. Med., 1995, 22, 608–616; DOI: https://doi.org/10.1007/BF01254561.
C. Müller, C. Vermeulen, K. Johnston, U. Köster, R. Schmid, A. Türler, N. P. van der Meulen, EJNMMI Res., 2016, 6, 35; DOI: https://doi.org/10.1186/s13550-016-0189-4.
F. Borgna, P. Barritt, P. V. Grundler, Z. Talip, S. Cohrs, J. R. Zeevaart, U. Köster, R. Schibli, N. P. van der Meulen, C. Müller, Pharmaceutics, 2021, 13, 536; DOI: https://doi.org/10.3390/pharmaceutics13040536.
M. F. Loncin, J. F. Desreux, E. Merciny, Inorg. Chem., 1986, 25, 2646–2648; DOI: https://doi.org/10.1021/ic00235a031.
J. Byegård, G. Skarnemark, M. Skælberg, J. Radioanal. Nucl. Chem., 1999, 241, 281–290; DOI: https://doi.org/10.1007/BF02347463.
P. Comba, U. Jermilova, C. Orvig, B. O. Patrick, C. F. Ramogida, K. Rück, C. Schneider, M. Starke, Chem.–A Eur. J., 2017, 23, 15945–15956; DOI: https://doi.org/10.1002/chem.201702284.
X. Wang, M. D. G. Jaraquemada-Peláez, C. Rodríguez-Rodríguez, Y. Cao, C. Buchwalder, N. Choudhary, U. Jermilova, C. F. Ramogida, K. Saatchi, U. O. Häfeli, B. O. Patrick, C. Orvig, J. Am. Chem. Soc., 2018, 140, 15487–15500; DOI: https://doi.org/10.1021/jacs.8b09964.
N. Choudhary, A. Dimmling, X. Wang, L. Southcott, V. Radchenko, B. O. Patrick, P. Comba, C. Orvig, Inorg. Chem., 2019, 58, 8685–8693; DOI: https://doi.org/10.1021/acs.inorgchem.9b01016.
A. P. Orlov, T. P. Trofimova, M. A. Orlova, Russ. Chem. Bull., 2022, 71, 415–429; DOI: https://doi.org/10.1007/s11172-022-3429-y.
P. A. Demakov, V. P. Fedin, Russ. Chem. Bull., 2022, 71, 967–973, DOI: https://doi.org/10.1007/s11172-022-3498-y.
C. Muller, K. Zhernosekov, U. Koster, K. Johnston, H. Dorrer, A. Hohn, N. T. van der Walt, A. Turler, R. Schibli, J. Nucl. Med., 2012, 53, 1951–1959; DOI: https://doi.org/10.2967/jnumed.112.107540.
D. Filosofov, E. Kurakina, V. Radchenko, Nucl. Med. Biol., 2021, 94–95, 1–19; DOI: https://doi.org/10.1016/j.nucmedbio.2020.12.001.
C. Müller, E. Fischer, M. Behe, U. Köster, H. Dorrer, J. Reber, S. Haller, S. Cohrs, A. Blanc, J. Grünberg, M. Bunka, K. Zhernosekov, N. van der Meulen, K. Johnston, A. Türler, R. Schibli, Nucl. Med. Biol., 2014, 41, 58–65; DOI: https://doi.org/10.1016/j.nucmedbio.2013.11.002.
F. Tárkáönyi, F. Ditrói, S. Takács, A. Hermanne, A. V. Ignatyuk, Ann. Nucl. Energy, 2013, 62, 375–381; DOI: https://doi.org/10.1016/j.anucene.2013.06.038.
F. Tárkányi, F. Ditrói, S. Takács, A. Hermanne, A. V. Ignatyuk, Appl. Radiat. Isot., 2015, 98, 87–95; DOI: https://doi.org/10.1016/j.apradiso.2015.01.015.
C. Favaretto, Z. Talip, F. Borgna, P. V. Grundler, G. Dellepiane, A. Sommerhalder, H. Zhang, R. Schibli, S. Braccini, C. Müller, N. P. van der Meulen, EJNMMI Radiopharm. Chem., 2021, 6, 37; DOI: https://doi.org/10.1186/s41181-021-00153-w.
F. Szelecsényi, Z. Kovács, K. Nagatsu, M.-R. Zhang, K. Suzuki, J. Radioanal. Nucl. Chem., 2016, 307, 1877–1881; DOI: https://doi.org/10.1007/s10967-015-4528-0.
A. N. Moiseeva, R. A. Aliev, E. B. Furkina, V. I. Novikov, V. N. Unezhev, Nucl. Med. Biol., 2022, 106–107, 52–61; DOI: https://doi.org/10.1016/j.nucmedbio.2021.12.004.
A. N. Moiseeva, R. A. Aliev, E. S. Kormazeva, S. T. Latushkin, T. Y. Malamut, K. A. Makoveeva, V. I. Novikov, V. N. Unezhev, E. B. Furkina, V. A. Zag-ryadskiy, Appl. Radiat. Isot., 2021, 170, 109609; DOI: https://doi.org/10.1016/j.apradiso.2021.109609.
A. Hermanne, A. V. Ignatyuk, R. Capote, B. V. Carlson, J. W. Engle, M. A. Kellett, T. Kibédi, G. Kim, F. G. Kondev, M. Hussain, O. Lebeda, A. Luca, Y. Nagai, H. Naik, A. L. Nichols, F. M. Nortier, S. V. Suryanarayana, S. Takács, F. T. Tárkányi, M. Verpelli, Nucl. Data Sheets, 2018, 148, 338–382; DOI: https://doi.org/10.1016/j.nds.2018.02.009.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare no competing interests.
Additional information
The authors are grateful to B. V. Egorova for valuable advice during planning of the experiment.
The production of the 155Tb radionuclide was financially supported by the Ministry of Science and Education of the Russian Federation (agreement No. 075-15-2021-1360). Analytical studies were performed using the research equipment of the Center for Collective Use of the Institute of Chemical Reagents and High-Purity Chemical Substances, National Research Center, Kurchatov Institute, with the financial support of the project from the Ministry of Science and Education of the Russian Federation (agreement No. 075-15-2022-1157 dated August 16, 2022). The study was supported by the National Research Center, Kurchatov Institute (order No. 2751).
No human or animal subjects were used in this research.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, Vol. 72, No. 9, pp. 2249—2254, September, 2023.
Rights and permissions
About this article
Cite this article
Moiseeva, A.N., Aliev, R.A., Osipov, V.N. et al. Preparation of 155Tb-labeled short somatostatin analog. Russ Chem Bull 72, 2249–2254 (2023). https://doi.org/10.1007/s11172-023-4022-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-023-4022-8