Abstract
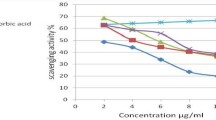
A series of new substituted chalcones with different positions of the isobornyl substituent in aromatic rings were synthesized in moderate and high yields by condensation of O-methoxy- and O-allyloxy derivatives of acetophenone with various benzaldehyde derivatives in the presence of sodium hydride in DMF. It was established that the presence of two allyloxy groups in the acylated derivatives of terpenophenols favors an increase in the yield of the substituted chalcone. A comparative evaluation of the antioxidant properties of the isobornylchalcones obtained was performed. It was shown that all compounds synthesized in this work exhibit pronounced antioxidant activity; however, particular characteristics depend strongly on the model system used. It was established that the presence, number, and positions of methoxy groups in the A- and B-rings are of importance for the implementation of the antioxidant activity of isobornylchalcones.
Similar content being viewed by others
References
R. Saxena, in Aging: Oxidative Stress and Dietary Antioxidants, Ed. V. R. Preedy, Academic Press, Oxford, 2014, p. 49; DOI: https://doi.org/10.1016/B978-0-12-405933-7.00005-6.
A. M. Pisoschi, A. Pop, Eur. J. Med. Chem., 2015, 97, 55; DOI: https://doi.org/10.1016/j.ejmech.2015.04.040.
N. A. Al Zahrani, R. M. El-Shishtawy, M. M. Elaasser, A. M. Asiri, Molecules, 2020, 25, 4566; DOI: https://doi.org/10.3390/molecules25194566.
G. Yang, H. E. Lee, S. H. Yeon, H. C. Kang, Y. Y. Cho, H. S. Lee, C. C. Zouboulis, S. H. Han, J. H. Lee, J. Y. Lee, Phytotherapy Res., 2018, 32, 2551; DOI: https://doi.org/10.1002/ptr.6195.
Z. Rozmer, P. Perjési, Phytochem. Rev., 2016, 15, 87; DOI: https://doi.org/10.1007/s11101-014-9387-8.
V. R. D. Pereira, I. J. A. Junior, L. S. Silveira, R. B. Geraldo, P. F. Pinto, F. S. Teixeira, M. C. Salvadori, M. P. Silva, L. A. Alves, P. V. S. Z. Capriles, A. C. Almeida, E. S. Coimbra, P. L. S. Pinto, M. R. C. Couri, J. Moraes, A. A. S. Filho, Chem. Biodiversity, 2018, 15, e1800398; DOI: https://doi.org/10.1002/cbdv.201800398.
M. J. Mphahlele, S. J. Zamisa, T. M. El-Gogary, J. Mol. Struct., 2021, 1245, 131090; DOI: https://doi.org/10.1016/j.molstruc.2021.131090.
T. I. Adelusi, G. R. Akinbolaji, X. Yin, K. S. Ayinde, O. T. Olaoba, Eur. J. Pharmacol., 2021, 891, 173695; DOI: https://doi.org/10.1016/j.ejphar.2020.173695.
T. I. Adelusi, L. Du, A. Chowdhury, G. Xiaoke, Q. Lu, X. Yin, Life Sci., 2021, 284, 118982; DOI: https://doi.org/10.1016/j.lfs.2020.118982.
R. M. Hassan, M. E. Aboutabl, M. Bozzi, M. F. El-Behairy, A. M. El Kerdawy, B. Sampaolese, C. Desiderio, F. Vincenzoni, F. Sciandra, I. A. Y. Ghannam, Bioorg. Chem., 2021, 115, 105170; DOI: https://doi.org/10.1016/j.bioorg.2021.105170.
S. Ammaji, S. Masthanamma, R. R. Bhandare, S. Annadurai, A. Basha Shaik, Arab. J. Chem., 2022, 15, 103581; DOI: https://doi.org/10.1016/j.arabjc.2021.103581.
O. A. Nurkenov, M. K. Ibraev, M. B. Isabaeva, I. A. Schepetkin, A. I. Khlebnikov, T. M. Seilkhanov, A. E. Arinova, Russ. J. Gen. Chem., 2019, 89, 1360; DOI: https://doi.org/10.1134/S0044460X19070023.
I. S. Odin, S. Cao, D. Hughes, E. V. Zamaratskii, Yu. P. Zarubin, P. P. Purygin, A. A. Golovanov, S. S. Zlotskii, Dokl. Chem., 2020, 492, 89; DOI: https://doi.org/10.1134/S0012500820360021.
E. V. Buravlev, O. G. Shevchenko, A. V. Kutchin, Russ. Chem. Bull., 2021, 70, 183; DOI: https://doi.org/10.1007/s11172-021-3075-9.
E. V. Buravlev, I. V. Fedorova, O. G. Shevchenko, A. V. Kutchin, Russ. Chem. Bull., 2020, 69, 1573; DOI: https://doi.org/10.1007/s11172-020-2937-x.
N. A. Zefirov, A. V. Mamaeva, A. I. Krasnoperova, Yu. A. Evteeva, E. R. Milaeva, S. A. Kuznetsov, O. N. Zefirova, Russ. Chem. Bull., 2021, 70, 549; DOI: https://doi.org/10.1007/s11172-021-3123-5.
P. M. Sivakumar, P. K. Prabhakar, M. Doble, Med. Chem. Res., 2011, 20, 482; DOI: https://doi.org/10.1007/s00044-010-9342-1.
B.-T. Kim, K.-Z. O, J.-C. Chun, K. J. Hwang, Bull. Korean. Chem. Soc., 2008, 29, 1125; DOI: https://doi.org/10.5012/bkcs.2008.29.6.1125.
V. P. Osipova, M. A. Polovinkina, L. R. Telekova, A. V. Velikorodov, N. N. Stepkina, N. T. Berberova, Rus. Chem. Bull., 2020, 69, 504; DOI: https://doi.org/10.1007/s11172-020-2790-y.
M. A. Polovinkina, V. P. Osipova, A. D. Osipova, N. T. Berberova, A. V. Velikorodov, G. G. Matishov, Dokl. Chem., 2021, 500, 184; DOI: https://doi.org/10.1134/S0012500821090032.
S. Marquina, M. Maldonado-Santiago, J. N. Sánchez-Carranza, M. Antúnez-Mojica, L. González-Maya, R. S. Razo-Hernández, L. Alvarez, Bioorg. Med. Chem., 2019, 27, 43; DOI: https://doi.org/10.1016/j.bmc.2018.10.045.
E. Saavedra, H. Del Rosario, I. Brouard, J. Quintana, F. Estévez, Chem. Bio. Int., 2019, 298, 137; DOI: https://doi.org/10.1016/j.cbi.2018.12.010.
D. K. Mahapatra, S. K. Bharti, V. Asati, Eur. J. Med. Chem., 2015, 98, 69; DOI: https://doi.org/10.1016/j.ejmech.2015.05.004.
S. A. Popova, E. V. Pavlova, I. Yu. Chukicheva, Russ. Chem. Bull., 2020, 69, 2198; DOI: https://doi.org/10.1007/s11172-020-3022-1.
S. A. Popova, E. V. Pavlova, I. Yu. Chukicheva, ARKIVOC, 2021, 8, 179; DOI: https://doi.org/10.24820/ark.5550190.p011.471.
S. A. Popova, E. V. Pavlova, O. G. Shevchenko, I. Yu. Chukicheva, A. V. Kutchin, Molecules, 2021, 26, 3579; DOI: https://doi.org/10.3390/molecules26123579.
S. A. Popova, O. G. Shevchenko, I. Yu. Chukicheva, A. V. Kutchin, Chem. Biodiversity, 2019, 16, e1800317.
S. A. Popova, O. G. Shevchenko, I. Yu. Chukicheva, Chem. Biol. Drug Des., 2021, 00, 1; DOI: https://doi.org/10.1111/cbdd.13955.
A. V. Samet, O. G. Shevchenko, V. V. Rusak, E. M. Chartov, A. Myshlyavtsev, D. Rusanov, M. N. Semenova, V. V. Semenov, J. Nat. Prod., 2019, 82, 1451.
I. A. Dvornikova, E. V. Buravlev, O. G. Shevchenko, I. Yu. Chukicheva, A. V. Kutchin, Russ. Chem. Bull., 2021, 70, 2185; DOI: https://doi.org/10.1007/s11172-021-3330-0.
I. A. Dvornikova, E. V. Buravlev, I. V. Fedorova, O. G. Shevchenko, I. Yu. Chukicheva, A. V. Kutchin, Russ. Chem. Bull., 2019, 68, 1000; DOI: https://doi.org/10.1007/s11172-019-2510-7.
I. Yu. Chukicheva, E. V. Buravlev, D. V. Belykh, I. S. Khudyaeva, I. V. Fedorova, O. G. Shevchenko, M. A. Maksimova, L. F. Zainullina, Yu. V. Vakhitova, A. V. Kutchin, Russ. Chem. Bull., 2018, 67, 548; DOI: https://doi.org/10.1007/s11172-018-2109-4.
S. A. Popova, E. V. Pavlova, I. Yu. Chukicheva, Vestn. Bashkirskogo universiteta [Bull. Bashkir Univ.], 2021, 26, 909 (in Russian).
L. Cesari, F. Mutelet, L. Canabady-Rochelle, Industrial Crops & Products, 2019, 129, 480; DOI: https://doi.org/10.1016/j.indcrop.2018.12.010.
R. Farhoosh, S. Johnny, M. Asnaashari, N. Molaahmadibahraseman, A. Sharif, Food Chem., 2016, 194, 128; DOI: https://doi.org/10.1016/j.foodchem.2015.08.003.
M. Güra, H. Muglu, M. S. Çavus, A. Güder, H. S. Sayiner, F. Kandemirli, J. Mol. Struct., 2017, 1134, 40; DOI: https://doi.org/10.1016/j.molstruc.2016.12.041.
I. Yu. Chukicheva, I. V. Fedorova, E. V. Buravlev, K. Yu. Suponitskii, A. V. Kuchin, Russ. J. Org. Chem., 2012, 82, 1425; DOI: https://doi.org/10.1134/S1070363212080154.
W. L. Mendelson, S. Hayden, Synth. Commun., 1996, 26, 603; DOI:.
K. Sevgi, B. Tepe, C. Sarikurkcu, Food Chem. Toxicol., 2015, 77, 12; DOI: https://doi.org/10.1016/j.fct.2014.12.006.
S. N. Lim, P. C. K. Cheung, V.E.C. Ooi, P. O. Ang, J. Agric. Food Chem., 2002, 50, 3862; DOI: https://doi.org/10.1021/jf020096b.
C. I. Acker, R. Brandão, A. R. Rosário, C. W. Nogueira, Environment. Toxicol. Pharmacol., 2009, 28, 280; DOI: https://doi.org/10.1016/j.etap.2009.05.002.
C.-R. Wu, W.-H. Lin, Y.-C. Hseu, J.-C. Lien, Y.-T. Lin, T.-P. Kuo, H. Ching, Food Chem., 2011, 127, 564; DOI: https://doi.org/10.1016/j.foodchem.2011.01.041.
J.-S. Kim, Food Nutrit. Sci., 2013, 4, 177; DOI: https://doi.org/10.4236/fns.2013.42025.
S. T. Stefanello, A. S. Prestes, T. Ogunmoyole, S. M. Salman, R. S. Schwab, C. R. Brender, L. Dornelles, J. B. T. Rocha, F. A. A. Soares, Toxicol. in Vitro, 2013, 27, 1433; DOI: https://doi.org/10.1016/j.tiv.2013.03.001.
R. Chawla, R. Arora, R. Kumar, A. Sharma, J. Prasad, S. Singh, R. Sagar, P. Chaundhary, S. Shukla, G. Kaur, R. K. Sharma, S. C. Puri, K. L. Dhar, G. Handa, V. K. Gupta, G. N. Qazi, Mol. Cell. Biochem., 2005, 273, 193; DOI: https://doi.org/10.1007/s11010-005-0821-5.
T. Asakawa, S. Matsushita, Lipids, 1980, 15, 137; DOI: https://doi.org/10.1007/BF02540959.
J. Takebayashi, J. Chen, A. A. Tai, in Advanced Protocols in Oxidative Stress II (Methods in Molecular Biology, Vol. 594), Ed. D. Armsrtong, Humana Press, New York, Dordrecht, Heidelberg, London, 2010, 594, p. 287.
J. J. M. Van den Berg, J. A. F. Op den Kamp, B. H. Lubin, B. Roelofsen, F. A. Kuypers, Free Radical Biol. Med., 1992, 12, 487; DOI: https://doi.org/10.1016/0891-5849(92)90102-M.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was financially supported by the Russian Science Foundation (Project No. 21-73-20091).
The synthesized compounds were analyzed using the equipment of the Center for Collective Usage “Chemistry” (Institute of Chemistry, Komi Scientific Center, Ural Branch of the Russian Academy of Sciences). The study of the in vitro activity of the compounds was carried out using the equipment of the Center for Collective Usage “Molecular Biology” (Institute of Biology, Komi Scientific Center, Ural Branch of the Russian Academy of Sciences).
Animals from the Scientific Collection of Experimental Animals at the Institute of Biology, Komi Science Center, Ural Branch of the Russian Academy of Sciences (http://www.ckp-rf.ru/usu/471933/) were used. The animals were handled in accordance with the “Regulations on the Vivarium of Experimental Animals” (Protocol No. 1, dated January 24, 2017) taking into account sanitary, hygienic, and bioethical aspects.
The authors declare no competing interests.
Based on the materials of the VI North Caucasus Organic Chemistry Symposium NCOCS-022 (April 18–22, 2022, Stavropol, Russia).
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 11, pp. 2383–2394, November, 2022.
Rights and permissions
About this article
Cite this article
Popova, S.A., Pavlova, E.V., Shevchenko, O.G. et al. Substituted chalcones with different positions of the isobornyl substituent: the synthesis and antioxidant activity. Russ Chem Bull 71, 2383–2394 (2022). https://doi.org/10.1007/s11172-022-3666-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-022-3666-0