Abstract
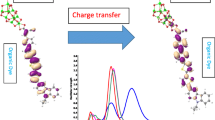
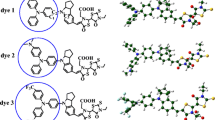
The geometry, electronic structure and properties, and composition of frontier molecular orbitals of the FeII complex with 4,4´,4´´,4´´´-substituted 2,2´:6´,2´´:6´´,2´´´-quaterpyridine were examined using quantum chemical simulation. This compound was proposed for use as a photoactive component in dye-sensitized solar cells. The regularities were established of the influence of the number of carboxyl groups in the molecular structure on the composition and energy of the frontier orbitals determining the electronic absorption spectra of the studied compounds. The presence of two COOH groups in the structure of the studied complex is the most optimal for use as a sensitizer.
Similar content being viewed by others
References
B. O´Regal, M. Grätzel, Nature, 1991, 353, 737.
A. Hagfeldt, G. Boschloo, L. Sun, L. Kloo, H. Pettersson, Chem. Rev., 2010, 110, 6595.
L. Zhang, J. M. Cole, ACS Appl. Mater. Interfaces, 2015, 7, 3427.
K. Ladomenou, T. N. Kitsopoulos, G. D. Sharma, A. G. Coutsolelos, RSC Adv., 2014, 4, 21379.
M. Pastore, F. De Angelis, Phys. Chem. Chem. Phys., 2012, 14, 920.
W. Du, H.-B. Li, Y. Geng, Y. Wu, M. Zhang, Z.-M. Su, J. Photochem. Photobiol., A, 2015, 301, 40.
S. G. Yan, J. T. Hupp, J. Phys. Chem., 1996, 100, 6867.
F. De Angelis, S. Fantacci, A. Selloni, M. K. Nazeeruddin, M. Gratzel, J. Phys. Chem. C, 2010, 114, 6054.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, J. A. Montgomery, Jr., T. Vreven, K. N. Kudin, J. C. Burant, J. M. Millam, S. S. Iyengar, J. Tomasi, V. Barone, B. Mennucci, M. Cossi, G. Scalmani, N. Rega, G. A. Petersson, H. Nakatsuji, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, M. Klene, X. Li, J. E. Knox, H. P. Hratchian, J. B. Cross, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, P. Y. Ayala, K. Morokuma, G. A. Voth, P. Salvador, J. J. Dannenberg, V. G. Zakrzewski, S. Dapprich, A. D. Daniels, M. C. Strain, O. Farkas, D. K. Malick, A. D. Rabuck, K. Raghavachari, J. B. Foresman, J. V. Ortiz, Q. Cui, A. G. Baboul, S. Clifford, J. Cioslowski, B. B. Stefanov, G. Liu, A. Liashenko, P. Piskorz, I. Komaromi, R. L. Martin, D. J. Fox, T. Keith, M. A. AlLaham, C. Y. Peng, A. Nanayakkara, M. Challacombe, P. M. W. Gill, B. Johnson, W. Chen, M. W. Wong, C. Gonzalez, J. A. Pople, Gaussian03, Revision E.1, Gaussian, 2003.
P. J. Stephens, F. J. Devlin, C. F. Chabalowski, M. J. Frisch, J. Phys. Chem., 1994, 98, 11623.
A. D. Becke, J. Chem. Phys., 1993, 98, 5648.
C. Lee, W. Yang, R. G. Parr, Phys. Rev. B, 1988, 37, 785.
S. Miertus, E. Scrocco, J. Tomasi, Chem. Phys., 1981, 55, 117.
R. Cammi, J. Tomasi, J. Comput. Chem., 1995, 16, 1449.
G. A. Zhurko, D. A. Zhurko, Chemcraft Version 1.6 (build 338); http://www.chemcraftprog.com.
Chemissian 1.770; http://www. chemissian.com.
M. K. Nazeeruddin, S. M. Zakeeruddin, R. Humphry-Baker, M. Jirousek, P. Liska, N. Vlachopoulos, V. Shklover, C.-H. Fischer, M. Grätzel, Inorg. Chem., 1999, 38, 6298.
M. K. Nazeeruddin, P. Péchy, T. Renouard, S. M. Zakeeruddin, R. Humphry-Baker, P. Comte, P. Liska, L. Cevey, E. Costa, V. Shklover, L. Spiccia, G. B. Deacon, C. A. Bignozzi, M. Grätzel, J. Am. Chem. Soc., 2001, 123, 1613.
C. Coluccini, N. Manfredi, M. M. Salamone, R. Ruffo, M. G. Lobello, F. D. Angelis, A. Abbotto, J. Org. Chem., 2012, 77, 7945.
Xiaoqing Lu, Shuxian Wei, Chi-Man Lawrence Wu, Ning Ding, Shaoren Li, Lianming Zhao, Wenyue Guo, Intern. J. Photoen., 2011, article 316952, 11 p., doi: 10.1155/2011/ 316952.
J. Kruger, U. Bach, M. Grätzel, Adv. Mater., 2000, 12, 447.
X. Liu, S. R. Scully, M. D. McGehee, J. S. Liu, C. K. Luscombe, J. M. J. Frechet, S. E. Shaheen, D. S. Ginley, J. Phys. Chem. B, 2006, 110, 3257.
G. Rothenberger, D. Fitzmaurice, M. Grätzel, J. Phys. Chem., 1992, 96, 5983.
I. Mora-Sero, J. Bisquert, J. Phys. Chem. Lett., 2010, 1, 3046.
I. Nazmul, C. G. Dulal, Int. J. Mol. Sci., 2012, 13, 2160.
R. Parthasarathi, V. Subramanian, D. R. Roy, P. K. Chattaraj, Bioorg. Med. Chem., 2004, 12, 5533.
Author information
Authors and Affiliations
Corresponding author
Additional information
Based on the materials of the XXVI International Chugaev Conference on Coordination Chemistry (October 6—10, 2014, Kazan).
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 8, pp. 1801—1807, August, 2015.
Rights and permissions
About this article
Cite this article
Tsaturyan, A.A., Shcherbakov, I.N., Kogan, V.A. et al. Influence of the number of anchor groups on the photophysical properties of coordination compounds as components of dye-sensitized solar cells. Russ Chem Bull 64, 1801–1807 (2015). https://doi.org/10.1007/s11172-015-1074-4
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-015-1074-4