Abstract
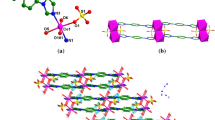
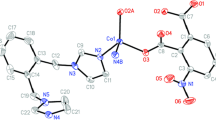
The reactions of MnII and CuII ions with deprotonated 2-hydroxy-3,6-di(tert-butyl)-1,4-benzoquinone (L) afford bis(chelates) ML2. In the solid phases of the compounds, the coordinated ligands L perform the bridging function leading to the formation of polymer structures: layered in MnL2 and framework in CuL2. In the coordination units ML2 the chelate fragments are in the trans and cis positions relative to one another for MnL2 and CuL2, respectively. Paramagnetism of the polymer structures MnL2 and CuL2, for which the effective magnetic moments in the high-temperature region are 5.85 and 1.91 μB, respectively, is determined by the metal ions, because the energy level of the triplet state of the coordinated L lies much higher, according to the calculated data, than that of the singlet state.
Similar content being viewed by others
References
R. Joshi, J. P. Kamat, T. Mukherjee, Chem.-Biol. Interact., 2007, 167, 125.
P. C. Lin, S. A. Li, S. O. Wang, Y. Yang, J. G. Shi, J. Nat. Prod., 2006, 69, 1629.
R. H. Thomson, Naturally Occurring Quinones, 2nd ed., Academic Press, London, 1971.
R. H. Thomson, Naturally Occurring Quinones III. Recent advances, 3rd ed., Chapman and Hall, London, 1987.
R. H. Thomson, Naturally Occurring Quinones IV. Recent advances, 4th ed., Blackie, London, 1997.
H. Shigemori, T. Madono, T. Sasaki, Y. Mikami, J. Kobayashi, Tetrahedron, 1994, 50, 8347.
J. Kobayashi, T. Madono, H. Shigemori, Tetrahedron, 1995, 51, 10867.
S. Aoki, D. Kong, K. Matsui, R. Rachmat, M. Kobayashi, Chem. Pharm. Bull., 2004, 52, 935.
Y. Takahashi, T. J. F. Kubota, J. Kobayashi, Tetrahedron, 2007, 63, 8770.
I. Kubo, M. Kim, I. Ganjian, T. Kamikawa, Y. Yamagiwa, Tetrahedron, 1987, 43, 2653.
S. Poigny, M. Guyot, M. Samadi, Tetrahedron, 1998, 54, 14791.
C. S. P. McErlean, C. J. Moody, J. Org. Chem., 2007, 72, 10298.
R. L. Danheiser, D. D. Cha, Tetrahedron Lett., 1990, 31, 1527.
P. Stahl, H. Waldmann, Angew. Chem., Int. Ed. Engl., 1999, 38, 3710.
A. R. MacKenzie, C. J. Moody, C. W. Rees, J. Chem. Soc., Chem. Commun., 1983, 1372.
K. S. Min, A. G. DiPasqual, J. A. Golen, A. L. Rheingold, A. M. Arif, J. S. Miller, J. Am. Chem. Soc., 2007, 129, 2360.
K. S. Min, A. G. DiPasqual, A. L. Rheingold, J. S. Miller, Inorg. Chem., 2007, 46, 1048.
K. S. Min, K. Swierczek, A. G. DiPasqual, A. L. Rheingold, W. M. Reiff, A. M. Arif, J. S. Miller, Chem. Commun., 2008, 317.
K. Yamada, H. Tanaka, S. Yagishita, K. Adachi, T. Uemura, S. Kitagawa, S. Kawata, Inorg. Chem., 2006, 45, 4322.
K. Yamada, S. Yagishita, H. Tanaka, K. Tohyama, K. Adachi, S. Kaizaki, H. Kumagai, K. Inoue, R. Kitaura, H.-C. Chang, S. Kitagawa, S. Kawata, Eur. J. Chem., 2004, 10, 2647.
S. Salunke-Gawalia, S. Y. Ranea, V. G. Puranikb, C. Guyard-Duhayonc, F. Varretd, Polyhedron, 2004, 23, 2541.
M. P. Mulay, P. L. Garge, S. B. Padhye, R. C. Haltiwanger, L. A. deLearie, C. Pierpont, Chem. Commun., 1987, 581.
M. A. Martinez, M. C. L. De Jimenez, E. E. Castellano, O. E. Piro, P. J. Aymonino, Coord. Chem. Rev., 2003, 56, 803.
P. Garge, R. Chikate, S. Padhye, J.-M. Savariault, P. de Loth, J.-P. Tuchagues, Inorg. Chem., 1990, 29, 3315.
N. H. Gokhale, S. B. Padhye, S. L. Croft, H. D. Kendrick, W. Davies, C. E. Anson, A. K. Powell, J. Inorg. Biochem., 2003, 95, 249.
C. L. Foster, X. Liu, C. A. Kilner, M. Thornton-Pett, M. A. Halcrow, J. Chem. Soc., Dalton Trans., 2000, 4563.
M. P. Bubnov, V. I. Nevodchikov, G. K. Fukin, V. K. Cherkasov, G. A. Abakumov, Inorg. Chem. Commun., 2007, 10, 989.
N. A. Malysheva, A. I. Prokof’ev, S. P. Solodovnikov, N. N. Bubnov, T. I. Prokof’eva, V. B. Vol’eva, M. I. Kabachnik, Izv. Akad. Nauk SSSR, Ser. Khim., 1990, 161 [Bull. Acad. Sci. USSR, Div. Chem. Sci. (Engl. Transl.), 1990, 145].
N. Yu. Kabarova, V. K. Cherkasov, L. N. Zakharov, G. A. Abakumov, Yu. T. Struchkov, L. G. Abakumova, Izv. Akad. Nauk SSSR, Ser. Khim., 1992, 2798 [Bull. Acad. Sci. USSR, Div. Chem. Sci. (Engl. Transl.), 1992, 2224].
A. L. Spek, J. Appl. Crystallogr., 2003, 36, 7.
Cambridge Structural Database, Version 5.30, November 2008 (Update February 2009).
F. Neese, ORCA — An ab Initio, Density Functional and Semiempirical Program Package, Version 2.6, Revision 35, University of Bonn, 2008.
C. Adamo, M. Cossi, V. Barone, J. Mol. Struct. Theochem., 1999, 493, 145.
C. A. Daul, I. Ciofini, A. Bencini, in Reviews of Modern Quantum Chemistry, part II, Ed. K. D. Sen, World Scientific, Singapore, 2002, 1247.
L. Noodleman, C. Y. Peng, D. A. Case, J. M. Mouesca, Coord. Chem. Rev., 1995, 144, 199.
L. Noodleman, J. G. Norman, J. Chem. Phys., 1979, 70, 4903.
A. J. H. Wachters, J. Chem. Phys., 1970, 52, 1033.
A. J. H. Wachters, IBM Tech. Rept., RJ584, 1969.
C. W. Bauschlicher, S. R. Langhoff, L. A. Barnes, J. Chem. Phys., 1989, 91, 2399.
E. R. Davidson, D. Feller, Chem. Rev., 1986, 86, 681.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Academician O. N. Chupakhin on the occasion of his 75th birthday.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 6, pp. 1110–1116, June, 2009.
Rights and permissions
About this article
Cite this article
Fursova, E.Y., Ovcharenko, V.I., Gorelik, E.V. et al. Coordination polymers based on CuII and MnII complexes with 2-hydroxy-3,6-di(tert-butyl)-1,4-benzoquinone. Russ Chem Bull 58, 1139–1145 (2009). https://doi.org/10.1007/s11172-009-0148-6
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-009-0148-6