Abstract
Aims
Cotton continuous cropping might cause the soil-borne disease Verticillium wilt in agricultural production. Crop rotation has been shown to be an effective method for controlling this disease. The objectives of this study were to evaluate the effects of cotton-maize rotation (CMR) and cotton-maize-broccoli residues rotation (CMBR) on incidence of cotton Verticillium wilt (CVW), soil microbial community structure and function.
Methods
qPCR was used to determine the populations of V. dahliae. Soil microbial community structure and function was analysed using metagenomics analysis.
Results
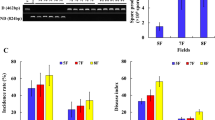
Results demonstrated that the disease incidence significantly decreased by 87.19% and 98.93%, and the populations of V. dahliae decreased by 67.07% and 84.36% under the CMR and CMBR treatments, respectively. There were significantly differences in bacterial communities under the CMR and CMBR treatments. Redundancy analysis revealed that the structures of microbial communities were mainly driven by available potassium (AK) and pH. Spearman’s correlation analysis showed that most of KEGG pathways in bacterial community were negatively with pH, positively with NH4+-N and AK. The abundances of NifH, AmoA, NirK, and NarG genes (involved in nitrogen cycling), PfkABC, FbaB, PGAM and KdpgA genes (carbon cycling), and Sir, Sat, and CysND (sulfur cycling) were enriched by the CMR and CMBR treatments.
Conclusions
CMR and CMBR could change the bacterial community structure and nutrients cycling in cotton rhizosphere, decrease the populations of V. dahliae in soil, therefore significantly reduce the incidence of CVW.







Similar content being viewed by others
Data availability
All raw sequences have been deposited at the National Center for Biotechnology Information (NCBI) under accession number PRJNA732914.
Code availability
Not applicable.
References
Alami MM, Xue JQ, Ma YT, Zhu DY, Abbas A, Gong ZD et al (2020) Structure, function, diversity, and composition of fungal communities in rhizospheric soil of Coptis chinensis franch under a successive cropping system. Plants 9(2):244. https://doi.org/10.3390/plants9020244
Amini J (2015) Induced resistance in potato plants against verticillium wilt invoked by chitosan and Acibenzolar-S-methyl. Aust J Crop Sci 9:570–576
Atallah ZK, Maruthachala K, Subbarao KV (2012) Sources of Verticillium dahliae affecting lettuce. Phytopathol 102:1071–1078. https://doi.org/10.1094/phyto-04-12-0067-r
Badri DV, Zolla G, Bakker MG, Manter DK, Vivanco JM (2013) Potential impact of soil microbiomes on the leaf metabolome and on herbivore feeding behavior. New Phytol 198(1):264–273. https://doi.org/10.1111/nph.12124
Banerjee S, Walder F, Buechi L, Meyer M, Held AY, Gattinger A et al (2019) Agricultural intensification reduces microbial network complexity and the abundance of keystone taxa in roots. ISME J 13:1722–1736. https://doi.org/10.1038/s41396-019-0383-2
Belofsky GN, Gloer KB, Gloer JB, Wicklow DT, Dowd PF (1998) New p-terphenyl and olyketide metabolites from the sclerotia of Penicillium raistrickii. J Nat Prod 61(9):1115–1119. https://doi.org/10.1021/np980188o
Berthrong ST, Jobbagy EG, Jackson RB (2009) A global meta-analysis of soil exchangeable cations, pH, carbon, and nitrogen with afforestation. Eco Appl 19(8):2228–2241. https://doi.org/10.1890/08-1730.1
Brankatschk G, Finkbeiner M (2017) Crop rotations and crop residues are relevant parameters for agricultural carbon footprints. Agron Sustain Dev 37(6):58. https://doi.org/10.1007/s13593-017-0464-4
Cal AD, Sagasta EM, Melgarejo P (1990) Biological control of peach twig blight (Monilinia laxa) with Penicillium frequentans. Plant Pathol 39(4):612–618. https://doi.org/10.1111/j.1365-3059.1990.tb02542.x
Campbell CA, Zentner RP (1993) Soil organic matter as influenced by crop rotations and fertilization. Soil Sci Soc Am J 57(4):1034–1034. https://doi.org/10.2136/sssaj1993.03615995005700040026x
Chang Y, Wang SS, Sekimoto S, Aerts A, Choi C, Clum A et al (2015) Phylogenomic analyses indicate that early fungi evolved digesting cell walls of algal ancestors of land plants. Genome Biol Evol 7(6):1590–1601. https://doi.org/10.1093/gbe/evv090
Chen JY, Xiao HL, Gui YJ, Zhang DD, Li L, Bao YM et al (2016) Characterization of the Verticillium dahliae exoproteome involves in pathogenicity from cotton-containing medium. Front Microbiol 7. https://doi.org/10.3389/fmicb.2016.01709
De Corato U, Patruno L, Avella N, Salimbeni R, Lacolla G, Cucci G et al (2020) Soil management under tomato-wheat rotation increases the suppressive response against Fusarium wilt and tomato shoot growth by changing the microbial composition and chemical parameters. Appl Soil Ecol 154:103601. https://doi.org/10.1016/j.apsoil.2020.103601
De Souza RSC, Armanhi JSL, Arruda P (2020) From microbiome to traits: designing synthetic microbial communities for improved crop resiliency. Front Plant Sci 11. https://doi.org/10.3389/fpls.2020.01179
Drigo B, Pijl AS, Duyts H, Kielak AM, Gamper HA, Houtekamer MJ et al (2010) Shifting carbon flow from roots into associated microbial communities in response to elevated atmospheric CO2. P Natl Acda Sci USA 107(24):10938–10942. https://doi.org/10.2307/20723996
El Ghaouth A, Wilson CL, Wisniewski M (2003) Control of postharvest decay of apple fruit with Candida saitoanaand induction of defense responses. Phytopathology 93(3):344–348. https://doi.org/10.1094/PHYTO.2003.93.3.344
Fan FL, Yin C, Tang YJ, Li ZJ, Song AL, Wakelin SA et al (2014) Probing potential microbial coupling of carbon and nitrogen cycling during decomposition of maize residue by 13C-DNA-SIP. Soil Biol Biochem 70:12–21. https://doi.org/10.1016/j.soilbio.2013.12.002
Feng YX, Hu YY, Wu JS, Chen JH, Yrjälä K, Yu WW (2019) Change in microbial communities, soil enzyme and metabolic activity in a Torreya grandis plantation in response to root rot disease. Forest Ecol Manag 432:932–941. https://doi.org/10.1016/j.foreco.2018.10.028
Fradin EF, Thomma BP (2006) Physiology and molecular aspects of Verticillium wilt diseases caused by V. dahliae and V. albo-atrum. Mol Plant Pathol 7:71–86. https://doi.org/10.1111/j.1364-3703.2006.00323.x
Gao ZY, Han MK, Hu YY, Xu JJ, Li ZQ, Liu CF et al (2019) Effects of continuous cropping of sweet potato on the fungal community structure in rhizospheric soil. Front Microbiol 10:2269. https://doi.org/10.3389/fmicb.2019.02269
Gao ZY, Hu YY, Han MK, Xu JJ, Wang X, Liu LF et al (2021) Effects of continuous cropping of sweet potatoes on the bacterial community structure in rhizospheric soil. BMC Microbiol 21:102. https://doi.org/10.1186/s12866-021-02120-6
Geisen S, Hu S, dela Cruz TEE, Veen GF(C) (2020) Protists as catalyzers of microbial litter breakdown and carbon cycling at different temperature regimes. ISME J 15:618–621. https://doi.org/10.1038/s41396-020-00792-y
Glockner FO, Kube M, Bauer M, Teeling H, Lombardot T, Ludwig W et al (2003) Complete genome sequence of the marine planctomycete Pirellula sp. strain 1. P Natl Acda Sci USA 100(14):8298–8303. https://doi.org/10.1073/pnas.1431443100
Gomes NCM, Fagbola O, Costa R, Rumjanek NG, Buchner A, Mendonca-Hagler L et al (2003) Dynamics of fungal communities in bulk and maize rhizosphere soil in the tropics. Appl Environ Microbiol 69(9):3758–3766. https://doi.org/10.1128/aem.69.9.5737.2003
Gong XY, Shi JB, Zhou XG, Yuan T, Gao DM, Wu FZ (2021) Crop rotation with cress increases cucumber yields by regulating the composition of the rhizosphere soil microbial community. Front Microbiol 12:556. https://doi.org/10.3389/fmicb.2021.631882
Gougoulias C, Clark JM, Shaw LJ (2014) The role of soil microbes in the global carbon cycle: tracking the below-ground microbial processing of plant-derived carbon for manipulating carbon dynamics in agricultural systems. J Sci Food Agric 94(12):2362–2371. https://doi.org/10.1002/jsfa.6577
Guo H, Liu XJ, Zhang Y, Shen JL, Han WX, Zhang WF et al (2010) Significant acidification in major Chinese croplands. Science 327(5968):1008–1010. https://doi.org/10.2307/40509964
Hong S, Jv HL, Lu M, Wang BB, Zhao Y, Ruan YZ (2020) Significant decline in banana Fusarium wilt disease is associated with soil microbiome reconstruction under chilli pepper-banana rotation. Eur J Soil Biol 97:103154. https://doi.org/10.1016/j.ejsobi.2020.103154
Hossain MM, Sultana F, Kubota M, Koyama H, Hyakumachi M (2007) The plant growth-promoting fungus Penicillium simplicissimum GP17-2 induces resistance in Arabidopsis thaliana by activation of multiple defense signals. Plant Cell Physiol 48(12):1724–1736. https://doi.org/10.1093/pcp/pcm144
Huang R, Li GQ, Zhang J, Yang L, Che HJ, Jiang DH et al (2011) Control of postharvest botrytis fruit rot of strawberry by volatile organic compounds of Candida intermedia. Phytopathology 101(7):859–869. https://doi.org/10.1094/PHYTO-09-10-0255
Huber DM (1990) Fertilizers and soil-borne diseases. Soil Use Manag 6(4):168–172. https://doi.org/10.1111/j.1475-2743.1990.tb00830.x
Huber DM, Thompson IA (2007) Nitrogen and plant disease. In: Datnoff LE, Elmer WH, Huber DM (eds) Mineral nutrition and plant disease. The American Phytopathological Society, St Paul, pp 31–44
Jian GL, Lu MG, Zhu HQ (2005) A preliminary study on the virulence of pathogenicity of Verticillium dahliae in cotton field. J Plant Protect 32(1):109–110
Jiang YJ, Liang YT, Li CM, Wang F, Sui YY, Suvannang N et al (2016) Crop rotations alter bacterial and fungal diversity in paddy soils across East Asia. Soil Biol Biochem 95:250–261. https://doi.org/10.1016/j.soilbio.2016.01.007
Klosterman SJ, Atallah ZK, Vallad GE, Subbarao KV (2009) Diversity, pathogenicity, and management of verticillium species. Annu Rev Phytopathol 47(1):39–62. https://doi.org/10.1146/annurev-phyto-080508-081748
Knight R, Vrbanac A, Taylo BC, Aksenov A, Callewaert C, Debelius J et al (2018) Best practices for analysing microbiomes. Nat Rev Microbiol 16(7):410–422. https://doi.org/10.1038/s41579-018-0029-9
Larkin R, Lynch R (2018) Use and effects of different Brassica and other rotation crops on soilborne diseases and yield of potato. Horticulturae 4(4):37. https://doi.org/10.3390/horticulturae4040037
Lei HY, Liu AK, Hou QW, Zhao QS, Guo J, Wang ZJ (2020) Diversity patterns of soil microbial communities in the Sophora flavescens rhizosphere in response to continuous monocropping. BMC Microbiol 20:272. https://doi.org/10.21203/rs.3.rs-28778/v2
Leoni C, Piancone E, Sasanelli N, Bruno GL, Manzari C, Pesole G et al (2020) Plant health and rhizosphere microbiome: effects of the bionematicide Aphanocladium album in tomato plants infested by Meloidogyne javanica. Microorganisms 8:1922. https://doi.org/10.3390/microorganisms8121922
Li XG, Zhang YN, Ding CF, Xu WH, Wang XX (2017) Temporal patterns of cotton Fusarium and Verticillium wilt in Jiangsu coastal areas of China. Sci Rep 7:12581. https://doi.org/10.1038/s41598-017-12985-1
Luo GW, Xue C, Jiang QH, Xiao Y, Zhang FG, Guo SW et al (2020) Soil carbon, nitrogen, and phosphorus cycling microbial populations and their resistance to global change depend on soil C:N:P Stoichiometry. mSystems 5(3):e00162–e00120. https://doi.org/10.1128/mSystems.00162-20
Maul J, Drinkwater L (2010) Short-term plant species impact on microbial community structure in soils with long-term agricultural history. Plant Soil 330:369–382. https://doi.org/10.1007/s11104-009-0211-y
Medina-Córdova N, López-Aguilar R, Ascencio F, Castellanos T, Campa-Córdova AI, Angulo C (2016) Biocontrol activity of the marine yeast Debaryomyces hansenii against phytopathogenic fungi and its ability to inhibit mycotoxins production in maize grain (Zea mays L.). Biol Control 97:70–79. https://doi.org/10.1016/j.biocontrol.2016.03.006
Nannipieri P, Giagnoni L, Renella G, Puglisi E, Ceccanti B, Masciandaro G et al (2012) Soil enzymology: classical and molecular approaches. Biol Fert Soils 48:743–762. https://doi.org/10.1007/s00374-012-0723-0
Navarrete AA, Diniz TR, Braga LPP, Silva GGZ, Franchini JC, Rossetto R et al (2015) Multi-analytical approach reveals potential microbial indicators in soil for sugarcane model systems. PLoS One 10:e0129765. https://doi.org/10.1371/journal.pone.0129765
Naylor D, Sadler N, Bhattacharjee A, Graham EB, Anderton CR, McClure R et al (2020) Soil microbiomes under climate change and implications for carbon cycling. Annu Rev Environ Resour 45(1):29–59. https://doi.org/10.1146/annurev-environ-012320-082720
Njoroge S, Kabir Z, Martin FN, Koike ST, Subbarao KV (2009) Comparison of crop rotation for Verticillium wilt management and effect on Pythium species in conventional and organic strawberry production. Plant Dis 93(5):519–527. https://doi.org/10.1094/pdis-93-5-0519
Osborne SL, Chim BK, Riedell WE, Schumacher TE (2020) Root length density of cereal and grain legume crops grown in diverse rotations. Crop Sci 60(5):2611–2620. https://doi.org/10.1002/csc2.20164
Pang ZQ, Dong F, Liu Q, Lin WX, Hu CH, Yuan ZN (2021) Soil metagenomics reveals effects of continuous sugarcane cropping on the structure and functional pathway of rhizospheric microbial community. Front Microbiol 12:627569. https://doi.org/10.3389/fmicb.2021.627569
Panichikkal J, Krishnankutty RE (2021) Root exudate components induced formation of plant beneficial metabolites by rhizospheric Pseudomonas spp. Rhizosphere 19:100366. https://doi.org/10.1016/j.rhisph.2021.100366
Pasche JS, Taylor RJ, David NL, Gudmestad NC (2014) Effect of soil temperature, injection depth, and metam sodium rate on the management of verticillium wilt of potato. Am J Potato Res 91:277–290. https://doi.org/10.1007/s12230-013-9348-6
Pegg GF, Brady BL (2002) Verticillium wilts. Cromwell Press, Trowbridge, UK
Reeves DW (1997) The role of soil organic matter in maintaining soil quality in continuous cropping systems. Soil Till Res 43(1–2):131–167. https://doi.org/10.1016/s0167-1987(97)00038-x
Ren CJ, Zhang W, Zhong ZK, Han XH, Yang GH, Feng YZ et al (2018) Differential responses of soil microbial biomass, diversity, and compositions to altitudinal gradients depend on plant and soil characteristics. Sci Total Environ 610–611:750–758. https://doi.org/10.1016/j.scitotenv.2017.08.110
Singh BK, Bardgett RD, Smith P, Reay DS (2010) Microorganisms and climate change: terrestrial feedbacks and mitigation options. Nat Rev Microbiol 8:779–790. https://doi.org/10.1262/jrd.17004
Singh RK, Singh P, Li HB, Song QQ, Guo DJ, Solanki MK et al (2020) Diversity of nitrogen-fixing rhizobacteria associated with sugarcane: a comprehensive study of plant-microbe interactions for growth enhancement in Saccharum spp. BMC Plant Biol 20:220. https://doi.org/10.1186/s12870-020-02400-9
Singh U, Choudhary AK, Sharma S (2021) Agricultural practices modulate the bacterial communities, and nitrogen cycling bacterial guild in rhizosphere: Field experiment with soybean. J Sci Food Agric 101(7):2687–2695. https://doi.org/10.1002/jsfa.10893
Subbarao KV, Hubbard JC, Koike ST (1999) Evaluation of broccoli residue incorporation into field soil for Verticillium wilt control in cauliflower. Plant Dis 83:124–129. https://doi.org/10.1094/pdis.1999.83.2.124
Su-ungkavatin P, Thongnueakhaeng W, Chaiprasert P (2019) Simultaneous removal of sulfur and nitrogen compounds with methane production from concentrated latex wastewater in two bioreactor zones of micro-oxygen hybrid reactor. J Chem Technol Biotechnol 94(10):3276–3291. https://doi.org/10.1002/jctb.6139
Tan Y, Cui YS, Li HY, Kuang AX, Li XR, Wei YL et al (2017) Rhizospheric soil and root endogenous fungal diversity and composition in response to continuous Panax notoginseng cropping practices. Microbiol Res 194:10–19. https://doi.org/10.1016/j.micres.2016.09.009
Tiemann LK, Grandy AS, Atkinson EE, Marin-Spiotta E, McDaniel MD (2015) Crop rotational diversity enhances belowground communities and functions in an agroecosystem. Ecol Lett 18:761–771. https://doi.org/10.1111/ele.12453
Uppal AK, El HA, Adams LR, Tenuta M, Daayt F (2008) Biological control of potato verticillium wilt under controlled and field conditions using selected bacterial antagonists and plant extracts. Biol Control 44:90–100. https://doi.org/10.1016/j.biocontrol.2007.10.020
Varo-Suárez A, Raya-Ortega MC, Agustí-Brisach C, García-Ortiz-Civantos C, Fernández-Hernández A, Mulero-Aparicio A et al (2017) Evaluation of organic amendments from agro-industry waste for the control of verticillium wilt of olive. Plant Pathol 67(4):860–870. https://doi.org/10.1111/ppa.12798
Wang C, Zheng MM, Song WF, Wen SL, Wang BR, Zhu CQ et al (2017) Impact of 25 years of inorganic fertilization on diazotrophic abundance and community structure in an acidic soil in southern China. Soil Biol Biochem 113:240–249. https://doi.org/10.1016/j.soilbio.2017.06.019
Wang YJ, Zhao WS, Chen D, Ji LJ, Li SZ, Kong LX et al (2020) Effect of growth substrate amended with broccoli residues on the spread of GFP-labeled Verticillium dahliae strain in cotton plant. Acta Phytopathologica Sinica 50(1):89–96. https://doi.org/10.13926/j.cnki.apps.000313
Wheeler TA, Woodward JE (2016) Field assessment of commercial cotton cultivars for Verticillium wilt resistance and yield. Crop Prot 88:1–6. https://doi.org/10.1016/j.cropro.2016.05.006
Wheeler TA, Bordovsky JP, Keeling JW (2019) The effectiveness of crop rotation on management of Verticillium wilt over time. Crop Prot 121:157–162. https://doi.org/https://doi.org/10.1016/j.cropro.2019.03.021
Winter M, Mol FD, Tiedemann AV (2014) Cropping systems with maize and oilseed rape for energy production may reduce the risk of stem base diseases in wheat. Field Crop Res 156:249–257. https://doi.org/10.1016/j.fcr.2013.10.009
Xi H, Zhang XK, Qu Z, Yang DY, Alariqi M, Yang ZG et al (2021) Effects of cotton–maize rotation on soil microbiome structure. Mol Plant Pathol 1-10. https://doi.org/10.1111/mpp.13053
Zhang WW, Wang C, Xue R, Wang LJ (2019a) Effects of salinity on the soil microbial community and soil fertility. J Integr Agric 18(6):1360–1368. https://doi.org/10.1016/s2095-3119(18)62077-5
Zhang JC, Yang XQ, Zhou H, Yang YB, Ding ZT (2019b) New natural products of rare actinomycetes from 2006 to 2018. Chinese J Org Chem 39(4):982–1012. https://doi.org/10.6023/cjoc201810006
Zhao WS, Guo QG, Li SZ, Wang YJ, Lu XY, Wang PP et al (2019) Control efficacy of broccoli residues on cotton Verticillium wilt and its effect on soil bacterial community at different growth stages. Sci Agric Sin 52(24):4505–4517. https://doi.org/10.3864/j.issn.0578-1752.2019.24.006
Zhao WS, Guo QG, Li SZ, Wang PP, Dong LH, Su ZH et al (2021) Effects of Bacillus subtilis NCD-2 and broccoli residues return on potato Verticillium wilt and soil fungal community structure. Biol Control 159:104628. https://doi.org/10.1016/j.biocontrol.2021.104628
Zhou TT, Zhao YL, Guo HS (2017) Secretory proteins are delivered to the septin-organized penetration interface during root infection by Verticillium dahliae. PLoS Pathog 13(3):e1006275. https://doi.org/10.1371/journal.ppat.1006275
Zhu HQ, Jian GL, Song XX (2004) Community structure of pathogenic type of Verticillium dahliae Kleb. in cotton field. Cotton Sci 16(3):147–151
Zhu SY, Wang YZ, Xu XM, Liu TM, Wu DQ, Zheng X et al (2018) Potential use of high-throughput sequencing of soil microbial communities for estimating the adverse effects of continuous cropping on ramie (Boehmeria nivea L. Gaud). PLoS One 13:e0197095. https://doi.org/10.1371/journal.pone.0197095
Acknowledgments
This work was supported by the China Agriculture Research System of MOF and MARA (CARS-15-19), the National Key Research and Development Program of China (2017YFD0200601), and the Special Fund for Agro-Scientific Research in the Public Interest (201503109).
Author information
Authors and Affiliations
Contributions
WZ, QG, SL, and PM planned, designed the research, and experiments. WZ, PW, LD, XZ, ZS, and XL performed the experiments. WZ and PM analyzed the data and wrote the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval
Not applicable.
Conflicts of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Responsible Editor: Sven Marhan.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1
(DOC 4.51 MB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, W., Li, S., Dong, L. et al. Effects of different crop rotations on the incidence of cotton Verticillium wilt and structure and function of the rhizospheric microbial community. Plant Soil 485, 457–474 (2023). https://doi.org/10.1007/s11104-022-05842-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-022-05842-2