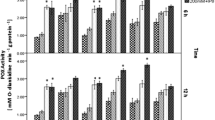
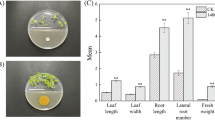
Abstract
Aims
Pseudomonas koreensis, a subgroup of the P. fluorescens complex, is a potential plant growth-promoting rhizobacterium. This study explored the mechanisms of plant growth promotion by P. koreensis and its potential to induce stress tolerance in tomato.
Methods
Tomato plants in pots were inoculated with P. koreensis GS and cultured for 60 days. RNA sequencing and gas chromatography-mass spectrometry were used to detect global transcriptomic and metabolomic changes in tomato leaves. Liquid chromatography-mass spectrometry was used for quantification of plant hormones.
Results
The inoculated plants showed more vigorous growth, with higher leaf chlorophyll content and shoot biomass compared with uninoculated controls. The activities of several defense enzymes (e.g., phenylalanine ammonia-lyase) were enhanced in the leaves of inoculated plants. A total of 737 differentially expressed genes were identified, which were related to plant hormone biosynthesis, MAPK signaling transduction, and polyphenol biosynthesis. The contents of specific metabolites related to plant growth and stress tolerance, including polyphenols (phenylpropanoids) and amino acids (tryptophanand and proline), were increased after inoculation. Plant hormones such as indole-3-acetic acid participated in plant growth promotion by P. koreensis.
Conclusions
P. koreensis promoted plant growth and showed potential to induce stress tolerance in tomato by enhancing auxin and polyphenol-related pathways.







Similar content being viewed by others
Abbreviations
- ABA:
-
Abscisic acid
- BAK1:
-
Brassinosteroid insensitive 1-associated receptor kinase 1
- DEGs:
-
Differentially expressed gene
- GA:
-
Gibberellic acid
- GID2:
-
GA-insensitive dwarf 2
- JA:
-
Jasmonic acid
- PAL:
-
Phenylalanine ammonia-lyase
- PIF3:
-
Phytochrome-interacting factor 3
- PPO:
-
Polyphenol oxidase
- RNA-Seq:
-
RNA sequencing
- TCH4:
-
xyloglucan:xyloglucosyl transferase TCH4
- WRKY33:
-
WRKY transcription factor 33
- Aux/IAA:
-
Auxin-responsive protein/IAA
- CAT:
-
Catalase
- FLS2:
-
Flagellin-sensing 2
- GH3:
-
Gretchen Hagen 3
- IAA:
-
Indole-3-acetic acid
- KEGG:
-
Kyoto Encyclopedia of Genes and Genomes
- PGPR:
-
Plant growth-promoting rhizobacterium
- POD:
-
Peroxidase
- PR1:
-
Pathogenesis-related protein1
- SA:
-
Salicylic acid
- VIP1:
-
VirE2-interacting protein 1
References
Acuna JJ, Jorquera MA, Martinez OA, Menezes-Blackburn D, Fernandez MT, Marschner P, Greiner R, Mora ML (2011) Indole acetic acid and phytase activity produced by rhizosphere bacilli as affected by pH and metals. J Plant Nutr Soil Sci 11:1–12. https://doi.org/10.4067/s0718-95162011000300001
Ahmed EA, Hassan EA, Tobgy KMKE, Ramadan EM (2014) Evaluation of rhizobacteria of some medicinal plants for plant growth promotion and biological control. Ann Agric Sci 59:273–280. https://doi.org/10.1016/j.aoas.2014.11.016
Ali-Benali MA, Alary R, Joudrier P, Gautier MF (2005) Comparative expression of five lea genes during wheat seed development and in response to abiotic stresses by real-time quantitative RT-PCR. Biochim Biophys Acta 1730:56–65. https://doi.org/10.1016/j.bbaexp.2005.05.011
Andreasson E et al (2005) The MAP kinase substrate MKS1 is a regulator of plant defense responses. EMBO J 24:2579–2589. https://doi.org/10.1038/sj.emboj.7600737
Atkinson NJ, Dew TP, Orfila C, Urwin PE (2011) Influence of combined biotic and abiotic stress on nutritional quality parameters in tomato (Solanum lycopersicum). J Agr Food Chem 59:9673–9682. https://doi.org/10.1021/jf202081t
Bajad SU, Lu WY, Kimball EH, Yuan J, Peterson C, Rabinowitz JD (2006) Separation and quantitation of water soluble cellular metabolites by hydrophilic interaction chromatography-tandem mass spectrometry. J Chromatogr A 1125:76–88. https://doi.org/10.1016/j.chroma.2006.05.019
Catala R, Medina J, Salinas J (2011) Integration of low temperature and light signaling during cold acclimation response in Arabidopsis. Proc Natl Acad Sci U S A 108:16475–16480. https://doi.org/10.1073/pnas.1107161108
Chen MJ, Liu QW, Tan JY, Xue-Mei LI (2015) Research advance on effects of heavy metal stress on plant organic acid metabolism. Guangdong Agric Sci 24:86–91. https://doi.org/10.16768/j.issn.1004-874x.2015.24.059
Cohen JD, Slovin JP, Hendrickson AM (2003) Two genetically discrete pathways convert tryptophan to auxin: more redundancy in auxin biosynthesis. Trends Plant Sci 8:0–199. https://doi.org/10.1016/S1360-1385(03)00058-X
Cuong DM, Kwon SJ, Jeon J, Park YJ, Park JS, Park SU (2018) Identification and characterization of phenylpropanoid biosynthetic genes and their accumulation in bitter melon (Momordica charantia). Molecules 23. https://doi.org/10.3390/molecules23020469
de Lucas M, Prat S (2014) PIFs get BRright: Phytochrome interacting factors as integrators of light and hormonal signals. New Phytol 202:1126–1141. https://doi.org/10.1111/nph.12725
de Werra P, Pechy-Tarr M, Keel C, Maurhofer M (2009) Role of gluconic acid production in the regulation of biocontrol traits of Pseudomonas fluorescens CHA0. Appl Environ Microbiol 75:4162–4174. https://doi.org/10.1128/aem.00295-09
Fahimi A, Ashouri A, Ahmadzadeh M, Naveh VH, Asgharzadeh A, Maleki F, Felton GW (2014) Effect of PGPR on population growth parameters of cotton aphid. Arch Phytopathol Plant Prot 47:1274–1285. https://doi.org/10.1080/03235408.2013.840099
Figueiredo MVB, Martinez CR, Burity HA, Chanway CP (2008) Plant growth-promoting rhizobacteria for improving nodulation and nitrogen fixation in the common bean (Phaseolus vulgaris L.). World J Microb Biot 24:1187–1193. https://doi.org/10.1007/s11274-007-9591-4
Fu XP, Dou CM, Hu SP, Chen XC, Shi JY, Chen YX (2010) A review of progress in roles of organic acids on heavy metal resistance and detoxification in plants. Chin J Plant Ecol 34:1354–1358. https://doi.org/10.3773/j.issn.1005-264x.2010.11.013
Gao JF (2006) Experimental techniques of plant physiology. Higher Education, Beijing, pp 214–215
Ghosh D, Sen S, Mohapatra S (2017) Modulation of proline metabolic gene expression in Arabidopsis thaliana under water-stressed conditions by a drought-mitigating Pseudomonas putida strain. Ann Microbiol 67:655–668. https://doi.org/10.1007/s13213-017-1294-y
Guo Q, Shi MD, Chen L, Zhou JH, Zhang LX, Li YL, Xue QH, Lai HX (2020) The biocontrol agent Streptomyces pactum increases Pseudomonas koreensis populations in the rhizosphere by enhancing chemotaxis and biofilm formation. Soil Biol Biochem 144:107755. https://doi.org/10.1016/j.soilbio.2020.107755
Habibi S, Djedidi S, Prongjunthuek K, Mortuza MF, Ohkama-Ohtsu N, Sekimoto H, Yokoyoma T (2014) Physiological and genetic characterization of rice nitrogen fixer PGPR isolated from rhizosphere soils of different crops. Plant Soil 379:51–66. https://doi.org/10.1007/s11104-014-2035-7
Hayat R, Ali S, Amara U, Khalid R, Ahmed I (2010) Soil beneficial bacteria and their role in plant growth promotion: a review. Ann Microbiol 60:579–598. https://doi.org/10.1007/s13213-010-0117-1
Hirano K, Aya K, Hobo T, Sakakibara H, Kojima M, Shim RA, Hasegawa Y, Ueguchi-Tanaka M, Matsuoka M (2008) Comprehensive transcriptome analysis of phytohormone biosynthesis and signaling genes in microspore/pollen and tapetum of rice. Plant Cell Physiol 49:1429–1450. https://doi.org/10.1093/pcp/pcn123
Hultberg M, Alsberg T, Khalil S, Alsanius B (2010) Suppression of disease in tomato infected by Pythium ultimum with a biosurfactant produced by Pseudomonas koreensis. Biocontrol 55:435–444. https://doi.org/10.1007/s10526-009-9261-6
Incerti A, Navari-Izzo F, Pardossi A, Mensuali A, Izzo R (2007) Effect of sea water on biochemical properties of fruit of tomato (Lycopersicon esculentum Mill.) genotypes differing for ethylene production. J Sci Food Agric 87:2528–2537. https://doi.org/10.1002/jsfa.3020
Kohno M, Takato H, Horiuchi H, Fujita K, Suzuki S (2012) Auxin-nonresponsive grape Aux/IAA19 is a positive regulator of plant growth. Mol Biol Rep 39:911–917. https://doi.org/10.1007/s11033-011-0816-0
Kumar S et al (2016) Paenibacillus lentimorbus inoculation enhances tobacco growth and extenuates the virulence of cucumber mosaic virus. PLos One 11. https://doi.org/10.1371/journal.pone.0149980
Li B (1998) Changes in activities and eletrophretic patterns of peroxidase and polyphenoloxidase in cucumbers during infection with Cladosporium cucumerinum. Sci Agric Sin 31:86–88
Li J (2011) Chinese tomato breeding. China Ariculture, Beijing, p 275
Li J, Wen JQ, Lease KA, Doke JT, Tax FE, Walker JC (2002) BAK1, an Arabidopsis LRR receptor-like protein kinase, interacts with BRI1 and modulates brassinosteroid signaling. Cell 110:213–222. https://doi.org/10.1016/s0092-8674(02)00812-7
Li YL, Guo Q, Li YZ, Sun YF, Xue QH, Lai HX (2019) Streptomyces pactum Act12 controls tomato yellow leaf curl virus disease and alters rhizosphere microbial communities. Biol Fertil Soils 55:149–169. https://doi.org/10.1007/s00374-019-01339-w
Lim JH, Kim SD (2009) Synergistic plant growth promotion by the indigenous auxins-producing PGPR Bacillus subtilis AH18 and Bacillus licheniforims K11. J Korean Soc Appl Bi 52:531–538. https://doi.org/10.3839/jksabc.2009.090
Lopes LD et al (2018) Genome variations between rhizosphere and bulk soil ecotypes of a Pseudomonas koreensis population. Environ Microbiol 20:4401–4414. https://doi.org/10.1111/1462-2920.14363
Manuel SP, Mayra AM, Sixta LP (2017) Biodegradable polymer hydrogels based in sorbitol and citric acid for controlled release of bioactive substances from plants (polyphenols). Curr Chem Biol 11:36–43. https://doi.org/10.2174/2212796810666161028114432
Mao GH, Meng XZ, Liu YD, Zheng ZY, Chen ZX, Zhang SQ (2011) Phosphorylation of a WRKY transcription factor by two pathogen-responsive MAPKs drives phytoalexin biosynthesis in Arabidopsis. Plant Cell 23:1639–1653. https://doi.org/10.1105/tpc.111.084996
Martins SJ, de Medeiros FHV, de Souza RM, de Resende MLV, Ribeiro PM (2013) Biological control of bacterial wilt of common bean by plant growth-promoting rhizobacteria. Biol Control 66:65–71. https://doi.org/10.1016/j.biocontrol.2013.03.009
Meng XZ, Zhang SQ (2013) MAPK cascades in plant disease resistance signaling. Annu Rev Phytopathol 51:245–266. https://doi.org/10.1146/annurev-phyto-082712-102314
Moura JC, Bonine CA, de Oliveira Fernandes Viana J, Dornelas MC, Mazzafera P (2010) Abiotic and biotic stresses and changes in the lignin content and composition in plants. J Integr Plant Biol 52:360–376. https://doi.org/10.1111/j.1744-7909.2010.00892.x
Nakabayashi R et al (2014) Enhancement of oxidative and drought tolerance in Arabidopsis by overaccumulation of antioxidant flavonoids. Plant J 77:367–379. https://doi.org/10.1111/tpj.12388
Pieterse CMJ, Zamioudis C, Berendsen RL, Weller DM, Van Wees SCM, Bakker PAHM (2014) Induced systemic resistance by beneficial microbes. Annu Rev Phytopathol 52:347–375. https://doi.org/10.1146/annurev-phyto-082712-102340
Pitzschke A, Djamei A, Teige M, Hirt H (2009) VIP1 response elements mediate mitogen-activated protein kinase 3-induced stress gene expression. Proc Natl Acad Sci U S A 106:18414–18419. https://doi.org/10.1073/pnas.0905599106
Porcel R, Zamarreno AM, Garcia-Mina JM, Aroca R (2014) Involvement of plant endogenous ABA in Bacillus megaterium PGPR activity in tomato plants. Bmc Plant Biol 14:36. https://doi.org/10.1186/1471-2229-14-36
Rafikova GF, Korshunova TY, Minnebaev LF, Chetverikov SP, Loginov ON (2016) A new bacterial strain, Pseudomonas koreensis IB-4, as a promising agent for plant pathogen biological control. Microbiology 85:333–341. https://doi.org/10.1134/s0026261716030115
Rangel LI, Henkels MD, Shaffer BT, Walker FL, Davis EW, Stockwell VO, Bruck D, Taylor BJ, Loper JE (2016) Characterization of toxin complex gene clusters and insect toxicity of bacteria representing four subgroups of Pseudomonas fluorescens. PLoS One 11:e0161120. https://doi.org/10.1371/journal.pone.0161120
Ryu CM, Hu CH, Locy RD, Kloepper JW (2005) Study of mechanisms for plant growth promotion elicited by rhizobacteria in Arabidopsis thaliana. Plant Soil 268:285–292. https://doi.org/10.1007/s11104-004-0301-9
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative C-T method. Nat Protoc 3:1101–1108. https://doi.org/10.1038/nprot.2008.73
Schulze B, Mentzel T, Jehle AK, Mueller K, Beeler S, Boller T, Felix G, Chinchilla D (2010) Rapid heteromerization and phosphorylation of ligand-activated plant transmembrane receptors and their associated kinase BAK1. J Biol Chem 285:9444–9451. https://doi.org/10.1074/jbc.M109.096842
Seo M, Nambara E, Choi G, Yamaguchi S (2009) Interaction of light and hormone signals in germinating seeds. Plant Mol Biol 69:463–472. https://doi.org/10.1007/s11103-008-9429-y
Shi MZ, Xie DY (2014) Biosynthesis and metabolic engineering of anthocyanins in Arabidopsis thaliana. Recent Pat Biotechnol 8:47–60
Silva HSA, Romeiro RD, Macagnan D, Halfeld-Vieira BD, Pereira MCB, Mounteer A (2004) Rhizobacterial induction of systemic resistance in tomato plants: non-specific protection and increase in enzyme activities. Biol Control 29:288–295. https://doi.org/10.1016/s1049-9644(03)00163-4
Staff SS (2010) Keys to Soil Taxonomy, 11th edn. USDA-Natural Resources Conservation Service, Washington, DC
Sun TP (2010) Gibberellin-GID1-DELLA: a pivotal regulatory module for plant growth and development. Plant Physiol 154:567–570. https://doi.org/10.1104/pp.110.161554
Thimm O et al (2004) MAPMAN: a user-driven tool to display genomics data sets onto diagrams of metabolic pathways and other biological processes. Plant J 37:914–939. https://doi.org/10.1111/j.1365-313X.2004.02016.x
Trapnell C, Williams BA, Pertea G, Mortazavi A, Kwan G, van Baren MJ, Salzberg SL, Wold BJ, Pachter L (2010) Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat Biotechnol 28:511–174. https://doi.org/10.1038/nbt.1621
Trinh CS, Jeong CY, Lee WJ, Truong HA, Chung N, Han J, Hong SW, Lee H (2018) Paenibacillus pabuli strain P7S promotes plant growth and induces anthocyanin accumulation in Arabidopsis thaliana. Plant Physiol Biochem 129:264–272. https://doi.org/10.1016/j.plaphy.2018.06.001
Ueguchi-Tanaka M et al (2007) Molecular interactions of a soluble gibberellin receptor, GID1, with a rice DELLA protein, SLR1, and gibberellin. Plant Cell 19:2140–2155. https://doi.org/10.1105/tpc.106.043729
Upadhyay SK, Singh JS, Saxena AK, Singh DP (2012) Impact of PGPR inoculation on growth and antioxidant status of wheat under saline conditions. Plant Biol (Stuttg) 14:605–611. https://doi.org/10.1111/j.1438-8677.2011.00533.x
Van den Ende W, El-Esawe SK (2014) Sucrose signaling pathways leading to fructan and anthocyanin accumulation: A dual function in abiotic and biotic stress responses? Environ Exp Bot 108:4–13. https://doi.org/10.1016/j.envexpbot.2013.09.017
Wang FB, Kong WL, Wong G, Fu LF, Peng RH, Li ZI, Yao QH (2016) AtMYB12 regulates flavonoids accumulation and abiotic stress tolerance in transgenic Arabidopsis thaliana. Mol Genet Genomics 291:1545–1559. https://doi.org/10.1007/s00438-016-1203-2
Wang YQ, Zhang YX, Gao ZP, Yang WC (2018) Breeding for resistance to tomato bacterial diseases in China: challenges and prospects. Hortic Plant J 4:193–207. https://doi.org/10.1016/j.hpj.2018.08.004
Wasilewska LD, Bralczyk J, Szczegielniak J (1987) The role of gibberellin in regulation of dwarf plants development. Plant Sci 53:11–19. https://doi.org/10.1016/0168-9452(87)90172-5
Xie YL, Wu LM, Zhu BC, Wu HJ, Gu Q, Rajer FU, Gao XW (2017) Digital gene expression profiling of the pathogen-resistance mechanism of Oryza sativa 9311 in response to Bacillus amyloliquefaciens FZB42 induction. Biol Control 110:89–97. https://doi.org/10.1016/j.biocontrol.2017.04.009
Xu W, Purugganan MM, Polisensky DH, Antosiewicz DM, Fry SC, Braam J (1995) Arabidopsis TCH4, regulated by hormones and the environment, encodes a xyloglucan endotransglycosylase. Plant Cell 7:1555–1567. https://doi.org/10.1105/tpc.7.10.1555
Yang X et al (2004) The IAA1 protein is encoded by AXR5 and is a substrate of SCFTIR1. Plant J 40:772–782. https://doi.org/10.1111/j.1365-313X.2004.02254.x
Zenoni S, Ferrarini A, Giacomelli E, Xumerle L, Fasoli M, Malerba G, Bellin D, Pezzotti M, Delledonne M (2010) Characterization of transcriptional complexity during berry development in vitis vinifera using RNA-SEq. Plant Physiol 152:1787–1795. https://doi.org/10.1104/pp.109.149716
Zhao YD (2010) Auxin biosynthesis and its role in plant development. Annu Rev Plant Biol 61:49–64. https://doi.org/10.1146/annurev-arplant-042809-112308
Zhao XM, Liu Y, Liu X, Jiang J (2018) Comparative transcriptome profiling of two tomato genotypes in response to potassium-deficiency stress. Int J Mol Sci 19. https://doi.org/10.3390/ijms19082402
Acknowledgements
We thank Dr. Chaofeng Lin (Transcend Envirotech Consulting. Co., Ltd., Qingdao, China) for improving the English. This work was supported by the Youth Fund of National Natural Science Foundation of China (31600407), the 2019 Frontier and Interdisciplinary Research Project of Basic Business Expenses in China (2452019177), and the Key R&D Program of Shaanxi Province, China (2019ZDLNY01-07-01).
Author information
Authors and Affiliations
Contributions
H.L. and Q.G. conceived the study and proposed the experimental approaches; Y.S., M.S., H.L., Y.L., and Q.G. conducted the experiments; Y.S., Q.G., X.H., and Y.J. contributed to data processing and analysis; and Q.G. and Y.S. wrote the manuscript. All authors read, commented on, and approved the final version of the manuscript.
Corresponding author
Ethics declarations
All authors are aware with the content of the manuscript and have agreed with the submission to Plant and Soil. The manuscript is not currently being considered for publication in another journal and the manuscript has not been published in whole or in part elsewhere.
Conflict of interest
The authors declare that they have no conflict of interest.
Human and animal rights
This article does not contain any studies with human or animal subjects.
Additional information
Responsible Editor: Luz E. de-Bashan.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 350 KB)
Rights and permissions
About this article
Cite this article
Guo, Q., Sun, Y., Shi, M. et al. Pseudomonas koreensis promotes tomato growth and shows potential to induce stress tolerance via auxin and polyphenol‐related pathways. Plant Soil 462, 141–158 (2021). https://doi.org/10.1007/s11104-021-04837-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-021-04837-9