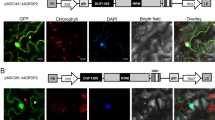
Abstract
AtMSI4/FVE/ACG1, one of five Arabidopsis thaliana genes encoding MSI1-like proteins, helps determine plant growth and development (including control of flowering), as well as responses to certain biotic and abiotic stresses. We reasoned that the product of this gene, AtMSI4, acts through protein partners, which we have co-immunopurified with AtMSI4 from A. thaliana suspension culture cells and identified by liquid chromatography–mass spectrometry (LC–MS). Many of the proteins associated with AtMSI4 have distinct RNA recognition motif (RRM) domains, which we determined to be responsible for association with AtMSI4; and most of the associated RRM domain proteins also contain PWWP domains that are specific to plants. We propose these novel ATMSI4-associated proteins help form nucleoprotein complexes that determine pleiotropic functional properties of AtMSI4/FVE/ACG1 involving plant development and responses to stress.









Similar content being viewed by others
Abbreviations
- LC–MS:
-
Liquid chromatography–mass spectrometry
- GST:
-
Glutathione-S-transferase
- RRM:
-
RNA recognition motif
- SET:
-
Suppressor of variegation, enhancer of zeste and trithorax domain
- HDA:
-
Histone deacetylase
References
Ach RA, Taranto P, Gruissem W (1997) A conserved family of WD-40 proteins binds to the retinoblastoma protein in both plants and animals. Plant Cell 9:1595–1606. doi:10.1105/tpc.9.9.1595
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410. doi:10.1016/S0022-2836(05)80360-2
Alvarez-Venegas R, Avramova Z (2012) Evolution of the PWWP-domain encoding genes in the plant and animal lineages. BMC Evol Biol 12:101. doi:10.1186/1471-2148-12-101
Ausin I, Alonso-Blanco C, Jarillo JA, Ruiz-Garcia L, Martinez-Zapater JM (2004) Regulation of flowering time by FVE, a retinoblastoma-associated protein. Nat Genet 36:162–166. doi:10.1038/ng1295
Baek IS, Park HY, You MK, Lee JH, Kim JK (2008) Functional conservation and divergence of FVE genes that control flowering time and cold response in rice and Arabidopsis. Mol Cells 26:368–372
Barak O, Lazzaro MA, Lane WS, Speicher DW, Picketts DJ, Shiekhattar R (2003) Isolation of human NURF: a regulator of Engrailed gene expression. EMBO J 22:6089–6100. doi:10.1093/emboj/cdg582
Bäurle I, Dean C (2008) Differential interactions of the autonomous pathway RRM proteins and chromatin regulators in the silencing of Arabidopsis targets. PLoS One 3:e2733. doi:10.1371/journal.pone.0002733
Campi M, D’Andrea L, Emiliani J, Casati P (2012) Participation of chromatin-remodeling proteins in the repair of ultraviolet-B-damaged DNA. Plant Physiol 158:981–995. doi:10.1104/pp.111.191452
Casati P et al (2008) Histone acetylation and chromatin remodeling are required for UV-B—dependent transcriptional activation of regulated genes in maize. Plant Cell 20:827–842. doi:10.1105/tpc.107.056457
Clery A, Blatter M, Allain FH (2008) RNA recognition motifs: boring? Not quite. Curr Opin Struct Biol 18:290–298. doi:10.1016/j.sbi.2008.04.002
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J Cell Molecul Biol 16:735–743
Cole C, Barber JD, Barton GJ (2008) The Jpred 3 secondary structure prediction server. Nucleic Acids Res 36:W197–W201. doi:10.1093/nar/gkn238
Corpet F (1988) Multiple sequence alignment with hierarchical clustering. Nucleic Acids Res 16:10881–10890
Eidahl JO et al (2013) Structural basis for high-affinity binding of LEDGF PWWP to mononucleosomes. Nucleic Acids Res. doi:10.1093/nar/gkt074
Garcia V, Salanoubat M, Choisne N, Tissier A (2000) An ATM homologue from Arabidopsis thaliana: complete genomic organisation and expression analysis. Nucleic Acids Res 28:1692–1699
Ge YZ, Pu MT, Gowher H, Wu HP, Ding JP, Jeltsch A, Xu GL (2004) Chromatin targeting of de novo DNA methyltransferases by the PWWP domain. J Biol Chem 279:25447–25454. doi:10.1074/jbc.M312296200
Gu J, Stephenson CG, Iadarola MJ (1994) Recombinant proteins attached to a nickel-NTA column: use in affinity purification of antibodies. BioTechniques 17(257):260–262
Gu X, Jiang D, Yang W, Jacob Y, Michaels SD, He Y (2011) Arabidopsis homologs of retinoblastoma-associated protein 46/48 associate with a histone deacetylase to act redundantly in chromatin silencing. PLoS Genet 7:e1002366. doi:10.1371/journal.pgen.1002366
Hajdukiewicz P, Svab Z, Maliga P (1994) The small, versatile pPZP family of Agrobacterium binary vectors for plant transformation. Plant Mol Biol 25:989–994
He Y, Michaels SD, Amasino RM (2003) Regulation of flowering time by histone acetylation in Arabidopsis. Science 302:1751–1754. doi:10.1126/science.1091109
Hennig L, Taranto P, Walser M, Schonrock N, Gruissem W (2003) Arabidopsis MSI1 is required for epigenetic maintenance of reproductive development. Development 130:2555–2565
Hennig L, Bouveret R, Gruissem W (2005) MSI1-like proteins: an escort service for chromatin assembly and remodeling complexes. Trends Cell Biol 15:295–302. doi:10.1016/j.tcb.2005.04.004
Hohenstatt ML (2012) Functional analysis of SCI1—A PWWP domain protein involved in Polycomb group mediated gene regulation in Arabidopsis. Dissertation, University of Duesseldorf
Jeon J, Kim J (2011) FVE, an Arabidopsis homologue of the retinoblastoma-associated protein that regulates flowering time and cold response, binds to chromatin as a large multiprotein complex. Mol Cells 32:227–234. doi:10.1007/s10059-011-1022-6
Jullien PE, Mosquna A, Ingouff M, Sakata T, Ohad N, Berger F (2008) Retinoblastoma and its binding partner MSI1 control imprinting in Arabidopsis. PLoS Biol 6:e194. doi:10.1371/journal.pbio.0060194
Jung JH, Park JH, Lee S, To TK, Kim JM, Seki M, Park CM (2013) The cold signaling attenuator HIGH EXPRESSION OF OSMOTICALLY RESPONSIVE GENE1 activates FLOWERING LOCUS C transcription via chromatin remodeling under short-term cold stress in Arabidopsis. Plant Cell 25:4378–4390. doi:10.1105/tpc.113.118364
Jung J-H, Lee H-J, Park M-J, Park C-M (2014) Beyond ubiquitination: proteolytic and nonproteolytic roles of HOS1. Trends Plant Sci. doi:10.1016/j.tplants.2014.03.012
Kaufman PD, Kobayashi R, Stillman B (1997) Ultraviolet radiation sensitivity and reduction of telomeric silencing in Saccharomyces cerevisiae cells lacking chromatin assembly factor-I. Genes Dev 11:345–357
Kaya H, Shibahara KI, Taoka KI, Iwabuchi M, Stillman B, Araki T (2001) FASCIATA genes for chromatin assembly factor-1 in Arabidopsis maintain the cellular organization of apical meristems. Cell 104:131–142
Kenan DJ, Query CC, Keene JD (1991) RNA recognition: towards identifying determinants of specificity. Trends Biochem Sci 16:214–220
Kenzior AL, Folk WR (1998) AtMSI4 and RbAp48 WD-40 repeat proteins bind metal ions. FEBS Lett 440:425–429
Kim HJ et al (2004) A genetic link between cold responses and flowering time through FVE in Arabidopsis thaliana. Nat Genet 36:167–171. doi:10.1038/ng1298
Kohler C, Hennig L, Bouveret R, Gheyselinck J, Grossniklaus U, Gruissem W (2003) Arabidopsis MSI1 is a component of the MEA/FIE polycomb group complex and required for seed development. EMBO J 22:4804–4814. doi:10.1093/emboj/cdg444
Korenjak M et al (2004) Native E2F/RBF complexes contain Myb-interacting proteins and repress transcription of developmentally controlled E2F target genes. Cell 119:181–193. doi:10.1016/j.cell.2004.09.034
Kortschak RD, Tucker PW, Saint R (2000) ARID proteins come in from the desert. Trends Biochem Sci 25:294–299
Kuzmichev A, Nishioka K, Erdjument-Bromage H, Tempst P, Reinberg D (2002) Histone methyltransferase activity associated with a human multiprotein complex containing the Enhancer of Zeste protein. Genes Dev 16:2893–2905. doi:10.1101/gad.1035902
Lewis PW, Beall EL, Fleischer TC, Georlette D, Link AJ, Botchan MR (2004) Identification of a Drosophila Myb-E2F2/RBF transcriptional repressor complex. Genes Dev 18:2929–2940. doi:10.1101/gad.1255204
Li D, Roberts R (2001) Human genome and diseases: WD-repeat proteins: structure characteristics, biological function, and their involvement in human diseases CMLS. Cell Mol Life Sci 58:2085–2097. doi:10.1007/pl00000838
Liu X et al (2012) HDA6 directly interacts with DNA methyltransferase MET1 and maintains transposable element silencing in Arabidopsis. Plant Physiol 158:119–129. doi:10.1104/pp.111.184275
Maris C, Dominguez C, Allain FHT (2005) The RNA recognition motif, a plastic RNA-binding platform to regulate post-transcriptional gene expression. FEBS J 272:2118–2131. doi:10.1111/j.1742-4658.2005.04653.x
Martinez-Balbas MA, Tsukiyama T, Gdula D, Wu C (1998) Drosophila NURF-55, a WD repeat protein involved in histone metabolism. Proc Natl Acad Sci 95:132–137
Maurer-Stroh S, Dickens NJ, Hughes-Davies L, Kouzarides T, Eisenhaber F, Ponting CP (2003) The Tudor domain ‘Royal Family’: tudor, plant agenet, chromo, PWWP and MBT domains. Trends Biochem Sci 28:69–74. doi:10.1016/S0968-0004(03)00004-5
Morel P, Tréhin C, Breuil-Broyer S, Negrutiu I (2009) Altering FVE/MSI4 results in a substantial increase of biomass in Arabidopsis–the functional analysis of an ontogenesis accelerator. Mol Breed 23:239–257
Nameki N et al (2005) Solution structure of the PWWP domain of the hepatoma-derived growth factor family. Protein Sci 14:756–764. doi:10.1110/ps.04975305
Pazhouhandeh M, Molinier J, Berr A, Genschik P (2011) MSI4/FVE interacts with CUL4-DDB1 and a PRC2-like complex to control epigenetic regulation of flowering time in Arabidopsis. Proc Natl Acad Sci 108:3430–3435. doi:10.1073/pnas.1018242108
Perkins DN, Pappin DJ, Creasy DM, Cottrell JS (1999) Probability-based protein identification by searching sequence databases using mass spectrometry data. Electrophoresis 20:3551–3567. doi:10.1002/(SICI)1522-2683(19991201)20:18<3551:AID-ELPS3551>3.0.CO;2-2
Qiu C, Sawada K, Zhang X, Cheng X (2002) The PWWP domain of mammalian DNA methyltransferase Dnmt3b defines a new family of DNA-binding folds. Nat Struct Biol 9:217–224. doi:10.1038/nsb759
Qiu Y et al (2012) Solution structure of the Pdp1 PWWP domain reveals its unique binding sites for methylated H4K20 and DNA. Biochem J 442:527–538. doi:10.1042/bj20111885
Rossi V et al (2001) The maize WD-repeat gene ZmRbAp1 encodes a member of the MSI/RbAp sub-family and is differentially expressed during endosperm development. Molecul Genet Genomics 265:576–584
Ruggieri R, Tanaka K, Nakafuku M, Kaziro Y, Toh-e A, Matsumoto K (1989) MSI1, a negative regulator of the RAS-cAMP pathway in Saccharomyces cerevisiae. Proc Natl Acad Sci 86:8778–8782
Smith TF, Gaitatzes C, Saxena K, Neer EJ (1999) The WD repeat: a common architecture for diverse functions. Trends Biochem Sci 24:181–185. doi:10.1016/S0968-0004(99)01384-5
Song JJ, Garlick JD, Kingston RE (2008) Structural basis of histone H4 recognition by p55. Genes Dev 22:1313–1318. doi:10.1101/gad.1653308
Taylor-Harding B, Binne UK, Korenjak M, Brehm A, Dyson NJ (2004) p55, the Drosophila ortholog of RbAp46/RbAp48, is required for the repression of dE2F2/RBF-regulated genes. Mol Cell Biol 24:9124–9136. doi:10.1128/MCB.24.20.9124-9136.2004
Tyler JK, Bulger M, Kamakaka RT, Kobayashi R, Kadonaga JT (1996) The p55 subunit of Drosophila chromatin assembly factor 1 is homologous to a histone deacetylase-associated protein. Mol Cell Biol 16:6149–6159
Veley KM, Michaels SD (2008) Functional redundancy and new roles for genes of the autonomous floral-promotion pathway. Plant Physiol 147:682–695. doi:10.1104/pp.108.118927
Vermeulen M et al (2010) Quantitative interaction proteomics and genome-wide profiling of epigenetic histone marks and their readers. Cell 142:967–980. doi:10.1016/j.cell.2010.08.020
Verreault A, Kaufman PD, Kobayashi R, Stillman B (1996) Nucleosome assembly by a complex of CAF-1 and acetylated histones H3/H4. Cell 87:95–104
Verreault A, Kaufman PD, Kobayashi R, Stillman B (1998) Nucleosomal DNA regulates the core-histone-binding subunit of the human Hat1 acetyltransferase. Curr Biol 8:96–108
Vezzoli A et al (2010) Molecular basis of histone H3K36me3 recognition by the PWWP domain of Brpf1. Nat Struct Mol Biol 17:617–619. doi:10.1038/nsmb.1797
Wang Y, Reddy B, Thompson J, Wang H, Noma K, Yates JR 3rd, Jia S (2009) Regulation of Set9-mediated H4K20 methylation by a PWWP domain protein. Mol Cell 33:428–437. doi:10.1016/j.molcel.2009.02.002
Wilsker D, Probst L, Wain HM, Maltais L, Tucker PW, Moran E (2005) Nomenclature of the ARID family of DNA-binding proteins. Genomics 86:242–251. doi:10.1016/j.ygeno.2005.03.013
Xie Z et al (2004) Genetic and functional diversification of small RNA pathways in plants. PLoS Biol 2:E104. doi:10.1371/journal.pbio.0020104
Xu Y et al (2013) A matrix protein silences transposons and repeats through interaction with retinoblastoma-associated proteins. Curr Biol 23:345–350. doi:10.1016/j.cub.2013.01.030
Yu CW et al (2011) HISTONE DEACETYLASE6 interacts with FLOWERING LOCUS D and regulates flowering in Arabidopsis. Plant Physiol 156:173–184. doi:10.1104/pp.111.174417
Zhang Y, Sun ZW, Iratni R, Erdjument-Bromage H, Tempst P, Hampsey M, Reinberg D (1998) SAP30, a novel protein conserved between human and yeast, is a component of a histone deacetylase complex. Mol Cell 1:1021–1031
Zilberman D, Cao X, Jacobsen SE (2003) ARGONAUTE4 control of locus-specific siRNA accumulation and DNA and histone methylation. Science 299:716–719. doi:10.1126/science.1079695
Acknowledgments
We thank Dr. Tim Ulmasov for providing the cDNA library and CaMV35S transformation cassette vector, Dr. Barbara Ulmasov for help with transgenic plants, Ms. Linh Ngo for assistance with protein domain modeling, and Dr. Brian Mooney for assistance in LC–MS analyses. Financial support from the University of Missouri is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. S1 Interaction of PWWP/RRM proteins with AtMSI5, AtMSI1 and RbAp48 (TIFF 7753 kb)
11103_2014_280_MOESM3_ESM.tif
Supplementary Fig. S2 Sequences at N-terminal regions of MSI1 like proteins. N-terminal region α-helix is involved in interaction with histone H4 (TIFF 13671 kb)
11103_2014_280_MOESM4_ESM.tif
Supplementary Fig. S3 Comparison of sequences from N-terminal region of At3G09670 involved in interaction with MSI4 with similar region of Arabidopsis homolog At5G02950 possessing RRM domain (Fig. S3a) and regions from homologs from relative species lacking RRM domain (Fig. S3b). (TIFF 34627 kb)
11103_2014_280_MOESM5_ESM.docx
Supplementary Table. S1 AtMSI4 associated proteins (* Proteins with only one peptide present in LC–MS pool. Interaction of RRM domain of At3G48190 with AtMSI4 was confirmed by GST-pull down assay) (DOCX 13 kb)
11103_2014_280_MOESM6_ESM.docx
Supplementary Table. S2 Combined list of AtMSI4 associated proteins from four independent immunoprecipitation identified by LC–MS (IPC-immunoprecipitation from cytoplasmic fraction; IPNE0.25 – 0.25 M NaCl nuclear extract; IPNE0.6 – 0.25-0.6 M nuclear extract; IPNL-nucleolar extract) (DOCX 16 kb)
Rights and permissions
About this article
Cite this article
Kenzior, A., Folk, W.R. Arabidopsis thaliana MSI4/FVE associates with members of a novel family of plant specific PWWP/RRM domain proteins. Plant Mol Biol 87, 329–339 (2015). https://doi.org/10.1007/s11103-014-0280-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-014-0280-z