ABSTRACT
Purpose
The HDAC shuttling inhibitor, YK-4-272 functions by restricting nuclear shuttling of Class II HDACs. Pre-clinical investigations of YK-4-272 bioavailability, pharmacokinetics, in vivo toxicity and tumor growth inhibition were performed to determine its potential as an HDAC shuttling disruptor for use in clinical applications.
Methods
The solubility, lipophilicity, in vitro metabolic stability, in vitro intestinal permeability, and in vivo pharmacokinetics of YK-4-272 were determined by HPLC methods. The anti-tumor activity of YK-4-272 was determined by monitoring athymic Balb/c nude mice bearing PC-3 xenografts.
Results
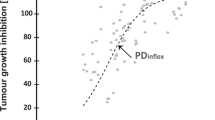
Oral bioavailability of YK-4-272 is supported by its solubility (0.537 mg/mL) and apparent partition coefficient of 2.0. The compound was chemically and metabolically stable and not a substrate for CYP450. In Caco-2 cell transport studies, YK-4-272 was highly permeable. The time-concentration profile of YK-4-272 in plasma resulted in a C max of 2.47 μg/mL at 0.25 h with a AUC of 3.304 μg × h/mL. Treatment of PC-3 tumor xenografts with YK-4-272 showed significant growth delay.
Conclusions
YK-4-272 is stable and bio-available following oral administration. Growth inhibition of cancer cells and tumors was observed. These studies support advancing YK-4-272 for further evaluation as a novel HDAC shuttling inhibitor for use in cancer treatment.




Similar content being viewed by others
Abbreviations
- AMMC:
-
3-[2-(N,N-diehtyl-N-methylamino)ethyl]-7-methoxy-4-methylcoumarin
- BQ:
-
7-benzoyloxyquinoline
- CEC:
-
3-cyano-7-ethoxycoumarin
- CYP450:
-
Cytochrome P450
- MFC:
-
7-methoxy-4-triflouromethylcoumarin
- MRP:
-
multidrug resistance-associated protein
- P-gp:
-
P-glycoprotein
- TEER:
-
transepithelial electrical resistance
REFERENCES
Laherty CD, Yang WM, Sun JM, Davie JR, Seto E, Eisenman RN. Histone deacetylases associated with the mSin3 corepressor mediate mad transcriptional repression. Cell. 1997;89:349–56.
Imai S, Armstrong CM, Kaeberlein M, Guarente L. Transcriptional silencing and longevity protein Sir2 is an NAD-dependent histone deacetylase. Nature. 2000;403:795–800.
de Ruijter AJ, van Gennip AH, Caron HN, Kemp S, van Kuilenburg AB. Histone deacetylases (HDACs): characterization of the classical HDAC family. Biochem J. 2003;370:737–49.
Finnin MS, Donigian JR, Pavletich NP. Structure of the histone deacetylase SIRT2. Nat Struct Biol. 2001;8:621–5.
Gallinari P, Di Marco S, Jones P, Pallaoro M, Steinkuhler C. HDACs, histone deacetylation and gene transcription: from molecular biology to cancer therapeutics. Cell Res. 2007;17:195–211.
Ashburner BP, Westerheide SD, Baldwin Jr AS. The p65 (RelA) subunit of NF-kappaB interacts with the histone deacetylase (HDAC) corepressors HDAC1 and HDAC2 to negatively regulate gene expression. Mol Cell Biol. 2001;21:7065–77.
Blagosklonny MV, Robey R, Sackett DL, Du L, Traganos F, Darzynkiewicz Z, et al. Histone deacetylase inhibitors all induce p21 but differentially cause tubulin acetylation, mitotic arrest, and cytotoxicity. Mol Cancer Ther. 2002;1:937–41.
Naryzhny SN, Lee H. The post-translational modifications of proliferating cell nuclear antigen: acetylation, not phosphorylation, plays an important role in the regulation of its function. J Biol Chem. 2004;279:20194–9.
Varshochi R, Halim F, Sunters A, Alao JP, Madureira PA, Hart SM, et al. ICI182,780 induces p21Waf1 gene transcription through releasing histone deacetylase 1 and estrogen receptor alpha from Sp1 sites to induce cell cycle arrest in MCF-7 breast cancer cell line. J Biol Chem. 2005;280:3185–96.
Halkidou K, Cook S, Leung HY, Neal DE, Robson CN. Nuclear accumulation of histone deacetylase 4 (HDAC4) coincides with the loss of androgen sensitivity in hormone refractory cancer of the prostate. Eur Urol. 2004;45:382–9.
Kong Y, Jung M, Wang K, Grindrod S, Velena A, Lee S, et al. Histone deacetylase cytoplasmic trapping by a novel fluorescent HDAC inhibitor. Mol Cancer Ther 2011;10:1591–99.
Doh MJ, Jung YJ, Kim I, Kong HS, Kim YM. Synthesis and in vitro properties of prednisolone 21-sulfate sodium as a colon-specific prodrug of prednisolone. Arch Pharm Res. 2003;26:258–63.
Lee J, Rho J, Yang Y, Kong H, Jung Y, Kim Y. Synthesis and in vitro evaluation of N-nicotinoylglycyl-2-(5-fluorouracil-1-yl)-D, L-glycine as a colon-specific prodrug of 5-fluorouracil. J Drug Target. 2007;15:199–205.
Uchida M, Fukazawa T, Yamazaki Y, Hashimoto H, Miyamoto Y. A modified fast (4 day) 96-well plate Caco-2 permeability assay. J Pharmacol Toxicol Methods. 2009;59:39–43.
Frederick KS, Maurer TS, Kalgutkar AS, Royer LJ, Francone OL, Winter SM, et al. Pharmacokinetics, disposition and lipid-modulating activity of 5-{2-[4-(3,4-difluorophenoxy)-phenyl]-ethylsulfamoyl}-2-methyl-benzoic acid, a potent and subtype-selective peroxisome proliferator-activated receptor alpha agonist in preclinical species and human. Xenobiotica. 2009;39:766–81.
Bey E, Marchais-Oberwinkler S, Werth R, Negri M, Al-Soud YA, Kruchten P, et al. Design, synthesis, biological evaluation and pharmacokinetics of bis(hydroxyphenyl) substituted azoles, thiophenes, benzenes, and aza-benzenes as potent and selective nonsteroidal inhibitors of 17beta-hydroxysteroid dehydrogenase type 1 (17beta-HSD1). J Med Chem. 2008;51:6725–39.
Madgula VL, Avula B, Pawar RS, Shukla YJ, Khan IA, Walker LA, et al. in vitro metabolic stability and intestinal transport of P57AS3 (P57) from Hoodia gordonii and its interaction with drug metabolizing enzymes. Planta Med. 2008;74:1269–75.
Obach RS, Baxter JG, Liston TE, Silber BM, Jones BC, MacIntyre F, et al. The prediction of human pharmacokinetic parameters from preclinical and in vitro metabolism data. J Pharmacol Exp Ther. 1997;283:46–58.
OECD. Acute oral toxicity-up and down procedure. OECD Guides for testing of chemicals. 2001.
Sheikh KD, Banerjee PP, Jagadeesh S, Grindrod SC, Zhang L, Paige M, et al. Fluorescent epigenetic small molecule induces expression of the tumor suppressor ras-association domain family 1A and inhibits human prostate xenograft. J Med Chem. 2010;53:2376–82.
Jeanne E. Phillips AA. MultiScreen® Caco‐2 Assay System Protocol Note. Optimization of Caco‐2 cell growth and differentiation for drug transport assay studies using a 96‐well assay system. Millipore Corporation 2003:1–12.
Artursson P, Karlsson J. Correlation between oral drug absorption in humans and apparent drug permeability coefficients in human intestinal epithelial (Caco-2) cells. Biochem Biophys Res Commun. 1991;175:880–5.
GENTEST/BDBiosciencesCompany. CYP2C9/MFC high throuoghput inhibitor screening kit instruction manual. 2002.
Sambuy Y, De Angelis I, Ranaldi G, Scarino ML, Stammati A, Zucco F. The Caco-2 cell line as a model of the intestinal barrier: influence of cell and culture-related factors on Caco-2 cell functional characteristics. Cell Biol Toxicol. 2005;21:1–26.
Konsoula Z, Jung M. Involvement of P-glycoprotein and multidrug resistance associated protein 1 on the transepithelial transport of a mercaptoacetamide-based histone-deacetylase inhibitor in Caco-2 cells. Biol Pharm Bull. 2009;32:74–8.
Kerns EH, Di L. Pharmaceutical profiling in drug discovery. Drug Discov Today. 2003;8:316–23.
FDA/CDER. Guidance for industry. Waiver of in vivo bioavailability and bioequivalence studies for immediate-release solid oral dosage forms based on a biopharmaceutics classification system. http://www.fda.gov/cder/guidance/index.htm 2000:1–13.
Royer I, Monsarrat B, Sonnier M, Wright M, Cresteil T. Metabolism of docetaxel by human cytochromes P450: interactions with paclitaxel and other antineoplastic drugs. Cancer Res. 1996;56:58–65.
Long L, Dolan ME. Role of cytochrome P450 isoenzymes in metabolism of O(6)-benzylguanine: implications for dacarbazine activation. Clin Cancer Res. 2001;7:4239–44.
Crespi CL, Miller VP, Penman BW. Microtiter plate assays for inhibition of human, drug-metabolizing cytochromes P450. Anal Biochem. 1997;248:188–90.
Clayton AL, Hazzalin CA, Mahadevan LC. Enhanced histone acetylation and transcription: a dynamic perspective. Mol Cell. 2006;23:289–96.
Amidon GL, Lennernas H, Shah VP, Crison JR. A theoretical basis for a biopharmaceutic drug classification: the correlation of in vitro drug product dissolution and in vivo bioavailability. Pharm Res. 1995;12:413–20.
Yazdanian M, Briggs K, Jankovsky C, Hawi A. The "high solubility" definition of the current FDA Guidance on Biopharmaceutical Classification System may be too strict for acidic drugs. Pharm Res. 2004;21:293–9.
Artursson P, Palm K, Luthman K. Caco-2 monolayers in experimental and theoretical predictions of drug transport. Adv Drug Deliv Rev. 2001;46:27–43.
Wils P, Warnery A, Phung-Ba V, Legrain S, Scherman D. High lipophilicity decreases drug transport across intestinal epithelial cells. J Pharmacol Exp Ther. 1994;269:654–8.
FDA/CDER. Guidance for industry. Bioavailability and Bioequivalence Studies for Orally Administered Drug Products-General Considerations. http://www.fda.gov/cder/guidance/index.htm 2003:1–23.
Novotny-Diermayr V, Sangthongpitag K, Hu CY, Wu X, Sausgruber N, Yeo P, et al. SB939, a novel potent and orally active histone deacetylase inhibitor with high tumor exposure and efficacy in mouse models of colorectal cancer. Mol Cancer Ther. 2010;9:642–52.
Obach RS, Walsky RL, Venkatakrishnan K. Mechanism-based inactivation of human cytochrome p450 enzymes and the prediction of drug-drug interactions. Drug Metab Dispos. 2007;35:246–55.
Obach RS, Walsky RL, Venkatakrishnan K, Houston JB, Tremaine LM. in vitro cytochrome P450 inhibition data and the prediction of drug-drug interactions: qualitative relationships, quantitative predictions, and the rank-order approach. Clin Pharmacol Ther. 2005;78:582–92.
ACKNOWLEDGMENTS AND DISCLOSURES
We thank Youngmi Kim, Yunjin Jung, and Mikell Paige for helpful discussions and technical assistance. This study was supported by National Institutes of Health grants (NIH/NCI P30 CA51008).
Author information
Authors and Affiliations
Corresponding author
Additional information
Hye-Sik Kong and Shuo Tian contributed equally to this publication.
ELECTRONIC SUPPLEMENTARY MATERIAL
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 31 kb)
Rights and permissions
About this article
Cite this article
Kong, HS., Tian, S., Kong, Y. et al. Preclinical Studies of YK-4-272, an Inhibitor of Class II Histone Deacetylases by Disruption of Nucleocytoplasmic Shuttling. Pharm Res 29, 3373–3383 (2012). https://doi.org/10.1007/s11095-012-0832-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-012-0832-3