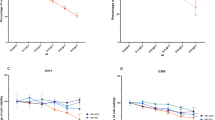
Chronic myelogenous leukemia (CML), also called chronic myeloid leukemia, is a myeloproliferative neoplasm considered one of the three most common types of leukemia. Apoptosis restriction is one of the major problems in CML patients. This process is mainly regulated by the Bcl-2 family protein divided into anti-apoptotic (such as Bcl-2 and Bcl-xL) and pro-apoptotic proteins such as Bax and Bad. In fact, the Bcl-2 family activates caspases – proteolytic enzymes triggering cell destruction. Furthermore, another main element of the apoptotic process is known as p53 protein that is capable of activating apoptosis. Tyrosine kinase inhibitors are a group of enzymes preventing cell apoptosis. Although tyrosine kinase inhibitor (TKI) imatinib is being administered to CMLpatients as a primary therapy, something in the region of 20 – 30% of patients at chronic phase develop acquired resistance to TKI treatment especially due to mutations in ABL1 kinase domain. Stem cell transplantation, another practical remedy, is not without untold harms. Not only is early relapse bound to occur, the risk of mortality from chronic graft-versus-host disease also exists. Hence, investigating new therapeutic approaches using the untapped potential of nature appears to be necessary. The purpose of this study was to assess the mRNA expression levels of Bax, Bcl-2, BCL-XL, and P53 genes in K562 cells after treatment with gum hydroalcoholic extract of Ferula asafoetida. In order to standardize the F. asafoetida gum extract, the total phenolic content (TPC) and total flavonoid content (TFC) of the extract were measured. The MTT assay, PI staining flow cytometry, and RT-PCR assay were applied after a 48 h incubation period in order to evaluate the cytotoxicity of extract on leukemia cells. The TPC and TFC of the extract were 26 mg gallic acid equivalent/g extract and 5.45 mg rutin/g, respectively.With regard to cell survival, there was a statically significant decline in the cell viability at drug doses of 500 and 1000 μg/mL. In addition, flow cytometry analysis showed a considerable apoptosis rate after 48 h treatment. Furthermore, after the 48 h incubation period, there was a rise in P53 and BAX mRNAexpression and decline in BCL-2 and BCL-XL at almost all concentrations of F. asafoetida extract. All the tests were validated by hydroxyurea (5 mM) as the positive control. In conclusion, the current study provides evidence indicating the antiproliferative potency of F. asafoetida extract against human CML (K562) cells. This herbal extract inhibits cell survival and induces apoptosis by rectifying the expression of BAX, BCL-2, BCL-XL, and P53 genes.





Similar content being viewed by others
References
J. Ferlay, I. Soerjomataram, R. Dikshit, et al., Int. J. Cancer., 136(5), 359 – 386 (2015).
H. Zhao, L. Yan, X. Xu, et al., AMB Express., 8(1), 1 – 16 (2018).
F. Bray, J. Ferlay, I. Soerjomataram, et al., Cancer J. Clin., 68(6), 394 – 424 (2018).
K. Krysiak, M. J. Christopher, Z. L. Skidmore, et al., Blood, 6(4), 413 – 413 (2016).
Y. Guo, Y. Li, Q. Shan, et al., Int. J. Biochem. Cell. Biol., 65, 1 – 11 (2015).
A. Rana, S. H. Shah, N. Rehman, et al., J. Cancer Res. Exp. Oncol., 3(6), 62 – 66 (2011).
E. Chamani, Z. Rezaei, K. Dastjerdi, et al., J. Biomol. Struct. Dyn., 38(16), 4838 – 4849 (2020).
E. Marzetti, G. Privitera, V. Simili, et al., Sci. World J., 10, 340 – 349 (2010).
M. S Ola, M. Nawaz, H. Ahsan, et al., Mol. Cell. Biochem., 351(1), 41 – 58 (2011).
J. S Fridman and S. W Lowe, Oncogene, 22(56), 9030 – 9040 (2003).
A. Quintás-Cardama, H. Kantarjian, J. Cortes, et al., Nat. Rev. Clin. Oncol., 6(9), 535 (2009).
R. Kc, B. Thapa, A. Ubeda, et al., Stem Cells Dev., 28(11), 734 – 744 (2019).
I. Sloma, X. Jiang, A. C. Eaves, et al., Leukemia, 24(11), 1823 – 1833 (2010).
J. M Goldman and J. V. Melo, New Engl. J. Med., 349(15), 1451 – 1464 (2003).
M. Baccarani, J. Cortes, F. Pane, et al., J. Clin. Oncol., 27(35), 6041 (2009).
H. Cai, X. He, and C. Yang, Phytother. Res., 32(9), 1764 – 1769 (2018).
M. Martinez-Castillo, N. Villegas-Sepúlveda, M. A. Meraz-Rios, et al., Oncol. Lett., 15(5), 6777 – 6783 (2018).
A. Amalraj and S. Gopi, J. Complement. Med. Res., 7(3), 347 – 359 (2017).
A. A. Dehpour,M. A. Ebrahimzadeh, N. S. Fazel, et al., Grasas Aceites, 60(4), 405 – 412 (2009).
G. Kavoosi and V. Rowshan, Appl. Food Protein Chem., 138(4), 2180 – 2187 (2013).
V. Shrivastava, U. Bhardwaj, V. Sharma, et al., J. Pharm. Res., 5(10), 5022 – 5024 (2012).
S. D Patil, S. Shinde, P. Kandpile, et al., Int. J. Pharm. Sci. Res., 6(2), 722 (2015).
P. Angelini, R. Pagiotti, R. Venanzoni, et al., Allelopathy, 23(2), 357 – 68 (2009).
A. Rani, S. Jain, and P. Dureja, J. Pestic. Sci., 2009, 0908030091 (2009).
Ahmed Saber Abu-Zaiton, Pak. J. Biol. Sci., 13(2), 97 – 100 (2010).
E. Latifi, A. A. Mohammadpour, B. Fathi, et al., Biomed. Pharmacother., 110, 197 – 202 (2019)
G. S. Tayeboon, F. Tavakoli, S. Hassani, et al., In Vitro Cell. Dev. Biol., 49(9), 706 – 715 (2013).
M. Moghaddam and N. Farhadi, J. Med. Arom. Plant Sci., 2(3), 69 – 76 (2015).
G. U. Mallikarjuna, S. Dhanalakshmi, S. Raisuddin, et al., Breast Cancer Res. Treat. 81(1), 1 – 10 (2003).
M. Saleem, A. Alam, and S. Sultana., Am. J. Biomed. Life Sci., 68(16), 1913 – 1921 (2001).
R. Panwar, S. Rana, D. K. Dhawan, et al., J. Phytopharm., 4(6), 282 – 6 (2015).
S. M. Bagheri, A. Abdian-Asl, M. T. Moghadam, et al., J. Ayurveda Integr. Med., 8(3) 152 – 158 (2017).
P. K. Upadhyay, Int. J. Green Pharm., 11(02) (2017).
M. Khalili, Z. Kiasalari, M. Roghani, and Y. Azizi, J. Med. Plants Res., 5(7), 1140 – 1146 (2011).
M. Jalili-Nik, H. Sabri, E. Zamiri, et al., Drug Res., 69(12), 665–670 (2019).
A. U. Ezirim, V. I. Okochi, A. B. James, et al., J. Med. Plants Res., 2(3), 142 (2013).
S. M. Bagheri, A. A. Asl, A. Shams, et al., Indian J. Med. Pediatr. Oncol., 38(2), 116 (2017).
S. D. Sadooghi, K. Nezhad Shahrokh Abadi, and S. Z. Balanzhad., Int. Arch. Health Sci. 17(4), 323 – 330 (2013).
J. Y. Lin and C. Y Tang, Food. Chem., 101(1), 140 – 147 (2007).
H. Fatima, K. Khan, M. Zia, et al., BMC Complement. Altern. Med., 15(1), 1 – 18 (2015).
C. Riccardi and I. Nicoletti, Nat. Protoc., 1(3), 1458 (2006).
E. Chamani, Z. Rezaei, K. Dastjerdi, et al., J. Biomol. Struct. Dyn., 36(16), 4838 – 4849 (2020).
L. Dubrez, B. Eymin, O. Sordet, et al., Blood, 91(7), 2415 – 2422 (1998).
G. P. Amarante-Mendes, C. Naekyung Kim, L. Liu, et al., Blood, 91(5), 1700 – 1705 (1998).
I. Sánchez-García and D. Martin-Zanca, J. Mol. Biol., 225 – 228 (1997).
T. Skorski, A. Bellacosa, M. Nieborowska-Skorska, et al., EMBO J., 16(20), 6151 – 6161 (1997).
B. L. Stiles, Adv. Drug Delivery Rev., 61(14), 1276 – 1282 (2009).
M. Martinez-Castillo, R. Bonilla-Moreno, L. A. Lazarini, et al., PloS One., 11(11) 0165971 (2016).
R. Niazmand and B. M. Razavizadeh, Int. J. Food Sci. Technol., 58(6), 2148 – 2159 (2021).
A. Akbarian, M. Rahimmalek, M. Sabzalian, et al., Pak. J. Med. Sci., 1165 (2018).
X. Zhang, Q. Wu, S. Yang, et al., Pak. J. Sci. Res., 33(1), 127 (2017).
T. Sarwar, M. Zafaryab, M. A. Husain, et al., Toxicol. Appl. Pharmacol., 289(2), 251 – 261 (2015).
T.Wang, X. Gong, R. Jiang, et al., Am. J. Transl. Res., 8(2), 968 (2016).
Z. Aas, E. Babaei, M. Feizi, and G. Dehghan, Asian Pac. J. Cancer Prev., 16(13), 5325 – 5329 (2015).
J. Kasaian and A. Mohammadi, J. Asian Nat. Prod. Res., 20(1), 27 – 35 (2018).
M. Eskandani, J. Abdolalizadeh, H. Hamishehkar, et al., Fitoterapia, 101, 1 – 11 (2015).
K. H. Kim, H. J. Lee, S. J. Jeong, et al., Indo-Am. J. Pharm. Res., 28(3), 597 – 609 (2011).
Y. H. Kim, E. A. Shin, J. H. Jung, et al., Eur. J. Pharmacol, 847, 91 – 96 (2019).
L. Zhang, J. Si, G. Li, et al., RSC Adv., 5(110), 91006 – 91017 (2015).
J. M. Lee and A. Bernstein, Cancer Metastasis Rev., 14(2), 149 – 161 (1995).
D. Hasanzadeh, M. Mahdavi, G. Dehghan, H. Nozad Charoudeh, et al., Toxicol. Rep., 4, 420 – 426 (2017).
M. Abroudi, A. G. Fard, G. Dadashizadeh, et al., Int. J. Biomed Eng. Sci., 6(3), 60 (2020).
J. Wang, H. Wang, M. Zhang, et al., Phytochemistry, 180, 112531 (2020).
A. R. Afshari, M. K. Roshan, M. Soukhtanloo, et al., Avicenna J. Phytomed., 9(4), 334 (2019).
R. T. Silver, S. H. Woolf, R. Hehlmann, et al., Blood, 94(5), 1517 – 1536 (1999).
M. Issaivanan, P. S. Mitu, C. Manisha, et al., J. Dermatol., 21(2), 124 – 127 (2004).
ACKNOWLEDGMENTS
The authors would like to acknowledge the Laboratory of Birjand University of Medical Sciences for providing all research facilities.
CONFLICT OF INTEREST
The authors declare that they have no conflicts of interest.
Funding
This work was supported by the Birjand University of Medical Science (BUMS), Iran.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Talebpour, A., Alipour, R., Sajjadi, S.M. et al. In Vitro Cytotoxicity of Ferula asafoetida Gum Extract on Human Chronic Myelogenous Leukemia K562 Cells. Pharm Chem J 56, 245–253 (2022). https://doi.org/10.1007/s11094-022-02627-w
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-022-02627-w