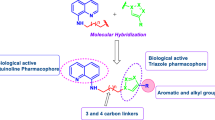
1,2-dihydro-3-methyl-2-oxoquinoxaline-6-carboxylic acid (S1) was prepared and coupled with various amino acid tri/tetrapeptide methyl esters to afford new quinoxalopeptide derivatives (S1a – S1i). The synthesized derivatives were characterized and screened for their antibacterial, antifungal and anthelmintic activity. Furthermore, molecular docking study revealed their potential in targeting human papillomavirus (HPV-16) E6 oncoprotein.



Similar content being viewed by others
References
M. M. Badran, K. A. Abonzid, and M.H. Hussein, Arch. Pharm. Res., 26, 107 – 113 (2003).
R. K. Griffith, S. V. Chittur, and Y. C. Chen, Med. Chem. Res., 2, 467 – 473 (1992).
A. A. El-Gendy, S. El-Meligie, A. El-Ansry, and A. M. Ahmedy, Arch. Pharm. Res., 18, 44 – 47 (1995).
C. V. Reddy-Sastry, K. Shrinivas-Rao, V. S. H. Krishanan, et al., Indian J. Chem., 29, 396 – 403 (1990).
S. A. El-Hawash, N. S. Habib, and N. H. Franki, Pharmazie, 54, 808 – 815 (1999).
G. Westphal, H. Wasiki, U. Zielinski, et al., Pharmazie, 35, 570 – 571 (1977).
A. Monge, F. J. Martinez-Crespo, A. L. Cerai, et al., J. Med. Chem., 38, 4488 – 4495, (1995).
J. W. Michael, T. Ben-Hadda, A. T. Kotchevan, et al., Molecules, 7, 641 – 656 (2002).
J. B. Rangisetty, C. N. Gupta, A. L. Prasad, et al., J. Pharm. Pharmacol., 53, 1409 – 1413 (2001).
A. Burguete, E. Pontiki, D. Hadjipavlou-Litina, et al., Bioorg. Med. Chem. Lett., 17, 6439 – 6443 (2007).
S. D. Undevia, F. Innocenti, J. Ramirez, et al., Eur. J. Cancer., 44, 1684 – 1692 (2008).
P. Corona, A. Carta, M. Loriga, et al., Eur. J. Med. Chem., 44, 1579 – 1591 (2009).
C. Urquiola, D. Gambino, M. Cabrera, et al., Inorg. Biochem., 102, 119 – 126 (2008).
Q. Weng, D. Wang, P. Guo, et al., Eur. J. Pharmacol., 581, 262 – 269 (2008).
S.Wagle, A. V. Adhikari, N. S. Kumari, Eur. J. Med. Chem., 44, 1135 – 1143 (2009).
E. Vicente, L. M. Lima, E. Bongard, et al., Eur. J. Med. Chem., 43, 1903 – 1910, (2008).
A. Burguete, E. Pontiki, D. Hadjipavlou-Litina, et al., Bioorg. Med. Chem. Lett., 17, 6439 – 6443 (2007).
A. Budakoti, A. R. Bhat, A. Azam, Eur. J. Med. Chem., 44, 1317 – 1325 (2009).
M. R. Myers, W. He, B. Hanney, et al., Bioorg. Med. Chem. Lett., 13, 3097 – 3100 (2003).
Y. B. Kim, Y. H. Kim, J. Y. Park, and S. K. Kim, Bioorg. Med. Chem. Lett., 14, 541 – 544 (2004).
B. Poojary, S. L. Belagali, K. Harish Kumar, and B. S. Holla, Farmaco., 58, 569 – 572 (2003).
R. Dahiya and R. Kaur. Int. J. Biol. Chem. Sci., 2, 1 – 13 (2008).
R. Dahiya, D. Pathak, and S. Bhatt, J. Saudi Chem. Soc., 10, 165 – 176 (2006).
R. Dahiya. Sci. Pharm., 76, 217 – 239 (2008).
K. Butz, C. Denk, A. Ullmann, et al., PNAS., 97, 6693 – 6697(2000).
Y. Zhang, J. Dasgupta, R. Z. Ma, et al., J. Virology., 81, 3618 – 3626 (2007).
M. Bodanszky and A. Bodanszky, The Practice of Peptide Synthesis, Springer, New York (1984), pp. 143 – 209.
L. C. Garg and C. K. Atal, Indian J. Pharm. Sci., 59, 240 – 245 (1963).
A. W. Bauer, W. M. Kriby, J. C. Sherris, and M. Turck, Am. J. Clin. pathol., 45, 493 – 496 (1966).
Z. K. Khan, In vitro and vivo screening techniques for bioactivity screening and evaluation, in: Proceedings of Int. Workshop UNIDO-CDR (1997), pp. 210 – 211.
T. L. Aceret, J. C. Coll, Y. Uchio, and P. W. Sammarco, Comp. Biochem. Physiol. C, Pharmacol. Toxicol. Endocrinol., 120, 21 – 126 (1998).
O. Trott and A.J. Olson, J. Comp. Chem., 31, 455 – 461 (2010).
K. Zanier1, S. Charbonnier, A.O.M.O. Sidi, et al., Science, 8, 694 – 698 (2013).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 32 kb)
Rights and permissions
About this article
Cite this article
Chaudhary, S., Kumar, S. & Tarazi, H. Peptide Derivatives of 1,2-Dihydro-3-Methyl-2-Oxoquinoxaline-6-Carboxylic Acid: Synthesis and Evaluation of Antimicrobial, Antifungal and Antiviral Potential. Pharm Chem J 50, 331–338 (2016). https://doi.org/10.1007/s11094-016-1447-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-016-1447-5