Abstract
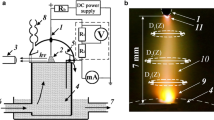
This paper reports the results of the experimental study and chemical composition modeling for a DC nitrogen discharge burning above water cathode in the pressure range of 0.1–1 bar and at discharge current of 40 mA. The gas temperature, vibrational temperatures, reduced electric field strength, cathode voltage drop, and emission intensities of some nitrogen bands were obtained from experiment. The modeling chemical composition of plasma was carried out on the basis of these data. At modeling, the combined solution of Boltzmann equation for electrons, equations of vibrational kinetics for ground states of N2, O2, H2O and NO molecules, equations of chemical kinetics and plasma conductivity equation were used. The calculations agree with the measured bands intensities for the second positive system of N2 and vibrational temperatures of \({\text{N}}_{2} ( {\text{C}}^{3}\Pi _{\text{u}} )\). In the frame of the model proposed, the data of other studies were explained. The second kind collision of electrons with N2 vibration excited states was shown to affect strongly the electron gas parameters. The electron average energy and electron density are given. The difference between the properties of the discharges in N2 and air at the same conditions are discussed as well.










Similar content being viewed by others
References
Bruggeman P, Leys C (2009) J Phys D Appl Phys 42(5):053001
Tatarova E, Bundaleska N, Sarrette J, Ferreira CM (2014) Plasma Sources Sci Technol 23(6):063002
Jiang B, Zheng J, Qiu S, Wu M, Zhang Q, Yan Z, Xue Q (2014) Chem Eng J 236:348–368
Titov VA, Rybkin VV, Maximov AI, Choi H-S (2005) Plasma Chem Plasma Process 25(5):502–518
Verreycken T, Schram DC, Leys C, Bruggeman P (2010) Plasma Sources Sci Technol 19(4):045004
Li L, Nikiforov A, Xiong Q, Lu X, Taghizadeh L, Leys C (2012) J Phys D Appl Phys 45(12):125201
Nikiforov A, Li L, Xiong Q, Leys C, Lu XP (2011) Eur Phys J Appl Phys 56(2):24009
Ito H, Kano H (2008) Appl Phys Express 1:10601–10603
Liu DX, Bruggeman P, Iza F, Rong MZ, Kong MC (2010) Plasma Sources Sci Technol 19(2):025018
Zhang S, Van Gaens W, Van Gessel B, Hofman S, Van Veldhuizen E, Bogaerts A, Bruggeman P (2013) J Phys D Appl Phys 46(20):205202
Van Gaens W, Bruggeman PJ, Bogaerts A (2014) New J Phys 16:063054
Shutov DA, Smirnov SA, Bobkova ES, Rybkin VV (2014) Plasma Chem. DOI, Plasma Process. doi:10.1007/s11090-014-9596-3
Bobkova ES, Smirnov SA, Zalipaeva YV, Rybkin VV (2014) Plasma Chem Plasma Process 34(4):721–743
Bruggeman P, Schram D, Gonzalez MA, Rego R, Kong MG, Leys C (2009) Plasma Sources Sci Technol 18(2):025017
Titov VA, Rybkin VV, Smirnov SA, Kulentsan AN, Choi H-S (2006) Plasma Chem Plasma Process 26(6):543–555
Raizer YuP (1991) Gas discharge physics. Springer, Berlin
Gordiets BF, Ferreira CM, Guerra VL, Loureiro J, Nahorny J, Pagnon D, Touzeau Vialle M (1995) IEEE Trans Plasma Sci 23(23):750–768
Kajita S, Ushiroda S, Kondo Y (1990) J Phys D Appl Phys 67(9):4015–4023
Rybkin VV, Titov VA, Kholodkov IV (2008) Izv Vyssh Uchebn Zaved Khim Khim Tekhnol 51(3):3–10 (in Russian)
Rybkin VV, Titov VA, Kholodkov IV (2009) Izv Vyssh Uchebn Zaved Khim Khim Tekhnol 52(12):3–10 (in Russian)
Laher RR, Gilmore FR (1990) J Phys Chem Ref Data 19(1):277–304
Louriero J, Ferreira CM (1989) J Phys D Appl Phys 22(11):1680–1691
Diamy A-M, Legrand J-C, Smirnov SA, Rybkin VV (2005) Contrib Plasma Phys 45(1):5–21
Schwartz RN, Slawsky ZI, Herzfeld KF (1952) J Chem Phys 20(10):1591
Capitelli M (ed) (1986) Nonequilibrium vibrational kinetics. Springer, Berlin
Kiefer JH (1972) J Chem Phys 57(5):938–1956
Mnatsakanyan AK, Naidis GV (1985) High Temperature (Teplofyzika Vysokikh Temperatur) 23(4):640–646 (in Russian)
Salnikov VA, Starik AM (1995) High Temperature (Teplofyzika Vysokikh Temperatur) 33(1):121–133 (in Russian)
Wysong IJ (1994) J Chem Phys 101(4):2800–2810
Frost MJ, Islam M, Smith IWM (1994) Can J Chem 72(3):606–611
Huber KP, Herzberg G (1979) Molecular spectra and molecular structure IV. Constants of diatomic molecules. Litton Educational Publishing, Inc, New York
Glushko VP (ed) (1978) Thermodynamic properties of individual substances. Handbook. Nauka, Moscow (in Russian)
Radtsig AA, Smirnov BM (1980) Handbook on atomic and molecular physics. Atomizdat, Moscow (in Russian)
Herron JT (1999) J Phys Chem Ref Data 28(5):1453–1483
Javoy S, Naudet V, Abid S, Paillard CE (2003) Exp Thermal Fluid Sci 27(4):371–377
Clyne MAA, Stedman DH (1967) J Phys Chem 71(9):3071–3073
Nicholls RW (1964) Ann Geophys 20:144–151
Pancheshnyi SV, Starikovskaia SM, Starikovskii AY (1998) Chem Phys Lett 294(6):523–527
Slovetskiy DI (1980) Mechanism of chemical reactions in the non-equilibrium plasma. Nauka, Moscow (in Russian)
Tikhonov AN, Arsenin VY (1974) Methods of solution of incorrect tasks. Nauka, Moscow (in Russian)
Grigoriev IS, Meiylikhov EZ (eds) (1991) Physical quantities. Handbook. Energoatomizdat, Moscow (in Russian)
Staack D, Farouk B, Gutsol A, Fridman A (2005) Plasma Sources Sci Technol 14(4):700–711
Kim YH, Hong YJ, Baik KY, Kwon GC, Vhoi JJ, Cho GS, Uhm HS, Kim DY, Choi EH (2014) Plasma Chem Plasma Process 34(3):457–472
Smirnov SA, Rybkin VV, Kholodkov IV, Titov VA (2002) High Temp 40(3):323–330
Bobkova ES, Rybkin VV (2013) High Temp 51(6):747–752
Acknowledgments
This study was supported by the RFBR grant, project number 14-02-01113 A.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Smirnov, S.A., Shutov, D.A., Bobkova, E.S. et al. Physical Parameters and Chemical Composition of a Nitrogen DC Discharge with Water Cathode. Plasma Chem Plasma Process 35, 639–657 (2015). https://doi.org/10.1007/s11090-015-9626-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11090-015-9626-9