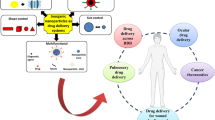
Abstract
Vascular dementia (VaD) has a serious impact on the patients’ quality of life. Icariin (Ica) possesses neuroprotective potential for treating VaD, yet its oral bioavailability and blood–brain barrier (BBB) permeability remain challenges. This research introduced a PEG–PLGA-loaded chitosan hydrogel-based binary formulation tailored for intranasal delivery, enhancing the intracerebral delivery efficacy of neuroprotective agents. The formulation underwent optimization to facilitate BBB crossing, with examinations conducted on its particle size, morphology, drug-loading capacity, in vitro release, and biodistribution. Using the bilateral common carotid artery occlusion (BCCAO) rat model, the therapeutic efficacy of this binary formulation was assessed against chitosan hydrogel and PEG–PLGA nanoparticles loaded with Ica. Post-intranasal administration, enhanced cognitive function was evident in chronic cerebral hypoperfusion (CCH) rats. Further mechanistic evaluations, utilizing immunohistochemistry (IHC), RT-PCR, and ELISA, revealed augmented transcription of synaptic plasticity-associated proteins like SYP and PSD-95, and a marked reduction in hippocampal inflammatory markers such as IL-1β and TNF-α, highlighting the formulation's promise in alleviating cognitive impairment. The brain-derived neurotrophic factor (BDNF)/tropomyosin related kinase B (TrkB) pathway was activated significantly in the binary formulation compared with the other two. Our study demonstrates that the intranasal application of chitosan hydrogel loaded with Ica-encapsulated PEG–PLGA could effectively deliver Ica into the brain and enhance its neuroprotective effect.










Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
Abbreviations
- AD:
-
Alzheimer’s disease
- BBB:
-
Blood brain barrier
- BCCAO:
-
Bilateral common carotid artery occlusion
- BDNF:
-
Brain-derived neurotrophic factor
- CaMKII:
-
Ca2+/calmodulin-dependent protein kinase II
- CCH:
-
Chronic cerebral hypoperfusion
- CREB:
-
cAMP-response element binding protein
- CSF:
-
Cerebrospinal fluid
- DLS:
-
Dynamic light scattering
- DMF:
-
N,N-dimethylformamide
- EA:
-
Ethyl acrylate
- EF:
-
Epimedium flavonoid
- ELISA:
-
Enzyme-linked immunosorbent assay
- ER:
-
Endoplasmic reticulum
- ERK:
-
Extracellular regulated protein kinases
- FDA:
-
Food and Drug Administration
- Gel:
-
Hydrogel
- HE:
-
Hematoxylin and eosin
- HPLC:
-
High performance liquid chromatography
- Ica:
-
Icariin
- Ica gel:
-
Ica chitosan hydrogel
- Ica ig:
-
Ica Intragastric administration
- Ica NPs and gel:
-
Ica-PEG–PLGA NPs and chitosan hydrogel hydrogel
- Ica NPs group:
-
Ica-PEG–PLGA NPs
- IHC:
-
immunohistochemistry
- IL-1β/IL-1b:
-
Interleukin 1β
- IL6:
-
Interleukin 6
- LTP:
-
Long-term potentiation
- MWM:
-
Morris water maze
- NMDA:
-
N-methyl d-aspartate
- NMDARs:
-
N-methyl D-aspartate receptors
- NPs:
-
nanoparticles
- GluN2B:
-
A main subunit of the NMDA receptor
- NSCs:
-
Neural stem cells
- PBS:
-
Phosphate buffered saline
- PDI:
-
Polydispersity Index
- PEG–PLGA:
-
Poly(ethyleneglycol)–poly(lactic-co-glycolic acid)
- P-gp:
-
P-glycoprotein
- PSD-95:
-
Post-synaptic density 95
- PVA:
-
Polyvinyl alcohol
- RCTs:
-
Randomized controlled trails
- RT-PCR:
-
Reverse transcription-polymerase chain reaction
- SD:
-
Sprague Dawley
- ß-GP:
-
β-Glycerophosphate
- SYP:
-
Synaptophysin
- TB:
-
Toluidine blue
- TEM:
-
Transmission electron microscopy
- TFE:
-
Epimedium koreanum
- TJ:
-
Tight junction
- TNF-α:
-
Tumor necrosis factor-α
- TrkB:
-
Tropomyosin receptor kinas B
- VaD:
-
Vascular dementia
- VCI:
-
Vascular cognitive impairment
References
Du SQ, Wang XR, Xiao LY, Tu JF, Zhu W, He T, Liu CZ (2017) Molecular mechanisms of vascular dementia: what can be learned from animal models of chronic cerebral hypoperfusion? Mol Neurobiol 54(5):3670–3682. https://doi.org/10.1007/s12035-016-9915-1
Yang Y, Zhao X, Zhu Z, Zhang L (2022) Vascular dementia: a microglia’s perspective. Ageing Res Rev 81:101734. https://doi.org/10.1016/j.arr.2022.101734
Tian Z, Ji X, Liu J (2022) Neuroinflammation in vascular cognitive impairment and dementia: current evidence, advances, and prospects. Int J Mol Sci 23(11):6224. https://doi.org/10.3390/ijms23116224
Zhou C, Sun P, Xu Y, Chen Y, Huang Y, Hamblin MH, Foley L, Hitchens TK, Li S, Yin KJ (2022) Genetic deficiency of MicroRNA-15a/16-1 confers resistance to neuropathological damage and cognitive dysfunction in experimental vascular cognitive impairment and dementia. Adv Sci 9(17):e2104986. https://doi.org/10.1002/advs.202104986
Zanon Zotin MC, Sveikata L, Viswanathan A, Yilmaz P (2021) Cerebral small vessel disease and vascular cognitive impairment: from diagnosis to management. Curr Opin Neurol 34(2):246–257. https://doi.org/10.1097/WCO.0000000000000913
Montine TJ, Bukhari SA, White LR (2021) Cognitive impairment in older adults and therapeutic strategies. Pharmacol Rev 73:152–162
Rundek T, Tolea M, Ariko T et al (2022) Vascular cognitive impairment (VCI). Neurotherapeutics. 19:68–88
Bai X, Zhang M (2021) Traditional Chinese medicine intervenes in vascular dementia: traditional medicine brings new expectations. Front Pharmacol 12:689625. https://doi.org/10.3389/fphar.2021.689625
Ke L, Duan X, Cui J et al (2023) Research progress on the extraction technology and activity study of epimedium polysaccharides. Carbohydr Polym 306:120602
Niu HM, Ma DL, Wang MY et al (2020) Epimedium flavonoids protect neurons and synapses in the brain via activating NRG1/ErbB4 and BDNF/Fyn signaling pathways in a chronic cerebral hypoperfusion rat model. Brain Res Bull 162:132–140. https://doi.org/10.1016/j.brainresbull.2020.06.012
Chen FJ, Liu B, Wu Q et al (2019) Icariin delays brain aging in senescence-accelerated mouse prone 8 (SAMP8) model via inhibiting autophagy. J Pharmacol Exp Ther 369:121–128. https://doi.org/10.1124/jpet.118.253310
Ma D, Zhao L, Zhang L et al (2021) Icariin promotes survival, proliferation, and differentiation of neural stem cells in vitro and in a rat model of Alzheimer’s disease. Stem Cells Int 2021:9974625. https://doi.org/10.1155/2021/9974625
Fu X, Li S, Zhou S et al (2018) Stimulatory effect of icariin on the proliferation of neural stem cells from rat hippocampus. BMC Complement Altern Med 18:1–9
Wang Y, Shang C, Zhang Y et al (2023) Regulatory mechanism of icariin in cardiovascular and neurological diseases. Biomed Pharmacotherapy 158:114156
Li F, Zhang Y, Lu X et al (2019) Icariin improves the cognitive function of APP/PS1 mice via suppressing endoplasmic reticulum stress. Life Sci 234:116739. https://doi.org/10.1016/j.lfs.2019.116739
Ramezani M, Meymand AZ, Khodagholi F et al (2023) A role for flavonoids in the prevention and/or treatment of cognitive dysfunction, learning, and memory deficits: a review of preclinical and clinical studies. Nutr Neurosci 26:156–172
Szabó R, Rácz CP, Dulf FV (2022) Bioavailability improvement strategies for icariin and its derivates: a review. Int J Mol Sci 23:7519
Khezri MR, Ghasemnejad-Berenji M (2022) Icariin: a potential neuroprotective agent in Alzheimer’s disease and Parkinson’s disease. Neurochem Res 47:2954–2962
Zhang Y, Wang Q-S, Cui Y-L et al (2012) Changes in the intestinal absorption mechanism of icariin in the nanocavities of cyclodextrins. Int J Nanomed. https://doi.org/10.2147/IJN.S33014
Wang J, Kong L, Guo R-B et al (2022) Multifunctional icariin and tanshinone IIA co-delivery liposomes with potential application for Alzheimer’s disease. Drug Deliv 29:1648–1662
Li X, Li S, Ma C et al (2022) Preparation of baicalin-loaded ligand-modified nanoparticles for nose-to-brain delivery for neuroprotection in cerebral ischemia. Drug Deliv 29:1282–1298. https://doi.org/10.1080/10717544.2022.2064564
Vaz G, Clementino A, Mitsou E et al (2022) In vitro evaluation of curcumin- and quercetin-loaded nanoemulsions for intranasal administration: effect of surface charge and viscosity. Pharmaceutics. https://doi.org/10.3390/pharmaceutics14010194
Bonaccorso A, Gigliobianco MR, Pellitteri R et al (2020) Optimization of curcumin nanocrystals as promising strategy for nose-to-brain delivery application. Pharmaceutics. https://doi.org/10.3390/pharmaceutics12050476
Agrawal M, Saraf S, Saraf S et al (2018) Nose-to-brain drug delivery: an update on clinical challenges and progress towards approval of anti-Alzheimer drugs. J Control Release 281:139–177. https://doi.org/10.1016/j.jconrel.2018.05.011
Kozlovskaya L, Abou-Kaoud M, Stepensky D (2014) Quantitative analysis of drug delivery to the brain via nasal route. J Control Release 189:133–140. https://doi.org/10.1016/j.jconrel.2014.06.053
Nagaraja S, Basavarajappa GM, Karnati RK et al (2021) Ion-triggered in situ gelling nanoemulgel as a platform for nose-to-brain delivery of small lipophilic molecules. Pharmaceutics. https://doi.org/10.3390/pharmaceutics13081216
Chung EP, Cotter JD, Prakapenka AV et al (2020) Targeting small molecule delivery to the brain and spinal cord via intranasal administration of rabies virus glycoprotein (RVG29)-modified PLGA nanoparticles. Pharmaceutics. https://doi.org/10.3390/pharmaceutics12020093
Revdekar A, Shende P (2021) Block copolymers in Alzheimer’s disease therapy: a perceptive to revolutionize biomaterials. J Control Release. 340:271–281. https://doi.org/10.1016/j.jconrel.2021.11.007
Cunha S, Swedrowska M, Bellahnid Y et al (2022) Thermosensitive in situ hydrogels of rivastigmine-loaded lipid-based nanosystems for nose-to-brain delivery: characterisation, biocompatibility, and drug deposition studies. Int J Pharm. 620:121720. https://doi.org/10.1016/j.ijpharm.2022.121720
Yu S, Xu X, Feng J et al (2019) Chitosan and chitosan coating nanoparticles for the treatment of brain disease. Int J Pharm 560:282–293
Colucci-D’Amato L, Speranza L, Volpicelli F (2020) Neurotrophic factor BDNF, physiological functions and therapeutic potential in depression, neurodegeneration and brain cancer. Int J Mol Sci 21(20):7777. https://doi.org/10.3390/ijms21207777
Tejeda GS, Díaz-Guerra M (2017) Integral characterization of defective BDNF/TrkB signalling in neurological and psychiatric disorders leads the way to new therapies. Int J Mol Sci 18(2):268. https://doi.org/10.3390/ijms18020268
Leal G, Comprido D, Duarte CB (2014) BDNF-induced local protein synthesis and synaptic plasticity. Neuropharmacology 76:639–656. https://doi.org/10.1016/j.neuropharm.2013.04.00
Arévalo JC, Deogracias R (2023) Mechanisms controlling the expression and secretion of BDNF. Biomolecules 13(5):789. https://doi.org/10.3390/biom13050789
Luo H, Xiang Y, Qu X, Liu H, Liu C, Li G, Han L, Qin X (2019) Apelin-13 suppresses neuroinflammation against cognitive deficit in a streptozotocin-induced rat model of Alzheimer’s disease through activation of BDNF-TrkB signaling pathway. Front Pharmacol 10:395. https://doi.org/10.3389/fphar.2019.00395
Khayatan D, Razavi SM, Arab ZN, Niknejad AH, Nouri K, Momtaz S, Gumpricht E, Jamialahmadi T, Abdolghaffari AH, Barreto GE, Sahebkar A (2022) Protective effects of curcumin against traumatic brain injury. Biomed Pharmacotherapy 154:113621. https://doi.org/10.1016/j.biopha.2022.113621
Su Y, Sun B, Gao X et al (2020) Chitosan hydrogel doped with PEG-PLA nanoparticles for the local delivery of miRNA-146a to treat allergic rhinitis. Pharmaceutics. https://doi.org/10.3390/pharmaceutics12100907
Moya-Alvarado G, Guerra MV, Tiburcio R, Bravo E, Bronfman FC (2022) The Rab11-regulated endocytic pathway and BDNF/TrkB signaling: roles in plasticity changes and neurodegenerative diseases. Neurobiol Dis 171:105796. https://doi.org/10.1016/j.nbd.2022.105796
Langdon KD, Cordova CA, Granter-Button S et al (2018) Executive dysfunction and blockage of brain microvessels in a rat model of vascular cognitive impairment. J Cereb Blood Flow Metab 38:1727–1740. https://doi.org/10.1177/0271678x17739219
Tian H, Ding N, Guo M et al (2019) Analysis of learning and memory ability in an Alzheimer’s disease mouse model using the Morris water maze. J Vis Exp. https://doi.org/10.3791/60055
Qiu L, Pan W, Luo D et al (2020) Dysregulation of BDNF/TrkB signaling mediated by NMDAR/Ca(2+)/calpain might contribute to postoperative cognitive dysfunction in aging mice. J Neuroinflamm 17:23. https://doi.org/10.1186/s12974-019-1695-x
Lu Y, Li CJ, Chen C et al (2016) Activation of GABAB2 subunits alleviates chronic cerebral hypoperfusion-induced anxiety-like behaviours: a role for BDNF signalling and Kir3 channels. Neuropharmacology 110:308–321. https://doi.org/10.1016/j.neuropharm.2016.08.007
Wang G, An T, Lei C et al (2022) Antidepressant-like effect of ginsenoside Rb1 on potentiating synaptic plasticity via the miR-134-mediated BDNF signaling pathway in a mouse model of chronic stress-induced depression. J Ginseng Res 46:376–386. https://doi.org/10.1016/j.jgr.2021.03.005
Miyanohara J, Kakae M, Nagayasu K et al (2018) TRPM2 channel aggravates CNS inflammation and cognitive impairment via activation of microglia in chronic cerebral hypoperfusion. J Neurosci 38:3520–3533. https://doi.org/10.1523/jneurosci.2451-17.2018
Zheng Y, Zhu G, He J et al (2019) Icariin targets Nrf2 signaling to inhibit microglia-mediated neuroinflammation. Int Immunopharmacol 73:304–311
Wolters FJ, Ikram MA (2019) Epidemiology of vascular dementia: nosology in a time of epiomics. Arterioscler Thromb Vasc Biol 39:1542–1549
Dichgans M, Leys D (2017) Vascular cognitive impairment. Circ Res 120:573–591
Van Der Flier WM, Skoog I, Schneider JA et al (2018) Vascular cognitive impairment. Nat Rev Dis Primers 4:1–16
Yang T, Sun Y, Lu Z et al (2017) The impact of cerebrovascular aging on vascular cognitive impairment and dementia. Ageing Res Rev 34:15–29
Iadecola C, Duering M, Hachinski V et al (2019) Vascular cognitive impairment and dementia: JACC scientific expert panel. J Am Coll Cardiol 73:3326–3344
Zhai M, He L, Ju X et al (2015) Icariin acts as a potential agent for preventing cardiac ischemia/reperfusion injury. Cell Biochem Biophys 72:589–597. https://doi.org/10.1007/s12013-014-0506-3
Huang R, Xu Y, Lu X et al (2021) Melatonin protects inner retinal neurons of newborn mice after hypoxia-ischemia. J Pineal Res 71:e12716. https://doi.org/10.1111/jpi.12716
Sanders O, Rajagopal L (2020) Phosphodiesterase inhibitors for Alzheimer’s disease: a systematic review of clinical trials and epidemiology with a mechanistic rationale. J Alzheimers Dis Rep 4:185–215. https://doi.org/10.3233/adr-200191
Lu Q, Zhu H, Liu X et al (2020) Icariin sustains the proliferation and differentiation of Aβ(25–35)-treated hippocampal neural stem cells via the BDNF-TrkB-ERK/Akt signaling pathway. Neurol Res 42:936–945. https://doi.org/10.1080/01616412.2020.1792701
Skaper SD, Facci L, Zusso M et al (2017) Synaptic plasticity, dementia and Alzheimer disease. CNS Neurol Disord Drug Targets 16:220–233. https://doi.org/10.2174/1871527316666170113120853
Català-Solsona J, Miñano-Molina AJ, Rodríguez-Álvarez J (2021) Nr4a2 transcription factor in hippocampal synaptic plasticity, memory and cognitive dysfunction: a perspective review. Front Mol Neurosci 14:287
Buffington SA, Huang W, Costa-Mattioli M (2014) Translational control in synaptic plasticity and cognitive dysfunction. Annu Rev Neurosci 37:17–38. https://doi.org/10.1146/annurev-neuro-071013-014100
Shang Y, Wang X, Shang X et al (2016) Repetitive transcranial magnetic stimulation effectively facilitates spatial cognition and synaptic plasticity associated with increasing the levels of BDNF and synaptic proteins in Wistar rats. Neurobiol Learn Mem 134:369–378. https://doi.org/10.1016/j.nlm.2016.08.016
Yao M, Meng M, Yang X et al (2022) POSH regulates assembly of the NMDAR/PSD-95/Shank complex and synaptic function. Cell Rep 39:110642. https://doi.org/10.1016/j.celrep.2022.110642
Wang M, Jiang L, Chen H et al (2019) Levetiracetam protects against cognitive impairment of subthreshold convulsant discharge model rats by activating protein kinase C (PKC)-growth-associated protein 43 (GAP-43)-calmodulin-dependent protein kinase (CaMK) signal transduction pathway. Med Sci Monit 25:4627–4638. https://doi.org/10.12659/msm.913542
Rosenberg GA (2017) Extracellular matrix inflammation in vascular cognitive impairment and dementia. Clin Sci (Lond) 131:425–437. https://doi.org/10.1042/cs20160604
Wang X, Zhang B, Xia R et al (2020) Inflammation, apoptosis and autophagy as critical players in vascular dementia. Eur Rev Med Pharmacol Sci 24:9601–9614. https://doi.org/10.26355/eurrev_202009_23048
Wu S, Pan B, Tsai S et al (2020) BDNF reverses aging-related microglial activation. J Neuroinflamm 17:210. https://doi.org/10.1186/s12974-020-01887-1
Acknowledgements
The authors would like to acknowledge financial supports from Science and Technology Department of Jilin Province [Grant Number: 20200201467JC].
Funding
This work was supported in part by Science and Technology Department of Jilin Province [Grant Number: 20200201467JC].
Author information
Authors and Affiliations
Contributions
Writing (original draft), graphics drawing and editing, tissue sample analysis, project administration, data curation and interpretation, T.L.; conceptualization and methodology, S.L.; writing (review and editing revising the manuscript critically for intellectual content), investigation, and data analysis, Y.X.; animal experimentation, X.L.; writing (manuscript revising), manufacturing, and characterization of binary formulation, C.M. and Z.G.; formulation design, project coordination, and funding acquisition, L.Y.; All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical Approval and Informed Consent
All animal experiments in this study were conducted in accordance with the protocols of the Laboratory Animal Ethics Committee of the School of Pharmacy, Jilin University (approval Document No.: 20220036). There are currently no alternatives that mimic the nose-to-brain pathway, which is very complex. The experiments in vitro could not fully disclose the delivery efficiency and safety assessment of the formulation in vivo. A total of 120 male Sprague Dawley (SD) rats (12 weeks old) with body weights between 300 to 320 g were housed at a temperature of 22 ± 2℃, a relative humidity of 50 ± 2%, and a 12-h dark light cycle. All the experimental rats took food and drank water as they pleased. In order to minimize rats suffering, the invasive surgeries on rats were performed under anesthesia (intraperitoneal injections with 3% sodium pentobarbital (1.5 mL/kg)). In addition, invasive operations were performed by skilled technicians in order to speed up operations and reduce the size of the surgical area for rats, which maximized the survival rate of rats and reduced the number of rats in the experiments. At the end of the study, the rats were anesthetized by intraperitoneal injection of sodium pentobarbital and then euthanized in a thermostatically controlled dark box filled with 70% CO2. The study has adhered to the ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, T., Li, S., Xiong, Y. et al. Binary Nano-inhalant Formulation of Icariin Enhances Cognitive Function in Vascular Dementia via BDNF/TrkB Signaling and Anti-inflammatory Effects. Neurochem Res (2024). https://doi.org/10.1007/s11064-024-04129-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11064-024-04129-5