Abstract
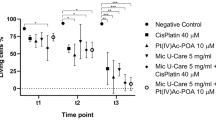
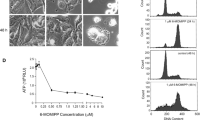
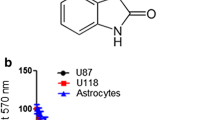
Colchicine, an anti-microtubule and antimitotic drug, is a common therapeutically agent for gout, which is thought to have potential anti-tumor effects. Owing to concerns of colchicines poisoning, the development of derivatives with low dose efficacy and less side effects is of obvious interest. In this study, we characterized the inhibitory effects of a colchicine derivative named AD1 on the cell proliferation of human malignant glioblastoma (MG) cell lines, U87MG and U373MG. We found that 50 % of U87MG and U373MG cells were reduced in the cultures after exposure to AD1 for 24 h at 10 and 50 nM, respectively. Moreover, α-tubulin immunostaining indicated that AD1 induced the disruption of the microtubule polymerization in glioma cells with apoptotic features including membrane budding/blebbing or fragmented nuclei. Increased levels of reactive oxygen species (ROS) were also detected in AD1-treated U87MG and U373MG cells compared to that observed in the control culture. Moreover, examination of microtubule-associated protein 1A/1B-light chain 3 (LC3I)/LC3II conversion and acridine orange staining for autophagic vesicles, combined with flow cytometry, showed that treatment with AD1 induced the autophagic pathway in U87MG and U373MG cells. Furthermore, we found that the intermittent intravenous administration of AD1 suppressed glioma growth in rat brain receiving intracerebral injection with rat C6 glioma cells. Taken together, our findings reveal that treatment with AD1 at nanomolar scales can reduce glioma cell viability effectively, with the occurrence of a rise in ROS and cellular autophagy. In conjunction with the observations from in vivo study, the colchicine derivative AD1 has chemotherapeutic potential to suppress glioma progression.





Similar content being viewed by others
References
Reardon DA, Wen PY (2006) Therapeutic advances in the treatment of glioblastoma: rationale and potential role of targeted agents. Oncologist 11:152–164
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO, European Organisation for R, Treatment of Cancer Brain T, Radiotherapy G, National Cancer Institute of Canada Clinical Trials G (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996
Perry J, Okamoto M, Guiou M, Shirai K, Errett A, Chakravarti A (2012) Novel therapies in glioblastoma. Neurol Res Int 2012:428565
Zhou J, Giannakakou P (2005) Targeting microtubules for cancer chemotherapy. Curr Med Chem Anticancer Agents 5:65–71
Marzo I, Naval J (2013) Antimitotic drugs in cancer chemotherapy: promises and pitfalls. Biochem Pharmacol 86:703–710
Jordan MA, Wilson L (2004) Microtubules as a target for anticancer drugs. Nat Rev Cancer 4:253–265
Zhang Q, Liu X, Li X, Li C, Zhou H, Yan B (2013) Antitumor activity of (2E,5Z)-5-(2-hydroxybenzylidene)-2-((4-phenoxyphenyl)imino) thiazolidin-4-one, a novel microtubule-depolymerizing agent, in U87MG human glioblastoma cells and corresponding mouse xenograft model. J Pharmacol Sci 122:223–231
Ahern MJ, Reid C, Gordon TP, McCredie M, Brooks PM, Jones M (1987) Does colchicine work? The results of the first controlled study in acute gout. Aust N Z J Med 17:301–304
Adler Y, Finkelstein Y, Guindo J, Rodriguez de la Serna A, Shoenfeld Y, Bayes-Genis A, Sagie A, Bayes de Luna A, Spodick DH (1998) Colchicine treatment for recurrent pericarditis. A decade of experience. Circulation 97:2183–2185
Bhattacharyya B, Panda D, Gupta S, Banerjee M (2008) Anti-mitotic activity of colchicine and the structural basis for its interaction with tubulin. Med Res Rev 28:155–183
Craig DH, Owen CR, Conway WC, Walsh MF, Downey C, Basson MD (2008) Colchicine inhibits pressure-induced tumor cell implantation within surgical wounds and enhances tumor-free survival in mice. J Clin Investig 118:3170–3180
De Vincenzo R, Scambia G, Ferlini C, Distefano M, Filippini P, Riva A, Bombardelli E, Pocar D, Gelmi ML, Benedetti Panici P, Mancuso S (1998) Antiproliferative activity of colchicine analogues on MDR-positive and MDR-negative human cancer cell lines. Anticancer Drug Des 13:19–33
Liu W, Sun P, Yang L, Wang J, Li L, Wang J (2010) Assay of glioma cell responses to an anticancer drug in a cell-based microfluidic device. Microfluid Nanofluid 9:717–725
Yang CS, Tzou BC, Liu YP, Tsai MJ, Shyue SK, Tzeng SF (2008) Inhibition of cadmium-induced oxidative injury in rat primary astrocytes by the addition of antioxidants and the reduction of intracellular calcium. J Cell Biochem 103:825–834
Jiang H, White EJ, Conrad C, Gomez-Manzano C, Fueyo J (2009) Autophagy pathways in glioblastoma. Methods Enzymol 453:273–286
Kanzawa T, Kondo Y, Ito H, Kondo S, Germano I (2003) Induction of autophagic cell death in malignant glioma cells by arsenic trioxide. Cancer Res 63:2103–2108
Fang KM, Lin TC, Chan TC, Ma SZ, Tzou BC, Chang WR, Liu JJ, Chiou SH, Yang CS, Tzeng SF (2013) Enhanced cell growth and tumorigenicity of rat glioma cells by stable expression of human CD133 through multiple molecular actions. Glia 61:1402–1417
Fang KM, Wang YL, Huang MC, Sun SH, Cheng H, Tzeng SF (2011) Expression of macrophage inflammatory protein-1alpha and monocyte chemoattractant protein-1 in glioma-infiltrating microglia: involvement of ATP and P2X(7) receptor. J Neurosci Res 89:199–211
Endo K, Mizuguchi M, Harata A, Itoh G, Tanaka K (2010) Nocodazole induces mitotic cell death with apoptotic-like features in Saccharomyces cerevisiae. FEBS Lett 584:2387–2392
Biggers JW, Nguyen T, Di X, Gupton JT, Henderson SC, Emery SM, Alotaibi M, White KL Jr, Brown R, Almenara J, Gewirtz DA (2013) Autophagy, cell death and sustained senescence arrest in B16/F10 melanoma cells and HCT-116 colon carcinoma cells in response to the novel microtubule poison, JG-03-14. Cancer Chemother Pharmacol 71:441–455
Kabeya Y, Mizushima N, Ueno T, Yamamoto A, Kirisako T, Noda T, Kominami E, Ohsumi Y, Yoshimori T (2000) LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. EMBO J 19:5720–5728
Paglin S, Hollister T, Delohery T, Hackett N, McMahill M, Sphicas E, Domingo D, Yahalom J (2001) A novel response of cancer cells to radiation involves autophagy and formation of acidic vesicles. Cancer Res 61:439–444
Fang KM, Wang YL, Huang MC, Sun SH, Cheng H, Tzeng SF (2011) Expression of macrophage inflammatory protein-1alpha and monocyte chemoattractant protein-1 in glioma-infiltrating microglia: involvement of ATP and P2X receptor. J Neurosci Res 89:199–211
Risinger AL, Giles FJ, Mooberry SL (2009) Microtubule dynamics as a target in oncology. Cancer Treat Rev 35:255–261
Finkelstein Y, Aks SE, Hutson JR, Juurlink DN, Nguyen P, Dubnov-Raz G, Pollak U, Koren G, Bentur Y (2010) Colchicine poisoning: the dark side of an ancient drug. Clin Toxicol 48:407–414
Nacoulma AP, Megalizzi V, Pottier LR, De Lorenzi M, Thoret S, Dubois J, Vandeputte OM, Duez P, Vereecke D, El Jaziri M (2013) Potent antiproliferative cembrenoids accumulate in tobacco upon infection with Rhodococcus fascians and trigger unusual microtubule dynamics in human glioblastoma cells. PLoS One 8:e77529
Larocque K, Ovadje P, Djurdjevic S, Mehdi M, Green J, Pandey S (2014) Novel analogue of colchicine induces selective pro-death autophagy and necrosis in human cancer cells. PLoS One 9:e87064
Van Meir EG, Kikuchi T, Tada M, Li H, Diserens AC, Wojcik BE, Huang HJ, Friedmann T, de Tribolet N, Cavenee WK (1994) Analysis of the p53 gene and its expression in human glioblastoma cells. Cancer Res 54:649–652
Lee HS, Daniels BH, Salas E, Bollen AW, Debnath J, Margeta M (2012) Clinical utility of LC3 and p62 immunohistochemistry in diagnosis of drug-induced autophagic vacuolar myopathies: a case-control study. PLoS One 7:e36221
Ju JS, Varadhachary AS, Miller SE, Weihl CC (2010) Quantitation of “autophagic flux” in mature skeletal muscle. Autophagy 6:929–935
Scherz-Shouval R, Elazar Z (2007) ROS, mitochondria and the regulation of autophagy. Trends Cell Biol 17:422–427
Espert L, Denizot M, Grimaldi M, Robert-Hebmann V, Gay B, Varbanov M, Codogno P, Biard-Piechaczyk M (2006) Autophagy is involved in T cell death after binding of HIV-1 envelope proteins to CXCR4. J Clin Investig 116:2161–2172
Ravikumar B, Berger Z, Vacher C, O’Kane CJ, Rubinsztein DC (2006) Rapamycin pre-treatment protects against apoptosis. Hum Mol Genet 15:1209–1216
Acknowledgments
We thank Ms. Chia-Hsin Ho for cell culture assistance. The study was supported in part by Ministry of Science and Technology of Taiwan (NSC 99-2628-B-006-030-MY3) and NCKU Aim for the Top University Project (D104-38006).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fang, KM., Liu, JJ., Li, CC. et al. Colchicine derivative as a potential anti-glioma compound. J Neurooncol 124, 403–412 (2015). https://doi.org/10.1007/s11060-015-1874-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-015-1874-2