Abstract
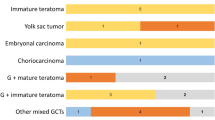
Human chorionic gonadotropin (hCG) production has been utilized as a diagnostic marker for germinoma with syncytiotrophoblastic giant cells (STGC) and choriocarcinoma. Elevated hCG in germinoma is considered to predict less favorable prognosis, and an intensive treatment strategy may accordingly be applied. However, there is some evidence that any germinoma may produce hCG to varying extent. We investigated mRNA expression of the hCG β subunit (hCGβ) using real time quantitative polymerase chain reaction in 94 germ cell tumors (GCTs). Most (93.3 %) GCTs showed higher expression levels compared with that of normal brain tissue (1.09 × 100–1.40 × 105 fold). The expression was the highest in GCTs which harbor choriocarcinoma or STGC components. The expression level of hCGβ in germinoma was highly variable (1.09 × 100–5.88 × 104 fold) in linear but not bimodal distribution. hCG concentrations in serum and CSF correlated with gene expression, especially when GCTs with single histological component were analyzed separately. The expression was not significantly associated with recurrence in pure germinoma. These results suggest that the serum/CSF hCG levels may need to be interpreted with caution as most GCTs appear to have the capacity of producing hCG irrespective of their histology. The clinical significance of ubiquitous hCG expression in GCTs needs further investigation.




Similar content being viewed by others
References
Shibui S (2014) Report of the brain tumor registry of Japan, 2001–2004
Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, Burger PC, Jouvet A, Scheithauer BW, Kleihues P (2007) The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol 114:97–109
Göbel U, Schneider DT, Calaminus G, Haas RJ, Schmidt P, Harms D (2000) Germ-cell tumors in childhood and adolescence. GPOH MAKEI and the MAHO study groups. Ann Oncol: Off J Eur Soc Med Oncol/ESMO 11:263–271
Inamura T, Nishio S, Ikezaki K, Fukui M (1999) Human chorionic gonadotrophin in CSF, not serum, predicts outcome in germinoma. J Neurol Neurosurg Psychiat 66:654–657
Utsuki S, Kawano N, Oka H, Tanaka T, Suwa T, Fujii K (1999) Cerebral germinoma with syncytiotrophoblastic giant cells: feasibility of predicting prognosis using the serum hCG level. Acta Neurochir 141:975 discussion 977-978
Ogino H, Shibamoto Y, Takanaka T, Suzuki K, Ishihara S, Yamada T, Sugie C, Nomoto Y, Mimura M (2005) CNS germinoma with elevated serum human chorionic gonadotropin level: clinical characteristics and treatment outcome. Int J Radiat Oncol Biol Phys 62:803–808. doi:10.1016/j.ijrobp.2004.10.026
Shibamoto Y, Takahashi M, Sasai K (1997) Prognosis of intracranial germinoma with syncytiotrophoblastic giant cells treated by radiation therapy. Int J Radiat Oncol Biol Phys 37:505–510
Matsutani M, Sano K, Takakura K, Fujimaki T, Nakamura O, Funata N, Seto T (1997) Primary intracranial germ cell tumors: a clinical analysis of 153 histologically verified cases. J Neurosurg 86:446–455. doi:10.3171/jns.1997.86.3.0446
Sawamura Y, Ikeda J, Shirato H, Tada M, Abe H (1998) Germ cell tumours of the central nervous system: treatment consideration based on 111 cases and their long-term clinical outcomes. Eur J Cancer 34:104–110
Fujimaki T, Matsutani M, Nishikawa R, Mishima K, Suzuki T, Katakami H (2011) All germinomas are capable of producing hCG-beta and might be treated in the same protocol. Neuro-oncology 13:I13
Katakami H, Hashida S, Yamaguchi H, Yazawa S, Nakano S, Wakisaka S, Matsutani M (2003) Diagnosis and follow-up of CNS germ cell tumors using an ultrasensitive assay of HCG-beta. Horm Clin 51:196–206
Utsuki S, Oka H, Tanizaki Y, Kondo K, Kawano N, Fujii K (2005) Pathological features of intracranial germinomas with reference to fibrous tissue and granulomatous change. Brain Tumor Pathol 22:9–13. doi:10.1007/s10014-004-0171-0
Zeng N, Liu L, McCabe MG, Jones DT, Ichimura K, Collins VP (2009) Real-time quantitative polymerase chain reaction (qPCR) analysis with fluorescence resonance energy transfer (FRET) probes reveals differential expression of the four ERBB4 juxtamembrane region variants between medulloblastoma and pilocytic astrocytoma. Neuropathol Appl Neurobiol 35:353–366. doi:10.1111/j.1365-2990.2008.01001.x
Fukushima S, Otsuka A, Suzuki T, Yanagisawa T, Mishima K, Mukasa A, Saito N, Kumabe T, Kanamori M, Tominaga T, Narita Y, Shibui S, Kato M, Shibata T, Matsutani M, Nishikawa R, Ichimura K (2014) Mutually exclusive mutations of KIT and RAS are associated with KIT mRNA expression and chromosomal instability in primary intracranial pure germinomas. Acta Neuropathol. doi:10.1007/s00401-014-1247-5
Allred DC, Harvey JM, Berardo M, Clark GM (1998) Prognostic and predictive factors in breast cancer by immunohistochemical analysis. Modern Pathol: An Off J United States and Can Acad of Pathol Inc 11:155–168
Mohsin SK, Weiss H, Havighurst T, Clark GM, Berardo M, le Roanh D, To TV, Qian Z, Love RR, Allred DC (2004) Progesterone receptor by immunohistochemistry and clinical outcome in breast cancer: a validation study. Modern Pathol: An Off J United States and Can Acad Pathol Inc 17:1545–1554. doi:10.1038/modpathol.3800229
Bjornsson J, Scheithauer BW, Okazaki H, Leech RW (1985) Intracranial germ cell tumors: pathobiological and immunohistochemical aspects of 70 cases. J Neuropathol Exp Neurol 44:32–46
Ikura Y, Sasaki M, Ohgami M, Ikebe T, Gotoh K, Bettoh H, Sakurai M (1996) Mixed germ-cell tumor of the brain. Pathologic study of six autopsy cases. Pathol Res Ractice 192:595–603. doi:10.1016/S0344-0338(96)80111-7
Tamaki N, Lin T, Shirataki K, Hosoda K, Kurata H, Matsumoto S, Ito H (1990) Germ cell tumors of the thalamus and the basal ganglia. Child’s Nerv Syst: ChNS: Off J Int Soc Pediatr Neurosurg 6:3–7
Teilum G (1965) Classification of endodermal sinus tumour (mesoblatoma vitellinum) and so-called “embryonal carcinoma” of the ovary. Acta Pathologica et Microbiologica Scandinavica 64:407–429
Sano K (1999) Pathogenesis of intracranial germ cell tumors reconsidered. J Neurosurg 90:258–264. doi:10.3171/jns.1999.90.2.0258
Yamagami T, Handa H, Yamashita J, Okumura T, Paine J, Haebara H, Furukawa F (1987) An immunohistochemical study of intracranial germ cell tumours. Acta Neurochir 86:33–41
Tan C, Scotting PJ (2013) Stem cell research points the way to the cell of origin for intracranial germ cell tumours. J Pathol 229:4–11. doi:10.1002/path.4098
Wong JM, Chi SN, Marcus KJ, Levine BS, Ullrich NJ, MacDonald S, Lechpammer M, Goumnerova LC (2010) Germinoma with malignant transformation to nongerminomatous germ cell tumor. J Neurosurg Pediatr 6:295–298. doi:10.3171/2010.6.PEDS09541
Mostofi FK (1973) Tumors of the male genital system. Armed Forces Institute of Pathology, Washington, D.C.
Madersbacher S, Kratzik C, Gerth R, Dirnhofer S, Berger P (1994) Human chorionic gonadotropin (hCG) and its free subunits in hydrocele fluids and neoplastic tissue of testicular cancer patients: insights into the in vivo hCG-secretion pattern. Cancer Res 54:5096–5100
Irie N, Weinberger L, Tang WW, Kobayashi T, Viukov S, Manor YS, Dietmann S, Hanna JH, Surani MA (2015) SOX17 is a critical specifier of human primordial germ cell fate. Cell 160:253–268. doi:10.1016/j.cell.2014.12.013
Louhimo J, Carpelan-Holmstrom M, Alfthan H, Stenman UH, Jarvinen HJ, Haglund C (2002) Serum HCG beta, CA 72-4 and CEA are independent prognostic factors in colorectal cancer. Int J Cancer 101:545–548. doi:10.1002/ijc.90009
Vartiainen J, Lassus H, Lehtovirta P, Finne P, Alfthan H, Butzow R, Stenman UH (2008) Combination of serum hCG beta and p53 tissue expression defines distinct subgroups of serous ovarian carcinoma. Int J Cancer 122:2125–2129. doi:10.1002/ijc.23322
Cole LA (2010) Biological functions of hCG and hCG-related molecules. Reprod Biol Endocrinol: RB&E 8:102. doi:10.1186/1477-7827-8-102
Cole LA, Butler S (2012) Hyperglycosylated hCG, hCGbeta and Hyperglycosylated hCGbeta: interchangeable cancer promoters. Mol Cell Endocrinol 349:232–238. doi:10.1016/j.mce.2011.10.029
Lempiainen A, Sankila A, Hotakainen K, Haglund C, Blomqvist C, Stenman UH (2014) Expression of human chorionic gonadotropin in testicular germ cell tumors. Urol Oncol 32:727–734. doi:10.1016/j.urolonc.2013.11.007
Oosterhuis JW, Stoop H, Honecker F, Looijenga LH (2007) Why human extragonadal germ cell tumours occur in the midline of the body: old concepts, new perspectives. Int J Androl 30:256. discussion 263-254 doi:10.1111/j.1365-2605.2007.00793.x
Jennings MT, Gelman R, Hochberg F (1985) Intracranial germ-cell tumors: natural history and pathogenesis. J Neurosurg 63:155–167. doi:10.3171/jns.1985.63.2.0155
Acknowledgments
This study was supported by a research program of the Project for Development of Innovative Research on Cancer Therapeutics (P-Direct) Ministry of Education, Culture, Sports, Science and Technology (MEXT) of Japan. We thank Dr Hideki Katakami for sharing valuable data and inspiring the project, and Dr Sylvia Kocialkowski for critical reading of the manuscript.
Conflict of interest
The authors declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
On behalf of the Intracranial Germ Cell Tumor Genome Analysis Consortium (the iGCT Consortium).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Takami, H., Fukushima, S., Fukuoka, K. et al. Human chorionic gonadotropin is expressed virtually in all intracranial germ cell tumors. J Neurooncol 124, 23–32 (2015). https://doi.org/10.1007/s11060-015-1809-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-015-1809-y