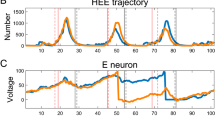
In vivo calcium imaging is widely used in neuroscience to assess the activity of neuronal ensembles. The advent of the single-photon miniature fluorescence microscope (miniscope) has made it possible to use intravital calcium imaging in freely moving animals. Various algorithms and analysis packages have been developed to analyze miniscope data. The present work uses model data with different noise levels as an example to examine the relationship between the accuracy of neuron detection and the values of parameters in Minian, a package for analyzing miniscope data. On the basis of the results obtained, recommendations are given for changing the values of the Minian parameters depending on the noise level in the processed data. The results obtained here provide preliminary guidance for selecting appropriate values for Minian parameters for processing experimental data. The results of this study are expected to be relevant to neuroscientists using intravital calcium imaging in freely moving animals.
Similar content being viewed by others
References
Aharoni, D., Khakh, B. S., Silva, A. J., and Golshani, P., “All the light that we can see: a new era in miniaturized microscopy,” Nat. Methods, 16, No. 1, 11–13 (2019).
Bao, Y., Soltanian-Zadeh, S., Farsiu, S., and Gong, Y., “Segmentation of neurons from fluorescence calcium beyond real time,” Nat. Machine Intell., 3, No. 7, 590–600 (2021).
Barbera, G., Jun, R., Zhang, Y., et al., “A miniature fluorescence microscope for multi-plane imaging,” Sci. Rep., 12, No. 1, 16686 (2022).
Cantu, D. A., Wang, B., Gongwer, M. W., et al., “EZcalcium: Opensource toolbox for analysis of calcium imaging data,” Front. Neural Circuits, 14 (2020).
de Kraker, L., Seignette, K., Thamizharasu, P., et al., “SpecSeg is a versatile toolbox that segments neurons and neurites in chronic calcium imaging datasets based on low-frequency cross-spectral power,” Cell Rep. Methods, 2, No. 10, 100299 (2022).
Denk, W., Strickler, J. H., and Webb, W. W., “Two-photon laser scanning fluorescence microscopy,” Science, 248, No. 4951, 73–76 (1990).
Dong, Z., Mau, W., Feng, Y., et al., “Minian, an open-source miniscope analysis pipeline,” eLife, 11, e70661 (2022).
Engelbrecht, C. J., Johnston, R. S., Seibel, E. J., and Helmchen, F., “Ultracompact fiber-optic two-photon microscope for functional fluorescence imaging in vivo,” Optics Express, 16, No. 8, 5556–5564 (2008).
Erofeev, A. I., Barinov, D. S., Gerasimov, E. I., et al., “NeuroInfoViewer: A software package for analysis of miniscope data,” Neurosci. Behav. Physiol., 51, No. 8, 1199–1205 (2021).
Escobet-Montalban, A., Gasparoli, F. M., Nylk, J., et al., “Three-photon light-sheet fluorescence microscopy,” Opt. Lett., 43, No. 21, 5484–5487 (2018).
Friedrich, J., Giovannucci, A., and Pnevmatikakis, E. A., “Online analysis of microendoscopic 1-photon calcium imaging data streams,” PLoS Comput. Biol., 17, No. 1, e1008565 (2021).
Ghosh, K. K., Burns, L. D., Cocker, E. D., et al., “Miniaturized integration of a fluorescence microscope,” Nat. Methods, 8, No. 10, 871–878 (2011).
Giovannucci, A., Friedrich, J., Gunn, P., Ket al., “CaImAn an open source tool for scalable calcium imaging data analysis,” eLife, 8, e38173 (2019).
Grienberger, C. and Konnerth, A., “Imaging calcium in neurons,” Neuron, 73, No. 5, 862–885 (2012).
Iwasaki, S. and Ikegaya, Y., “In vivo one-photon confocal calcium imaging of neuronal activity from the mouse neocortex,” J. Integr. Neurosci., 17, No. 3–4, 671–678 (2018).
Li, M., Liu, C., Cui, X., et al., “An open-source real-time motion correctionplug-in for single-photon calcium imaging of head-mounted microscopy,” Front. Neural Circuits, 16, (2022).
Liberti, W. A., Perkins, L. N., Leman, D. P., and Gardner, T. J., “An open source, wireless capable miniature microscope system,” J. Neural Eng., 14, No. 4, 045001 (2017).
Lu, J., Li, C., Singh-Alvarado, J., et al., “MIN1PIPE: A Miniscope 1-Photon-Based Calcium Imaging Signal Extraction Pipeline,” Cell Rep., 23, No. 12, 3673–3684 (2018b).
Mukamel, E. A., Nimmerjahn, A., and Schnitzer, M. J., “Automated analysis of cellular signals from large-scale calcium imaging data,” Neuron, 63, No. 6, 747–760 (2009).
Pachitariu, M., Stringer, C., Dipoppa, M., et al., “Suite2p: beyond 10000 neurons with standard two-photon microscopy,” BioRxiv, 061507 (2017).
Pnevmatikakis, E. A. and Giovannucci, A., “NoRMCorre: An online algorithm for piecewise rigid motion correction of calcium imaging data,” J. Neurosci. Methods, 291, 83–94 (2017).
Pnevmatikakis E., A., Soudry, D., Gao, Y., et al., “Simultaneous denoising, deconvolution, demixing of calcium imaging data,” Neuron, 89, No. 2, 285–299 (2016).
Pochechuev, M. S., Fedotov, I. V., Ivashkina, O. I., et al., “Reconnectable fiberscopes for chronic in vivo deep-brain imaging,” J. Biophotonics, 11, No. 4, e201700106 (2018).
Radstake, F. D. W., Raaijmakers, E. A. L., Luttge, R., et al., “CALIMA: The semi-automated open-source calcium imaging analyzer,” Comp. Meth. Progr. Biomed., 179, 104991 (2019).
Resendez, S. L. and Stuber, G. D., “In vivo calcium imaging to illuminate neurocircuit activity dynamics underlying naturalistic behavior,” Neuropsychopharmacology, 40, No. 1, 238–239 (2015).
Robbins, M., Christensen, C., Kaminski, C., and Zlatic, M., “Calcium imaging analysis ? how far have we come? [version 2; peer review: 3 approved],” F1000Res., 10, (258) (2021).
Russell, J. T., “Imaging calcium signals in vivo: a powerful tool in physiology and pharmacology,” Br. J. Pharmacol., 163, No. 8, 1605–1625 (2011).
Sità, L., Brondi, M., Lagomarsino de Leon Roig, P., et al., “A deep-learning approach for online cell identification and trace extraction in functional two-photon calcium imaging,” Nat. Commun., 13, No. 1, 1529 (2022).
ter Veer, M. J. T., Pfeiffer, T., and Nägerl, U. V., “Two-photon STED microscopy for nanoscale imaging of neural morphology in vivo. Super-resolution microscopy,” Methods Mol. Biol., 1663, 45–64 (2017).
Vogt, N., “Two-photon imaging in freely behaving mice,” Nat. Methods, 19, No. 5, 518 (2022).
Wu, X., Yang, X., Song, L., et al., “A modified miniscope system for simultaneous electrophysiology and calcium imaging in vivo,” Front. Integr. Neurosci., 15, (2021).
Zátonyi, A., Madarász, M., Szabó, Á., et al., “Transparent, low-autofluorescence microECoG device for simultaneous Ca2+ imaging and cortical electrophysiology in vivo,” J. Neural Eng., 17, No. 1, 016062 (2020).
Zhou, P., Resendez, S. L., Rodriguez-Romaguera, J., et al., “Efficient and accurate extraction of in vivo calcium signals from microendoscopic video data,” eLife, 7, e28728 (2018).
Zong, W., Obenhaus, H. A., Skytøen, E. R., et al., “Large-scale two-photon calcium imaging in freely moving mice,” Cell, 185, No. 7, 1240–1256.e1230 (2022).
Zong, W., Wu, R., Chen, S., et al., “Miniature two-photon microscopy for enlarged field-of-view, multi-plane and long-term brain imaging,” Nat. Methods, 18, No. 1, 46–49 (2021).
Author information
Authors and Affiliations
Corresponding author
Additional information
A. I. Erofeev and M. V. Petrushan contributed equally to this work and share first authorship.
Translated from Zhurnal Vysshei Nervnoi Deyatel’nosti imeni I. P. Pavlova, Vol. 73, No. 5, pp. 704–723, September–October, 2023.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Erofeev, A.I., Petrushan, M.V., Lysenko, L.V. et al. On Optimizing Miniscope Data Analysis with Simulated Data: A Study of Parameter Optimization in the Minian Analysis Pipeline. Neurosci Behav Physi 54, 251–262 (2024). https://doi.org/10.1007/s11055-024-01593-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11055-024-01593-y