Abstract
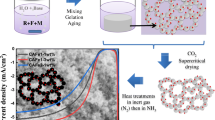
In this work, a process of thermal decomposition of metal carbonyls in oxygen-enriched supercritical carbon dioxide was studied. Manganese, iron, cobalt, and tungsten carbonyls were found to be soluble in supercritical CO2 at relatively mild conditions. For each metal carbonyl studied in this work, the thermal decomposition led to the formation of branched agglomerated structures throughout the high-pressure vessel. More specifically, for manganese and iron carbonyls, the procedure yielded partially monolithic aerogel-like structures, while for cobalt and tungsten carbonyls, the formation of branched aggregates was observed. The morphology of all the obtained metal oxides was studied by means of TEM. The study revealed that all materials consisted of individual nanoparticles with sizes between 2 and 40 nm. Pore structure was investigated by analyzing N2 adsorption/desorption curves. The specific surface area varied greatly depending on the type of oxide from 10 m2/g for CoOx to 130 m2/g for MnOx.








Similar content being viewed by others
References
Bozba SE, Aindow M, Deligöz H, Erkey C (2015) Electrochemical performance of fuel cell catalysts prepared by supercritical deposition : effect of different precursor conversion routes. J Supercrit Fluids 97:154–164
Cangül B, Zhang LC, Aindow M, Erkey C (2009) Preparation of carbon black supported Pd, Pt and Pd–Pt nanoparticles using supercritical CO2 deposition. J Supercrit Fluids 50:82–90. https://doi.org/10.1016/j.supflu.2009.04.001
Chamberlain TW, Zoberbier T, Biskupek J, Botos A, Kaiser U, Khlobystov AN (2012) Formation of uncapped nanometre-sized metal particles by decomposition of metal carbonyls in carbon nanotubes. Chem Sci 3:1919–1924. https://doi.org/10.1039/c2sc01026g
Combes JR, White LD, Tripp CP (1999) Chemical modification of metal oxide surfaces in supercritical CO2: in situ infrared studies of the adsorption and reaction of organosilanes on silica. Langmuir 15:7870–7875. https://doi.org/10.1021/la990495+
Crowley TA, Ziegler KJ, Lyons DM, Erts D, Olin H, Morris MA, Holmes JD (2003) Synthesis of metal and metal oxide nanowire and nanotube arrays within a mesoporous silica template. Chem Mater 15:3518–3522. https://doi.org/10.1021/cm034139v
Davis M, Gümeci C, Kiel C, Hope-Weeks LJ (2011) Preparation of porous manganese oxide nanomaterials by one-pot synthetic sol-gel method. J Sol-Gel Sci Technol 58:535–538. https://doi.org/10.1007/s10971-011-2424-9
Elmanovich IV, Naumkin AV, Gallyamov MO, Khokhlov AR (2012) Organometallic Pt precursor on graphite substrate: deposition from SC CO2, reduction and morphology transformation as revealed by SFM. J Nanopart Res 14:733. https://doi.org/10.1007/s11051-012-0733-8
Ewing AV, Gabrienko AA, Semikolenov SV, Dubkov KA, Kazarian SG (2015) How do intermolecular interactions affect swelling of polyketones with a differing number of carbonyl groups? An in situ ATR-FTIR spectroscopic study of CO2 sorption in polymers. J Phys Chem C 119:431–440. https://doi.org/10.1021/jp510208e
Fillman LM, Tang SC (1984) Thermal decomposition of metal carbonyls: a thermogravimetry-mass spectrometry study. Thermochim Acta 75:71–84. https://doi.org/10.1016/0040-6031(84)85008-X
Gash AE, Tillotson TM, Satcher JH et al (2001a) New sol - gel synthetic route to transition and main-group metal oxide aerogels using inorganic salt precursors. J Non-Cryst Solids 285:22–28. https://doi.org/10.1016/S0022-3093(01)00427-6
Gash AE, Tillotson TM, Satcher JH et al (2001b) Use of epoxides in the sol−gel synthesis of porous Iron(III) oxide monoliths from Fe(III) salts. Chem Mater 13:999–1007. https://doi.org/10.1021/cm0007611
Girard E, Tassaing T, Marty JD, Destarac M (2016) Structure-property relationships in CO2-philic (CO)polymers: phase behavior, self-assembly, and stabilization of water/CO2 emulsions. Chem Rev 116:4125–4169. https://doi.org/10.1021/acs.chemrev.5b00420
Gougousi T, Barua D, Young ED, Parsons GN (2005) Metal oxide thin films deposited from metal organic precursors in supercritical CO 2 solutions. Chem Mater 17:5093–5100. https://doi.org/10.1021/cm0510965
Gregg SJ, Sing KSW (1982) Adsorption, surface area and porosity, second edi. Academic Press, London
Gupta MK, Nigam AK (1972) X ray K absorption edge of cobalt in some of its compounds. J Phys F Met Phys 5:1251–1255. https://doi.org/10.1088/0022-3700/5/9/022
Hao E, Schatz GC, Hupp JT (2004) Synthesis and optical properties of anisotropic metal nanoparticles. J Fluoresc 14:331–341. https://doi.org/10.1023/B:JOFL.0000031815.71450.74
Kachala VV, Khemchyan LL, Kashin AS, Orlov NV, Grachev AA, Zalesskiy SS, Ananikov VP (2013) Target-oriented analysis of gaseous, liquid and solid chemical systems by mass spectrometry, nuclear magnetic resonance spectroscopy and electron microscopy. Russ Chem Rev 82:648–685. https://doi.org/10.1070/RC2013v082n07ABEH004413
Krämer J, Redel E, Thomann R, Janiak C (2008) Use of ionic liquids for the synthesis of Fe , Ru and Os nanoparticles from their metal carbonyl precursors. Organometallics 3:1976–1978. https://doi.org/10.1021/om800056z
Mos YM, Vermeulen AC, Buisman CJN, Weijma J (2018) X-ray diffraction of iron containing samples: the importance of a suitable configuration. Geomicrobiol J 35:511–517. https://doi.org/10.1080/01490451.2017.1401183
Nelson MR, Borkman RF (1998) Ab initio calculations on CO2 binding to carbonyl groups. J Phys Chem A 102:7860–7863. https://doi.org/10.1021/jp981824u
Nikam AV, Prasad BLV, Kulkarni AA (2018) Wet chemical synthesis of metal oxide nanoparticles: a review. CrystEngComm 20:5091–5107. https://doi.org/10.1039/C8CE00487K
Nikolaev AY, Khokhlov AA, Levin EE, Abramchuk SS, Kharitonova EP, Gallyamov MO (2019) Electrochemically active dispersed tungsten oxides obtained from tungsten hexacarbonyl in supercritical carbon dioxide. J Mater Sci 54:9426–9441. https://doi.org/10.1007/s10853-019-03591-9
Poe A (1981) Kinetic studies of thermal reactivities of metal—metal—bonded carbonyls. ACS Symp Ser Am Chem Soc Washington, DC 1981:135–166. https://doi.org/10.1021/bk-1981-0155.ch007
Sing KSW, Williams RT (2004) Physisorption hysteresis loops and the characterization of nanoporous materials. Adsorpt Sci Technol 22:773–782. https://doi.org/10.1260/0263617053499032
Türk M, Erkey C (2018) Synthesis of supported nanoparticles in supercritical fluids by supercritical fluid reactive deposition: current state, further perspectives and needs. J Supercrit Fluids 134:176–183. https://doi.org/10.1016/j.supflu.2017.12.010
Zefirov VV, Elmanovich IV, Levin EE, Abramchuk SS, Kharitonova EP, Khokhlov AA, Kondratenko MS, Gallyamov MO (2018) Synthesis of manganese oxide electrocatalysts in supercritical carbon dioxide. J Mater Sci 53:9449–9462. https://doi.org/10.1007/s10853-018-2242-3
Zefirov VV, Elmanovich IV, Pastukhov AV, Kharitonova EP, Korlyukov AA, Gallyamov MO (2019) Thermal decomposition of manganese carbonyl in supercritical CO2 as a simple and effective approach to obtain manganese oxide aerogels. J Sol-Gel Sci Technol 92:116–123. https://doi.org/10.1007/s10971-019-05092-2
Zhang Y, Erkey C (2006) Preparation of supported metallic nanoparticles using supercritical fluids: a review. J Supercrit Fluids 38:252–267. https://doi.org/10.1016/j.supflu.2006.03.021
Funding
This work was supported by Russian Foundation for Basic Research, grant № 18-29-06036_mk. Electron microscopy characterization was performed in the Department of Structural Studies of Zelinsky Institute of Organic Chemistry, Moscow. This work was supported by Ministry of Science and Higher Education of the Russian Federation. Research contributions of V.V.Z. and V.E.S. were also supported by “Basis” foundation stipendium for PhD students.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declear no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 3980 kb).
Rights and permissions
About this article
Cite this article
Elmanovich, I.V., Zefirov, V.V., Glotov, I.D. et al. Morphology study of metal oxide nanoparticles and aerogels produced via thermal decomposition of metal carbonyls in supercritical carbon dioxide. J Nanopart Res 23, 95 (2021). https://doi.org/10.1007/s11051-021-05138-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-021-05138-z