Abstract
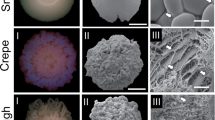
Switching between two cell types in fungi is called phenotypic switching, and it is commonly observed in pathogenic yeast. Candida lusitaniae undergoes antifungal resistance-associated phenotypic switching and results in three colony colors: light brown, brown and dark brown. In this study, we included C. lusitaniae as control. This study had two objectives. First, we wanted to evaluate whether also a prevalent human pathogen C. guilliermondii can undergo phenotypic switching. Second, our aim was to determine whether switching can change yeasts susceptibility to antifungals. Yeast suspension (1 × 103–5 × 103 c.f.u./ml) was plated on the YPD medium containing 1 mM CuSO4. Colonies exhibiting the original and variant phenotypes were counted and converted to percentage of the population. Minimum inhibitory concentrations of amphotericin B, formic acid and acetic acid for the cells from random colonies of the different phenotypes were determined by microdilution method. After 5 days of incubation, C. guilliermondii switched spontaneously and reversibly among two phenotypes distinguishable on CuSO4 containing agar, white and dark brown. Phenotypes occurred with greater frequency (10−1–10−2) than spontaneous mutations and were reversible, fulfilling the two phenotypic switching criteria. The study showed that phenotypic switching was associated with filamentation and affected antifungal resistance. Resistance to amphotericin B increased tenfold and was associated with C. lusitaniae dark brown phenotype. C. guilliermondii colonies with brown phenotype displayed 20 and 2 times higher resistance to amphotericin B and acetic acid, respectively.




Similar content being viewed by others
References
Pfaller MA, Diekema DJ. Epidemiology of invasive candidiasis: a persistent public health problem. Clin Microbiol Rev. 2007;20:133–63.
Pemán J, Salavert M. General epidemiology of invasive fungal disease. Enferm Infecc Microbiol Clin. 2012;30:90–8.
van der Woude MW. Re-examining the role and random nature of phase variation. FEMS Microbiol Lett. 2006;254:190–7.
Jain N, Hasan F, Fries BC. Phenotypic switching in fungi. Curr Fungal Infect Rep. 2008;2:180–8.
Soll DR. Candida commensalism and virulence: the evolution of phenotypic plasticity. Acta Trop. 2002;81:101–10.
Kolotila MP, Diamond RD. Effects of neutrophils and in vitro oxidants on survival and phenotypic switching of Candida albicans WO-1. Infect Immun. 1990;58:1174–9.
Lan CY, Newport G, Murillo LA, Jones T, Scherer S, Davis RW, Agabian N. Metabolic specialization associated with phenotypic switching in Candida albicans. Proc Natl Acad Sci USA. 2002;99:14907–12.
Favela A, Michel-Nguyenb A, Peyron F, Martin C, Thomachot L, Datry A, Bouchara JP, Challier S, Noel T, et al. Colony morphology switching of Candida lusitaniae and acquisition of multidrug resistance during treatment of a renal infection in a newborn: case report and review of the literature. Diagn Microbiol Infect Dis. 2003;47:331–9.
Pfaller MA. Antifungal drug resistance: mechanisms, epidemiology, and consequences for treatment. Am J Med. 2012;125(Suppl. 1):3–13.
Slutsky B, Staebell M, Anderson J, Risen L, Pfaller M, Soll DR. “White-opaque transition”: a second high-frequency switching system in Candida albicans. J Bacteriol. 1987;169:189–97.
Pujol C, Daniels KJ, Lockhart SR, Srikantha T, Radke JB, Geiger J, Soll DR. The closely related species Candida albicans and Candida dubliniensis can mate. Eukaryot Cell. 2004;3:1015–27.
Porman AM, Alby K, Hirakawa MP, Bennett RJ. Discovery of a phenotypic switch regulating sexual mating in the opportunistic fungal pathogen Candida tropicalis. Proc Natl Acad Sci USA. 2011;108:21158–63.
Lachke SA, Joly S, Daniels KJ, Soll DR. Phenotypic switching and filamentation in Candida glabrata. Microbiology. 2002;148:2661–74.
Brockert PJ, Lachke SA, Srikantha T, Pujol C, Galask R, Soll DR. Phenotypic switching and mating type switching of Candida glabrata at sites of colonization. Infect Immun. 2003;71:7109–18.
Miller NS, Dick JD, Merz WG. Phenotypic switching in Candida lusitaniae on copper sulfate indicator agar: association with amphotericin B resistance and filamentation. J Clin Microbiol. 2006;44:1536–9.
Lastauskienė E, Zinkevičienė A, Girkontaitė I, Kaunietis A, Kvedarienė V. Formic acid and acetic acid induce a programmed cell death in pathogenic Candida species. Curr Microbiol. 2014;69:303–10.
Zinkevičienė A, Vaičiulionienė N, Baranauskienė I, Kvedarienė V, Ėmužytė R, Čitavičius D. Cutaneous yeast microflora in patients with atopic dermatitis. Cent Eur J Med. 2011;6:713–9.
Lachke SA, Srikantha T, Tsai LK, Daniels K, Soll DR. Phenotypic switching in Candida glabrata involves phase-specific regulation of the metallothionein gene MT-11 and the newly discovered hemolysin gene HLP. Infect Immun. 2000;68:884–95.
Williamson EM. Synergy and other interactions in phytomedicines. Phytomedicine. 2001;8:401–9.
Thompson DS, Carlisle PL, Kadosh D. Coevolution of morphology and virulence in Candida spiecies. Eukaryot Cell. 2011;10:1173–82.
Brand A. Hyphal growth in human fungal pathogens and its role in virulence. Int J Microbiol. 2012;. doi:10.1155/2012/517529.
Lo HJ, Köhler JR, DiDomenico D, Loebenberg D, Cacciapuoti A, Fink GR. Nonfilamentous C. albicans mutants are avirulent. Cell. 1997;90:939–49.
Pfaller MA, Diekema DJ, Mendez M, Kibbler C, Erzsebet P, Chang SC, Gibbs DL, Newell VA, The Global Antifungal Surveillance Group Candida guilliermondii. An opportunistic fungal pathogen with decreased susceptibility to fluconazole: geographic and temporal trends from the ARTEMIS DISK antifungal surveillance program. J Clin Microbiol. 2006;44:3551–6.
Pfaller MA, Diekema DJ, Messer SA, Boyken L, Hollis RJ, Jones RN. In vitro susceptibilities of rare Candida bloodstream isolates to ravuconazole and three comparative antifungal agents. Diagn Microbiol Infect Dis. 2004;48:101–5.
Vargas K, Messer SA, Pfaller M, Lockhart SR, Stapleton JT, Hellstein J, Soll DR. Elevated phenotypic switching and drug resistance of Candida albicans from human immunodeficiency virus-positive individuals prior to first thrush episode. J Clin Microbiol. 2000;38:3595–607.
Yoon SA, Vazques JA, Steffan PE, Sobel JD, Akins RA. High-frequency, in vitro reversible switching of Candida lusitaniae clinical isolates from amphotericin B susceptibility to resistance. Antimicrob Agents Chemother. 1999;43:836–45.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lastauskienė, E., Čeputytė, J., Girkontaitė, I. et al. Phenotypic Switching of Candida guilliermondii is Associated with Pseudohyphae Formation and Antifungal Resistance. Mycopathologia 179, 205–211 (2015). https://doi.org/10.1007/s11046-014-9844-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11046-014-9844-3