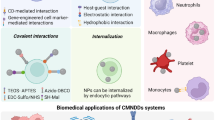
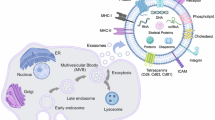
Abstract
Renal cell carcinoma (RCC) is the most common type of kidney cancer that accounts for approximately 2–3% of adult malignancies. Among the primary treatment methods for this type of cancer are surgery and targeted treatment. Still, due to less than optimal effectiveness, there are problems such as advanced distant metastasis, delayed diagnosis, and drug resistance that continue to plague patients. In recent years, therapeutic advances have increased life expectancy and effective treatment in renal cell carcinoma patients. One of these methods is the use of stem cells. Although the therapeutic effects of stem cells, especially mesenchymal stem cells, are still impressive, today, extracellular vesicles (EVs) as carrying molecules and various mediators in intercellular communications, having a central role in tumorigenesis, metastasis, immune evasion, and drug response, and on the other hand, due to its low immunogenicity and strong regulatory properties of the immune system, has received much attention from researchers and doctors. Despite the increasing interest in exosomes as the most versatile type of EVs, the heterogeneity of their efficacy presents challenges and, on the other hand, exciting opportunities for diagnostic and clinical interventions.
In the upcoming article, we will review the various aspects of exosomes’ effects in the prevention, treatment, and progress of renal cell carcinoma and also ways to optimize them to strengthen their positive sides.
Graphical Abstract




Similar content being viewed by others
Data availability
No datasets were generated or analysed during the current study.
References
Zhang Y et al (2021) Single-cell analyses of renal cell cancers reveal insights into tumor microenvironment, cell of origin, and therapy response. Proc Natl Acad Sci 118(24):e2103240118
Kim M et al (2020) Comprehensive immunoprofiles of renal cell carcinoma subtypes. Cancers 12(3):602
Malyszko J et al (2020) The link between kidney disease and cancer: complications and treatment. Lancet 396(10246):277–287
Bukavina L et al (2022) Epidemiology of renal cell carcinoma: 2022 update. Eur Urol 82(5):529–542
Larroquette M et al (2022) Therapeutic management of metastatic Clear Cell Renal Cell Carcinoma: A Revolution in every decade. Cancers 14(24):6230
Ballesteros PÁ et al (2021) Molecular mechanisms of resistance to immunotherapy and antiangiogenic treatments in clear cell renal cell carcinoma. Cancers 13(23):5981
Scholz H et al (2021) Kidney physiology and susceptibility to acute kidney injury: implications for renoprotection. Nat Rev Nephrol 17(5):335–349
Thurlow JS et al (2021) Global epidemiology of end-stage kidney disease and disparities in kidney replacement therapy. Am J Nephrol 52(2):98–107
Jalali F, Nassiri A, Hakemi MS (2021) Awareness and knowledge about kidney transplantation: a reflection on the current state among Iranian patients with end-stage renal disease (ESRD) treated by Dialysis. Iran J Kidney Dis, 15(6)
De Pasquale C et al (2020) Psychological and psychopathological aspects of kidney transplantation: a systematic review. Front Psychiatry 11:106
Ammirati A (2020) Chronic Kidney Disease 66:s03–s09
Padala SA et al (2020) Epidemiology of renal cell carcinoma. World J Oncol 11(3):79–87
Ljungberg B et al (2022) European Association of Urology guidelines on renal cell carcinoma: the 2022 update European urology
Rassy E et al (2020) New rising entities in cancer of unknown primary: is there a real therapeutic benefit? Crit Rev Oncol/Hematol 147:102882
Li J et al (2020) CircTLK1 promotes the proliferation and metastasis of renal cell carcinoma by sponging miR-136-5p. Mol Cancer 19:1–17
Huang WC et al (2020) Chronic kidney disease and kidney cancer surgery: new perspectives. J Urol 203(3):475–485
Díaz-Montero CM, Rini BI, Finke JH (2020) The immunology of renal cell carcinoma. Nat Rev Nephrol 16(12):721–735
Brown JE et al (2023) Temporary treatment cessation versus continuation of first-line tyrosine kinase inhibitor in patients with advanced clear cell renal cell carcinoma (STAR): an open-label, non-inferiority, randomised, controlled, phase 2/3 trial. Lancet Oncol 24(3):213–227
Savaliya M et al (2023) Posterior reversible Encephalopathy Syndrome after Pazopanib Therapy. Diseases 11(2):76
Kamli H, Li L, Gobe GC (2019) Limitations to the therapeutic potential of tyrosine kinase inhibitors and alternative therapies for kidney cancer. Ochsner J 19(2):138–151
Braun DA et al (2021) Beyond conventional immune-checkpoint inhibition—novel immunotherapies for renal cell carcinoma. Nat Reviews Clin Oncol 18(4):199–214
Mierzejewski B et al (2020) Human and mouse skeletal muscle stem and progenitor cells in health and disease. Seminars in cell & developmental biology. Elsevier
Rajabzadeh N, Fathi E, Farahzadi R (2019) Stem cell-based regenerative medicine. Stem cell Invest, 6
Franco ML, Beyerstedt S, Rangel ÉB (2021) Klotho and mesenchymal stem cells: a review on cell and gene therapy for chronic kidney disease and acute kidney disease. Pharmaceutics 14(1):11
Changizi-Ashtiyani S et al (2020) The effect of adipose-derived mesenchymal stem cells on renal function and histopathology in a rat model of ischemia-reperfusion induced acute kidney injury. Iran J Basic Med Sci 23(8):999
Liu D et al (2020) Stem cells: a potential treatment option for kidney diseases. Stem Cell Res Ther 11(1):1–20
Changizi-Ashtiyani S et al (2020) The effect of adipose-derived mesenchymal stem cells on renal function and histopathology in a rat model of ischemia-reperfusion induced acute kidney injury. 23(8):999
Fan X-L et al (2020) Mechanisms underlying the protective effects of mesenchymal stem cell-based therapy. Cell Mol Life Sci 77:2771–2794
Fu Z et al (2023) Optimization strategies of mesenchymal stem cell-based therapy for acute kidney injury. Stem Cell Res Ther 14(1):1–16
Li S et al (2021) Enhanced renoprotective effect of GDNF-modified adipose-derived mesenchymal stem cells on renal interstitial fibrosis. Stem Cell Res Ther 12(1):1–17
Hafazeh L et al (2019) Stem cell therapy ameliorates ischemia-reperfusion induced kidney injury after 24 hours reperfusion. Iran J Kidney Dis 13(6):380
Bochon B et al (2019) Mesenchymal stem cells—potential applications in kidney diseases. 20(10):2462
Hussen BM et al (2022) Strategies to overcome the main challenges of the use of exosomes as drug carrier for cancer therapy. Cancer Cell Int 22(1):1–23
Sidhom K, Obi PO, Saleem A (2020) A review of exosomal isolation methods: is size exclusion chromatography the best option? Int J Mol Sci 21(18):6466
Altyar AE et al (2023) Future regenerative medicine developments and their therapeutic applications. Biomed Pharmacother 158:114131
Gurung S et al (2021) The exosome journey: from biogenesis to uptake and intracellular signalling. Cell Communication Signal 19(1):1–19
Tschuschke M et al (2020) Inclusion biogenesis, methods of isolation and clinical application of human cellular exosomes. 9(2):436
Gonda A et al (2020) Cellular-defined microenvironmental internalization of exosomes Extracellular Vesicles and Their Importance in Human Health, : p. 1–30
Gurunathan S et al (2019) Review of the isolation, characterization, biological function, and multifarious therapeutic approaches of exosomes. 8(4):307
Amini H et al (2021) An examination of the putative role of melatonin in exosome biogenesis. Front Cell Dev Biology 9:686551
Frankel E, Audhya A (2018) ESCRT-dependent cargo sorting at multivesicular endosomes. In seminars in cell & developmental biology. Elsevier
Momen LT et al (2021) Regeneration and diagnosis of kidney Disease using Exosomes. Jentashapir J Cell Mol Biology, 12(4)
Xie JX et al (2017) MicroRNA profiling in kidney disease: plasma versus plasma-derived exosomes. Gene 627:1–8
Stefańska K et al (2023) The role of exosomes in Human Carcinogenesis and Cancer Therapy—recent findings from Molecular and Clinical Research. 12(3):356
Zhang C et al (2018) Exosome: function and role in cancer metastasis and drug resistance. Technol Cancer Res Treat 17:1533033818763450
Nam GH et al (2020) Emerging prospects of exosomes for cancer treatment: from conventional therapy to immunotherapy. Adv Mater 32(51):2002440
Hiltbrunner S et al (2016) Exosomal cancer immunotherapy is independent of MHC molecules on exosomes. Oncotarget 7(25):38707
Pitt JM et al (2016) Dendritic cell–derived exosomes for cancer therapy. J Clin Investig 126(4):1224–1232
Xu Y et al (2022) Role of macrophages in tumor progression and therapy. Int J Oncol 60(5):1–19
Paskeh MDA et al (2022) Emerging role of exosomes in cancer progression and tumor microenvironment remodeling. J Hematol Oncol 15(1):1–39
Mantovani A et al (2022) Macrophages as tools and targets in cancer therapy : p. 1–22
Ding X et al (2022) Eng Macrophages via Nanatechnol Genetic Manipulation cancer Therapy 11:786913
Yunna C et al (2020) Macrophage M1/M2 polarization. Eur J Pharmacol 877:173090
Wang H et al (2021) The impact of the tumor microenvironment on macrophage polarization in cancer metastatic progression. Int J Mol Sci 22(12):6560
Cassetta L, Pollard JW (2020) Tumor-associated macrophages. Curr Biol 30(6):R246–R248
Zou Z et al (2020) mTOR signaling pathway and mTOR inhibitors in cancer: Progress and challenges. 10(1):1–11
Tang T-T et al (2020) Extracellular vesicle–encapsulated IL-10 as novel nanotherapeutics against ischemic AKI. Sci Adv 6(33):eaaz0748
Ben-Sahra I, Manning BD (2017) mTORC1 signaling and the metabolic control of cell growth. Curr Opin Cell Biol 45:72–82
Collins SL et al (2021) mTORC1 signaling regulates proinflammatory macrophage function and metabolism. J Immunol 207(3):913–922
Novais AA et al (2021) Exosomes and melatonin: where their destinies intersect. Front Immunol 12:692022
Maleki M et al (2021) Multiple interactions between melatonin and non-coding RNAs in cancer biology, vol 98. Chemical Biology & Drug Design, pp 323–340. 3
Jin C et al (2021) Exosomes: emerging therapy delivery tools and biomarkers for kidney diseases 2021
Jin C et al (2021) Exosomes: emerging therapy delivery tools and biomarkers for kidney diseases Stem cells international, 2021
Tapparo M et al (2019) Renal regenerative potential of extracellular vesicles derived from miRNA-engineered mesenchymal stromal cells. 20(10):2381
Zhang C et al (2020) Supramolecular nanofibers containing arginine-glycine-aspartate (RGD) peptides boost therapeutic efficacy of extracellular vesicles in kidney repair. ACS Nano 14(9):12133–12147
Liao C et al (2022) Potential Therapeutic Effect Mech Mesenchymal stem cells-extracellular Vesicles Ren Fibros 10
Zhang W et al (2018) MicroRNAs in serum exosomes as potential biomarkers in clear-cell renal cell carcinoma. 4(3):412–419
Fang P et al (2020) Targeting strategies for renal cancer stem cell therapy. Curr Pharm Design 26(17):1964–1978
He X et al (2022) Circulating exosomal mRNA signatures for the early diagnosis of clear cell renal cell carcinoma. 20(1):1–13
Sun IO, Lerman LO (2020) Urinary extracellular vesicles as biomarkers of kidney disease: from diagnostics to therapeutics. Diagnostics 10(5):311
Chatterjee N, Bivona TG (2019) Polytherapy and targeted cancer drug resistance. Trends cancer 5(3):170–182
Barton S et al (2023) miR-23b-3p regulates human endometrial epithelial cell adhesion implying a role in implantation. Reproduction 165(4):407–416
Guo Y-X et al (2021) The role of miR-23b in cancer and autoimmune disease Journal of Oncology, 2021
Xu N et al (2022) Exosomes-mediated tumor treatment: one body plays multiple roles. 17(3):385–400
Zhang Y et al (2010) Exosomes derived from IL-12-anchored renal cancer cells increase induction of specific antitumor response in vitro: a novel vaccine for renal cell carcinoma. Int J Oncol 36(1):133–140
Shi L et al (2021) MicroRNAs in body fluids: a more promising biomarker for clear cell renal cell carcinoma. Cancer Manage Res, : p. 7663–7675
Boussios S et al (2023) Exosomes in the diagnosis and treatment of renal cell Cancer. Int J Mol Sci 24(18):14356
Chung I-M et al (2020) Exosomes: current use and future applications. Clin Chim Acta 500:226–232
Mataki H et al (2015) Downregulation of the microRNA-1/133a cluster enhances cancer cell migration and invasion in lung-squamous cell carcinoma via regulation of Coronin1C. J Hum Genet 60(2):53–61
Yu Z et al (2013) Identification of miR-7 as an oncogene in renal cell carcinoma. J Mol Histol 44(6):669–677
Liu L et al (2019) Long non-coding RNA GAS5 sensitizes renal cell carcinoma to sorafenib via miR-21/SOX5 pathway. Cell Cycle 18(3):257–263
Onyshchenko K et al (2020) Expression of micro-RNA hsa-miR-30c-5p and hsa-mir-138-1 in renal cell carcinoma. Exp Oncol 42:115–119
Liang L et al (2021) Retracted: miR-30d‐5p suppresses proliferation and autophagy by targeting ATG5 in renal cell carcinoma. FEBS open bio 11(2):529–540
Zeng J et al (2020) lncRNA 00312 attenuates cell proliferation and invasion and promotes apoptosis in renal cell carcinoma via miR-34a-5p/ASS1 axis Oxidative medicine and cellular longevity, 2020
Toraih EA et al (2017) MicroRNA-34a: a key regulator in the hallmarks of renal cell carcinoma Oxidative Medicine and Cellular Longevity, 2017
Osako Y et al (2019) Potential tumor–suppressive role of microRNA–99a–3p in sunitinib–resistant renal cell carcinoma cells through the regulation of RRM2. Int J Oncol 54(5):1759–1770
Sun K et al (2019) Long non-coding RNA XIST regulates miR-106b-5p/P21 axis to suppress tumor progression in renal cell carcinoma. Biochem Biophys Res Commun 510(3):416–420
Miao L-J et al (2019) miR-106b promotes proliferation and invasion by targeting Capicua through MAPK signaling in renal carcinoma cancer. OncoTargets Therapy 12:3595
Xu Y et al (2020) miR–133b affects cell proliferation, invasion and chemosensitivity in renal cell carcinoma by inhibiting the ERK signaling pathway. Mol Med Rep 22(1):67–76
Yan C et al (2022) MiR-1294 suppresses ROS-dependent inflammatory response in atopic dermatitis via restraining STAT3/NF-κB pathway. Cell Immunol 371:104452
Liu S et al (2023) HES1-mediated down-regulation of miR-138 sustains NOTCH1 activation and promotes proliferation and invasion in renal cell carcinoma. J Experimental Clin Cancer Res 42(1):1–15
Dasgupta P et al (2020) LncRNA CDKN2B-AS1/miR-141/cyclin D network regulates tumor progression and metastasis of renal cell carcinoma. Cell Death Dis 11(8):660
Wang J et al (2021) SNP-mediated lncRNA-ENTPD3-AS1 upregulation suppresses renal cell carcinoma via miR-155/HIF-1α signaling. Cell Death Dis 12(7):672
Wang X et al (2019) Serum exosomal miR-210 as a potential biomarker for clear cell renal cell carcinoma. J Cell Biochem 120(2):1492–1502
Dias F et al (2017) Plasmatic miR-210, miR-221 and miR-1233 profile: potential liquid biopsies candidates for renal cell carcinoma. Oncotarget 8(61):103315
Toraih EA et al (2022) Applications of noncoding RNAs in renal cancer patients, in clinical applications of non-coding RNAs in Cancer. Elsevier, pp 211–284
Wang Z, Xie W, Guan H (2023) Diverse functions of MiR-425 in Human Cancer. DNA Cell Biol 42(3):113–129
Jia G et al (2019) Mir-590-5p promotes liver cancer growth and chemotherapy resistance through directly targeting FOXO1. Am J Translational Res 11(4):2181
Cheng L et al (2021) Circ_RPL23A acts as a miR-1233 sponge to suppress the progression of clear cell renal cell carcinoma by promoting ACAT2. J Bioenerg Biomembr 53(4):415–428
Kalhori MR et al (2022) The potential role of miR-1290 in cancer progression, diagnosis, prognosis, and treatment: an oncomiR or onco‐suppressor microRNA? J Cell Biochem 123(3):506–531
Anbiyaee O et al (2023) The functions of long non-coding RNA (lncRNA)-MALAT-1 in the pathogenesis of renal cell carcinoma. BMC Nephrol 24(1):380
Bravo-Vázquez LA et al (2024) Exploring the therapeutic significance of microRNAs and lncRNAs in kidney diseases. Genes 15(1):123
Zhou W et al (2023) Yin Yang 1-Induced Long Noncoding RNA DUXAP9 drives the progression of oral squamous cell carcinoma by blocking CDK1‐Mediated EZH2 degradation. Adv Sci 10(25):2207549
Kulkarni P et al (2021) A lncRNA TCL6-miR-155 interaction regulates the src-Akt-EMT network to mediate kidney cancer progression and metastasis. Cancer Res 81(6):1500–1512
Li T et al (2022) Identification of a three-glycolysis-related lncRNA signature correlated with prognosis and metastasis in clear cell renal cell carcinoma. Front Med 8:777507
Xu P et al (2023) LncRNA AGAP2 antisense RNA 1 stabilized by insulin-like growth factor 2 mRNA binding protein 3 promotes macrophage M2 polarization in clear cell renal cell carcinoma through regulation of the microRNA-9-5p/THBS2/PI3K-Akt pathway. Cancer Cell Int 23(1):330
Fu S et al (2021) Prognostic value of long noncoding RNA DLEU2 and its relationship with immune infiltration in kidney renal clear cell carcinoma and liver hepatocellular carcinoma. Int J Gen Med, : p. 8047–8064
Zhou F-J et al (2022) LncRNA LINC00460 facilitates the proliferation and metastasis of renal cell carcinoma via PI3K/AKT signaling pathway. J Cancer 13(9):2844
Zhang D et al (2019) Down-regulation of circular RNA_000926 attenuates renal cell carcinoma progression through miRNA-411–dependent CDH2 inhibition. Am J Pathol 189(12):2469–2486
Luo S et al (2022) Circ_0005875 sponges miR-502-5p to promote renal cell carcinoma progression through upregulating E26 transformation specific-1 Anti-cancer drugs. 33(1):e286–e298
Gong L-J et al (2021) CircESRP1 inhibits clear cell renal cell carcinoma progression through the CTCF-mediated positive feedback loop. Cell Death Dis 12(11):1081
Fang L, Ye T, An Y (2021) Circular RNA FOXP1 induced by ZNF263 upregulates U2AF2 expression to accelerate renal cell carcinoma tumorigenesis and warburg effect through Sponging miR-423-5p Journal of Immunology Research, 2021
Li W et al (2021) Revealing potential lipid biomarkers in clear cell renal cell carcinoma using targeted quantitative lipidomics. Lipids Health Dis 20:1–13
Xu Z et al (2023) Identify AGAP2 as prognostic biomarker in clear cell renal cell carcinoma based on bioinformatics and IHC staining. Heliyon, 9(2)
Song Q et al (2022) Exosomes in urological diseases-biological functions and clinical applications. Cancer Lett, : p. 215809
Song Q et al (2022) Exosomes in urological diseases-Biological functions and clinical applications : p. 215809
Sávio-Silva C et al (2020) Mesenchymal stem cell therapy in acute kidney injury (AKI): review and perspectives. Revista Da Associação Médica Brasileira 66:s45–s54
Yoon YM et al (2020) Melatonin-stimulated exosomes enhance the regenerative potential of chronic kidney disease‐derived mesenchymal stem/stromal cells via cellular prion proteins. J Pineal Res 68(3):e12632
Yoon YM et al (2020) Melatonin-stimulated exosomes enhance the regenerative potential of chronic kidney disease‐derived mesenchymal stem/stromal cells via cellular prion proteins 68(3): p. e12632
Birtwistle L, Chen X-M, Pollock C (2021) Mesenchymal stem cell-derived extracellular vesicles to the rescue of renal injury. Int J Mol Sci 22(12):6596
Gao L et al (2020) Potential targeted therapy and diagnosis based on novel insight into growth factors, receptors, and downstream effectors in acute kidney injury and acute kidney injury-chronic kidney disease progression. 5(1):9
Su T et al (2021) Exosomal MicroRNAs mediating crosstalk between cancer cells with cancer-associated fibroblasts and tumor-associated macrophages in the tumor microenvironment. Front Oncol 11:631703
Baig MS et al (2020) Tumor-derived exosomes in the regulation of macrophage polarization. Inflamm Res 69:435–451
Zhang Wl et al (2020) Extracellular vesicle long non–coding RNA-mediated crosstalk in the tumor microenvironment: tiny molecules, huge roles. Cancer Sci 111(8):2726–2735
Zhang W et al (2022) Renal cell carcinoma-derived exosomes deliver lncARSR to induce macrophage polarization and promote tumor progression via STAT3 pathway. 18(8):3209–3222
Monti M et al (2022) Micro-RNAs predict response to systemic treatments in metastatic renal cell carcinoma patients: results from a systematic review of the literature. Biomedicines 10(6):1287
Peng J et al (2015) let-7b and let-7c are determinants of intrinsic chemoresistance in renal cell carcinoma. 13:1–8
Du M et al (2017) Plasma exosomal miRNAs-based prognosis in metastatic kidney cancer. Oncotarget 8(38):63703
Srivastava A et al (2018) Exosome RNAs as biomarkers and targets for cancer therapy. Diagnostic and therapeutic applications of exosomes in cancer. Elsevier, pp 129–159
Olejarz W et al (2020) Exosomes in angiogenesis and anti-angiogenic therapy in cancers. Int J Mol Sci 21(16):5840
Umezu T et al (2014) Exosomal miR-135b shed from hypoxic multiple myeloma cells enhances angiogenesis by targeting factor-inhibiting HIF-1 blood. J Am Soc Hematol 124(25):3748–3757
Olejarz W et al (2020) Exosomes in angiogenesis and anti-angiogenic therapy in cancers. 21(16):5840
Jahani M et al (2020) Regenerative medicine and angiogenesis; challenges and opportunities. Adv Pharm Bull 10(4):490
Long C et al (2023) Therapeutic potential of exosomes from adipose-derived stem cells in chronic wound healing Application of Stem Cell Therapy and Bioinformatics in Wound Repair and Skin Diseases. 16648714:118
Jahani M et al (2020) Regenerative medicine and angiogenesis; challenges and opportunities. 10(4):490
Kučuk N et al (2021) Exosomes engineering and their roles as therapy delivery tools, therapeutic targets, and biomarkers. Int J Mol Sci 22(17):9543
Chen L et al (2022) Exosomes as drug carriers in anti-cancer therapy. Front Cell Dev Biology 10:34
Chen J et al (2022) Review on strategies and technologies for exosome isolation and purification. Front Bioeng Biotechnol 9:811971
Liga A et al (2015) Exosome isolation: a microfluidic road-map. Lab Chip 15(11):2388–2394
Xu M et al (2020) Recent advancements in the loading and modification of therapeutic exosomes. 8:586130
Ghafouri-Fard S et al (2021) The impact of non-coding RNAs on normal stem cells. Biomed Pharmacother 142:112050
Choi H et al (2021) Biodistribution of exosomes and engineering strategies for targeted delivery of therapeutic exosomes. Tissue Eng Regenerative Med 18(4):499–511
Faruqu FN, Xu L, Al-Jamal KT (2018) Preparation of exosomes for siRNA delivery to cancer cells. JoVE (Journal Visualized Experiments), (142): p. e58814
Jafari D et al (2020) Designer exosomes: a new platform for biotechnology therapeutics. BioDrugs 34:567–586
Aslan C et al (2021) Exosomes for mRNA delivery: a novel biotherapeutic strategy with hurdles and hope. BMC Biotechnol 21:1–12
Yim N et al (2016) Exosome engineering for efficient intracellular delivery of soluble proteins using optically reversible protein–protein interaction module. Nat Commun 7(1):12277
Hadizadeh N et al (2022) Extracellular vesicles biogenesis, isolation, manipulation and genetic engineering for potential in vitro and in vivo therapeutics: an overview. Front Bioeng Biotechnol, 10
Kim J et al (2021) Platform technologies and human cell lines for the production of therapeutic exosomes. Extracell Vesicles Circulating Nucleic Acids 2(1):3–17
Uddin N et al (2022) Targeted delivery of RNAi to cancer cells using RNA-ligand displaying exosome. Acta Pharm Sinica B
Smyth T et al (2014) Surface functionalization of exosomes using click chemistry. Bioconjug Chem 25(10):1777–1784
Kumar S et al (2018) Cloaked exosomes: biocompatible, durable, and degradable encapsulation. Small 14(34):1802052
Nie W et al (2020) Responsive exosome nano-bioconjugates for synergistic cancer therapy. Angew Chem Int Ed 59(5):2018–2022
Johnsen KB et al (2016) Evaluation of electroporation-induced adverse effects on adipose-derived stem cell exosomes. Cytotechnology 68:2125–2138
Kim MS et al (2016) Development of exosome-encapsulated paclitaxel to overcome MDR in cancer cells Nanomedicine: Nanotechnology, Biology and Medicine, 12(3): p. 655–664
Pammi Guru KT, Praween N, Basu PK (2023) Isolation of exosomes from human serum using gold-nanoparticle-coated Silicon Surface. Nanomaterials 13(3):387
Chen H et al (2021) Exosomes, a new star for targeted delivery. Front Cell Dev Biology, : p. 2827
Whiteside TL (2016) J.A.i.c.c. Tumor-derived Exosomes Their role cancer Progression 74:103–141
Mu W, Rana S, Zöller MJN (2013) Host Matrix Modulation Tumor Exosomes Promotes Motil Invasiveness 15(8):875–IN4
Lokody IJNRC (2014) Exosomally derived miR-105 destroys tight junctions. 14(6):386–387
Zhou W et al (2014) Cancer-secreted miR-105 destroys vascular endothelial barriers to promote metastasis. 25(4):501–515
Zhang L (2019) D.J.B.e.B.A.-R.o.C. Yu. Exosomes cancer Dev Metastasis Immun 1871(2):455–468
Wong C-H (2019) And Y.-C.J.W.j.o.c.c. Chen. Clin Significance Exosomes as Potential Biomarkers cancer 7(2):171
Makler A (2020) W.J.E.r.o.m.d. Asghar. Exosomal Biomarkers cancer Diagnosis Patient Monit 20(4):387–400
Funding
The authors declare that no funds or grants were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Houshang Najafi, designed the study and reviewed and edited the manuscript, Mahan Mohammadi contributed to conceptualization, reviewed the literature and wrote the manuscript draft. Kamran Mansouri and Mehran Pournazeri participated in drafting the manuscript and editing. Pantea Mohammadi, contributed to figure designing.All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Ethical approval
This study was approved by the research deputy of Kermanshah University of Medical Sciences, Kermanshah, Iran (Ethics approval ID: IR.KUMS.REC.1401.253).
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mohammadi, M., Mansouri, K., Mohammadi, P. et al. Exosomes in renal cell carcinoma: challenges and opportunities. Mol Biol Rep 51, 443 (2024). https://doi.org/10.1007/s11033-024-09384-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11033-024-09384-x