Abstract
Background
Evidence suggests that enolase-phosphatase 1 (ENOPH1) is involved in the progression of some certain types of cancers and acts as an oncogenic factor in tumor progression. The present study aimed to identify the central role of ENOPH1 in the progression of breast cancer (BC), a highly proliferative and aggressive disease.
Methods and results
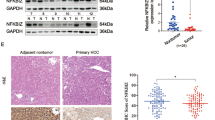
ENOPH1 expression in BC tissues was explored based on the online resource and 40 paired fresh BC and para-carcinoma samples. Functional assays were performed to evaluate the biological effect of ENOPH1 on cell proliferation and migration in ENOPH1-silenced or overexpressing BC cell lines. Blockade of NF-κB by BAY11-7082 was performed to evaluate whether ENOPH1 exerted tumor-promoting properties via regulating the NF-κB signaling pathway. Results of the present study demonstrated that ENOPH1 expression was profoundly upregulated in BC tissues compared with adjacent breast tissues, and ENOPH1 expression was associated with cancer stage, node metastasis status, and overall survival. Functional assays demonstrated that ENOPH1 overexpression significantly accelerated BC cell proliferation, migration, and invasion, while genetic knockdown of ENOPH1 yielded the opposite effects. Mechanistically, ENOPH1 activated the NF-κB pathway, as evidenced by increased expression of NF-κB downstream genes and enhanced NF-κB p65 nuclear translocation. Furthermore, the oncogenic properties of ENOPH1 in proliferation, migration, and invasion were restrained following inhibition of the NF-κB signaling pathway.
Conclusions
These findings indicated the significance of ENOPH1 in promoting cell proliferation and invasion, mainly through activating the NF-κB pathway, suggesting that ENOPH1 might be an attractive prognostic factor and a potential target for BC therapy.





Similar content being viewed by others
Data availability
The data used to support the findings of this study are available from the corresponding author upon request.
References
DeSantis CE, Ma J, Gaudet MM, Newman LA, Miller KD, Goding Sauer A et al (2019) Breast cancer statistics, 2019. CA Cancer J Clin 69:438–451. https://doi.org/10.3322/caac.21583
Bai X, Ni J, Beretov J, Graham P, Li Y (2018) Cancer stem cell in breast cancer therapeutic resistance. Cancer Treat Rev 69:152–163. https://doi.org/10.1016/j.ctrv.2018.07.004
Ngamcherdtrakul W, Yantasee W (2019) siRNA therapeutics for breast cancer: recent efforts in targeting metastasis, drug resistance, and immune evasion. Transl Res 214:105–120. https://doi.org/10.1016/j.trsl.2019.08.005
Karin M (2006) Nuclear factor-kappaB in cancer development and progression. Nature 441:431–436
Suhail M, Tarique M, Muhammad N, Naz H, Hafeez A, Zughaibi TA et al (2021) A critical transcription factor NF-κB as a cancer therapeutic target and its inhibitors as cancer treatment options. Curr Med Chem 28:4117–4132. https://doi.org/10.2174/0929867327666201111142307
Chaturvedi MM, Sung B, Yadav VR, Kannappan R, Aggarwal BB (2011) NF-κB addiction and its role in cancer: ‘one size does not fit all’. Oncogene 30:1615–1630. https://doi.org/10.1038/onc.2010.566
Wang W, Nag SA, Zhang R (2015) Targeting the NFκB signaling pathways for breast cancer prevention and therapy. Curr Med Chem 22:264–289
Khongthong P, Roseweir AK, Edwards J (2019) The NF-KB pathway and endocrine therapy resistance in breast cancer. Endocr Relat Cancer 26:R369–R380. https://doi.org/10.1530/ERC-19-0087
Jiang L, Ren L, Zhang X, Chen H, Chen X, Lin C et al (2019) Overexpression of PIMREG promotes breast cancer aggressiveness via constitutive activation of NF-κB signaling. EBioMedicine 43:188–200. https://doi.org/10.1016/j.ebiom.2019.04.001
Liu W, Yang Y, He B, Ma F, Sun F, Guo M et al (2021) ESM1 promotes triple-negative breast cancer cell proliferation through activating AKT/NF-κB/Cyclin D1 pathway. Ann Transl Med 9:533. https://doi.org/10.21037/atm-20-7005
Qu J, Li J, Zhang Y, He R, Liu X, Gong K et al (2021) AKR1B10 promotes breast cancer cell proliferation and migration via the PI3K/AKT/NF-κB signaling pathway. Cell Biosci 11:163. https://doi.org/10.1186/s13578-021-00677-3
Zhang L, Song L, Xu Y, Xu Y, Zheng M, Zhang P et al (2022) Midkine promotes breast cancer cell proliferation and migration by upregulating NR3C1 expression and activating the NF-κB pathway. Mol Biol Rep 49:2953–2961. https://doi.org/10.1007/s11033-022-07116-7
Luo M, Hou L, Li J, Shao S, Huang S, Meng D et al (2016) VEGF/NRP-1axis promotes progression of breast cancer via enhancement of epithelial-mesenchymal transition and activation of NF-κB and β-catenin. Cancer Lett 373. https://doi.org/10.1016/j.canlet.2016.01.010
Bhoj E, Halbach S, McDonald-McGinn D, Tan C, Lande R, Waggoner D et al (2013) Expanding the spectrum of microdeletion 4q21 syndrome: a partial phenotype with incomplete deletion of the minimal critical region and a new association with cleft palate and Pierre Robin sequence. Am J Med Genet Part A 161A:2327–2333. https://doi.org/10.1002/ajmg.a.36061
Wang H, Pang H, Bartlam M, Rao Z (2005) Crystal structure of human E1 enzyme and its complex with a substrate analog reveals the mechanism of its phosphatase/enolase activity. J Mol Biol 348:917–926
Barth A, Bilkei-Gorzo A, Drews E, Otte DM, Diaz-Lacava A, Varadarajulu J et al (2014) Analysis of quantitative trait loci in mice suggests a role of Enoph1 in stress reactivity. J Neurochem 128:807–817. https://doi.org/10.1111/jnc.12517
Wang B, Xu X, Liu X, Wang D, Zhuang H, He X et al (2021) Enolase-phosphatase 1 acts as an oncogenic driver in glioma. J Cell Physiol 236:1184–1194. https://doi.org/10.1002/jcp.29926
Zhuang H, Qiang Z, Shao X, Wang H, Dang Y, Wang Z et al (2019) Integration of metabolomics and expression of enolase-phosphatase 1 links to hepatocellular carcinoma progression. Theranostics 9:3639–3652. https://doi.org/10.7150/thno.31693
Su L, Yang K, Li S, Liu C, Han J, Zhang Y et al (2018) Enolase-phosphatase 1 as a novel potential malignant glioma indicator promotes cell proliferation and migration. Oncol Rep 40:2233–2241. https://doi.org/10.3892/or.2018.6592
Liu C, He X, Liu X, Yu J, Zhang M, Yu F et al (2019) RPS15A promotes gastric cancer progression via activation of the Akt/IKK-β/NF-κB signalling pathway. J Cell Mol Med 23:2207–2218. https://doi.org/10.1111/jcmm.14141
Chi F, Chen L, Jin X, He G, Liu Z, Han S (2022) CKAP2L, transcriptionally inhibited by FOXP3, promotes breast carcinogenesis through the AKT/mTOR pathway. Exp Cell Res 412:113035. https://doi.org/10.1016/j.yexcr.2022.113035
Kushwaha PP, Gupta S, Singh AK, Kumar S (2019) Emerging role of migration and invasion enhancer 1 (MIEN1) in cancer progression and metastasis. Front Oncol 9:868. https://doi.org/10.3389/fonc.2019.00868
Bonnet C, Andrieux J, Béri-Dexheimer M, Leheup B, Boute O, Manouvrier S et al (2010) Microdeletion at chromosome 4q21 defines a new emerging syndrome with marked growth restriction, mental retardation and absent or severely delayed speech. J Med Genet 47:377–384. https://doi.org/10.1136/jmg.2009.071902
Monteiro LS, Diniz-Freitas M, Warnakulasuriya S, Garcia-Caballero T, Forteza-Vila J, Fraga M (2018) Prognostic Significance of Cyclins A2, B1, D1, and E1 and Numerical Aberrations in Oral Squamous Cell Carcinomas. Anal Cell Pathol (Amst) 2018:7253510. https://doi.org/10.1155/2018/7253510
Choiniere J, Wu J, Wang L (2017) Pyruvate dehydrogenase Kinase 4 deficiency results in expedited cellular proliferation through E2F1-mediated increase of cyclins. Mol Pharmacol 91:189–196. https://doi.org/10.1124/mol.116.106757
Zhang Q, Lenardo MJ, Baltimore D (2017) 30 years of NF-κB: a blossoming of relevance to human pathobiology. Cell 168:37–57. https://doi.org/10.1016/j.cell.2016.12.012
Zhou Y, Eppenberger-Castori S, Marx C, Yau C, Scott GK, Eppenberger U et al (2005) Activation of nuclear factor-kappaB (NFkappaB) identifies a high-risk subset of hormone-dependent breast cancers. Int J Biochem Cell Biol 37:1130–1144
deGraffenried LA, Chandrasekar B, Friedrichs WE, Donzis E, Silva J, Hidalgo M et al (2004) NF-kappa B inhibition markedly enhances sensitivity of resistant breast cancer tumor cells to tamoxifen. Ann Oncol 15:885–890
Zhen Z-G, Ren S-H, Ji H-M, Ma J-H, Ding X-M, Feng F-Q et al (2017) Linarin suppresses glioma through inhibition of NF-κB/p65 and up-regulating p53 expression in vitro and in vivo. Biomed Pharmacother 95:363–374. https://doi.org/10.1016/j.biopha.2017.08.023
Huang L, Lin H, Chen Q, Yu L, Bai D (2019) MPPa-PDT suppresses breast tumor migration/invasion by inhibiting Akt-NF-κB-dependent MMP-9 expression via ROS. BMC Cancer 19:1159. https://doi.org/10.1186/s12885-019-6374-x
Liu B, Sun L, Liu Q, Gong C, Yao Y, Lv X et al (2015) A cytoplasmic NF-κB interacting long noncoding RNA blocks IκB phosphorylation and suppresses breast cancer metastasis. Cancer Cell 27:370–381. https://doi.org/10.1016/j.ccell.2015.02.004
Lu Z, Li Y, Wang J, Che Y, Sun S, Huang J et al (2017) Long non-coding RNA NKILA inhibits migration and invasion of non-small cell lung cancer via NF-κB/Snail pathway. J Exp Clin Cancer Res 36:54. https://doi.org/10.1186/s13046-017-0518-0
Lin H-H, Chen J-H, Chou F-P, Wang C-J (2011) Protocatechuic acid inhibits cancer cell metastasis involving the down-regulation of Ras/Akt/NF-κB pathway and MMP-2 production by targeting RhoB activation. Br J Pharmacol 162:237–254. https://doi.org/10.1111/j.1476-5381.2010.01022.x
Chen Z-D, Xu L, Tang K-K, Gong F-X, Liu J-Q, Ni Y et al (2016) NF-κB-dependent transcriptional upregulation of cyclin D1 exerts cytoprotection against hypoxic injury upon EGFR activation. Exp Cell Res 347:52–59. https://doi.org/10.1016/j.yexcr.2016.07.004
Liu J, Jin P, Lin X, Zhou Q, Wang F, Liu S et al (2018) Arsenite increases Cyclin D1 expression through coordinated regulation of the Ca/NFAT2 and NF-κB pathways via ERK/MAPK in a human uroepithelial cell line. Metallomics 10:486–495. https://doi.org/10.1039/c7mt00305f
Hinz M, Krappmann D, Eichten A, Heder A, Scheidereit C, Strauss M (1999) NF-kappaB function in growth control: regulation of cyclin D1 expression and G0/G1-to-S-phase transition. Mol Cell Biol 19:2690–2698
Acknowledgements
Not available.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Yuhui Bu and Li Ma designed the study, analyzed the result and prepared the manuscripts. Jun Hao analyzed TCGA data, and performed experiment with Xiaolong Li and Jianchao He. Yinfeng Liu and Yuhui Bu were collected data and prepared the figures. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest regarding the publication of this article.
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of the Fourth Hospital of Hebei Medical University (approval No. 2020KY113). Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bu, Y., Hao, J., He, J. et al. Tumor-promoting properties of enolase-phosphatase 1 in breast cancer via activating the NF-κB signaling pathway. Mol Biol Rep 50, 993–1004 (2023). https://doi.org/10.1007/s11033-022-08066-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-022-08066-w