Abstract
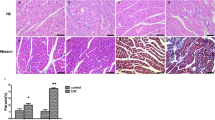
Cardiac complications are leading causes of death in diabetic patients. Imbalance of Ca2+ homeostasis is a hallmark of cardiac dysfunction in diabetes, while TRPV channels are non-selective for cations and are permeable to Ca2+. Our aim was to evaluate the expression levels of TRPV1, TRPV2, TRPV3, TRPV4, TRPV5, and TRPV6 genes and proteins in cardiac tissue at 3 days and 4, 8, and 12 weeks after induction of diabetes. Sprague-Dawley rats were assigned to control and DM groups. DM was induced by intraperitoneal injection of streptozotocin (60 mg/kg). The expression levels of TRPV genes were analyzed by the quantitative reverse transcription polymerase chain reaction, and TRPV proteins were determined by western blotting. Compared to controls, the expression levels of TRPV2, TRPV3, and TRPV6 in diabetic myocardium did not change, while TRPV1 decreased at 4, 8, and 12 weeks, TRPV4 was upregulated at 3 days and 4, 8, and 12 weeks, TRPV5 mRNA increased at 8 and 12 weeks, and TRPV5 protein increased at 4, 8, and 12 weeks. Our findings showed that TRPV1, TRPV4, and TRPV5 are associated with the diabetic heart.



Similar content being viewed by others
Abbreviations
- DM:
-
Diabetes mellitus
- qRT-PCR:
-
Quantitative reverse transcription polymerase chain reaction
- RT:
-
Reverse transcription
- PCR:
-
Polymerase chain reaction
- SD:
-
Standard deviation
- STZ:
-
Streptozotocin
- BCA:
-
Bicinchoninic acid
- TRP:
-
Transient receptor potential
- TRPV:
-
Transient receptor potential vanilloid
- ATP:
-
5'-Adenylate triphosphate
- GAPDH:
-
Glyceraldehyde-3-phosphate dehydrogenase
References
Mancini GBJ, Cheng AY, Connelly K et al (2017) Diabetes for cardiologists: practical issues in diagnosis and management. Can J Cardiol 33:366–377. https://doi.org/10.1016/j.cjca.2016.07.512
Zhang L, Ward M-L, Phillips ARJ et al (2013) Protection of the heart by treatment with a divalent-copper-selective chelator reveals a novel mechanism underlying cardiomyopathy in diabetic rats. Cardiovasc Diabetol 12:123. https://doi.org/10.1186/1475-2840-12-123
Suarez J, Cividini F, Scott BT et al (2018) Restoring mitochondrial calcium uniporter expression in diabetic mouse heart improves mitochondrial calcium handling and cardiac function. J Biol Chem 293:8182–8195. https://doi.org/10.1074/jbc.RA118.002066
Jacobson DA, Philipson LH (2007) TRP channels of the pancreatic beta cell. Handb Exp Pharmacol 179:409–424. https://doi.org/10.1007/978-3-540-34891-7_24
Falcon D, Galeano-Otero I, Calderon-Sanchez E et al (2019) TRP channels: current perspectives in the adverse cardiac remodeling. Front Physiol 10:159. https://doi.org/10.3389/fphys.2019.00159
Li H (2017) TRP channel classification. Adv Exp Med Biol 976:1–8. https://doi.org/10.1007/978-94-024-1088-4_1
Hof T, Chaigne S, Recalde A et al (2019) Transient receptor potential channels in cardiac health and disease. Nat Rev Cardiol 16:344–360. https://doi.org/10.1038/s41569-018-0145-2
Morine KJ, Paruchuri V, Qiao X et al (2016) Endoglin selectively modulates transient receptor potential channel expression in left and right heart failure. Cardiovasc Pathol 25:478–482. https://doi.org/10.1016/j.carpath.2016.08.004
Adapala RK, Thoppil RJ, Luther DJ et al (2013) TRPV4 channels mediate cardiac fibroblast differentiation by integrating mechanical and soluble signals. J Mol Cell Cardiol 54:45–52. https://doi.org/10.1016/j.yjmcc.2012.10.016
Heymann HM, Wu Y, Lu Y et al (2017) Transient receptor potential vanilloid 1 inhibitors block laparotomy- and opioid-induced infarct size reduction in rats. Br J Pharmacol 174:4826–4835. https://doi.org/10.1111/bph.14064
Liu Y, Qi H, E M et al (2018) Transient receptor potential vanilloid-3 (TRPV3) activation plays a central role in cardiac fibrosis induced by pressure overload in rats via TGF-beta1 pathway. Naunyn Schmiedeberg’s Arch Pharmacol 391:131–143. https://doi.org/10.1007/s00210-017-1443-7
Sun T, Guo Z, Liu C-J et al (2016) Preservation of CGRP in myocardium attenuates development of cardiac dysfunction in diabetic rats. Int J Cardiol 220:226–234. https://doi.org/10.1016/j.ijcard.2016.06.092
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-delta delta C(T)) method. Methods (San Diego, CA) 25:402–408. https://doi.org/10.1006/meth.2001.1262
Morgan JP, Erny RE, Allen PD et al (1990) Abnormal intracellular calcium handling, a major cause of systolic and diastolic dysfunction in ventricular myocardium from patients with heart failure. Circulation 81:III21–III32
Lebeche D, Davidoff AJ, Hajjar RJ (2008) Interplay between impaired calcium regulation and insulin signaling abnormalities in diabetic cardiomyopathy. Nat Clin Pract Cardiovasc Med 5:715–724. https://doi.org/10.1038/ncpcardio1347
Santoni G, Farfariello V, Amantini C (2011) TRPV channels in tumor growth and progression. Adv Exp Med Biol 704:947–967. https://doi.org/10.1007/978-94-007-0265-3_49
Jia Y, Lee L-Y (2007) Role of TRPV receptors in respiratory diseases. Biochim Biophys Acta 1772:915–927. https://doi.org/10.1016/j.bbadis.2007.01.013
Skrzypski M, Billert M, Mergler S et al (2017) Role of TRPV channels in regulating various pancreatic beta-cell functions: lessons from in vitro studies. Biosci Trends 11:9–15. https://doi.org/10.5582/bst.2016.01226
Robbins N, Koch SE, Rubinstein J (2013) Targeting TRPV1 and TRPV2 for potential therapeutic interventions in cardiovascular disease. Transl Res J Lab Clin Med 161:469–476. https://doi.org/10.1016/j.trsl.2013.02.003
Clapham DE (2003) TRP channels as cellular sensors. Nature 426:517–524. https://doi.org/10.1038/nature02196
Zahner MR, Li D-P, Chen S-R, Pan H-L (2003) Cardiac vanilloid receptor 1-expressing afferent nerves and their role in the cardiogenic sympathetic reflex in rats. J Physiol 551:515–523. https://doi.org/10.1113/jphysiol.2003.048207
Marshall NJ, Liang L, Bodkin J et al (2013) A role for TRPV1 in influencing the onset of cardiovascular disease in obesity. Hypertens (Dallas, TX 1979) 61:246–252. https://doi.org/10.1161/HYPERTENSIONAHA.112.201434
Lang H, Li Q, Yu H et al (2015) Activation of TRPV1 attenuates high salt-induced cardiac hypertrophy through improvement of mitochondrial function. Br J Pharmacol 172:5548–5558. https://doi.org/10.1111/bph.12987
Gao Y, Song J, Chen H et al (2015) TRPV1 activation is involved in the cardioprotection of remote limb ischemic postconditioning in ischemia-reperfusion injury rats. Biochem Biophys Res Commun 463:1034–1039. https://doi.org/10.1016/j.bbrc.2015.06.054
Chen J, Li L, Li Y et al (2015) Activation of TRPV1 channel by dietary capsaicin improves visceral fat remodeling through connexin43-mediated Ca2+ influx. Cardiovasc Diabetol 14:22. https://doi.org/10.1186/s12933-015-0183-6
Song J-X, Wang L-H, Yao L et al (2009) Impaired transient receptor potential vanilloid 1 in streptozotocin-induced diabetic hearts. Int J Cardiol 134:290–292
Filosa JA, Yao X, Rath G (2013) TRPV4 and the regulation of vascular tone. J Cardiovasc Pharmacol 61:113–119. https://doi.org/10.1097/FJC.0b013e318279ba42
Randhawa PK, Jaggi AS (2015) TRPV1 and TRPV4 channels: potential therapeutic targets for ischemic conditioning-induced cardioprotection. Eur J Pharmacol 746:180–185. https://doi.org/10.1016/j.ejphar.2014.11.010
Berrout J, Jin M, Mamenko M et al (2012) Function of transient receptor potential cation channel subfamily V member 4 (TRPV4) as a mechanical transducer in flow-sensitive segments of renal collecting duct system. J Biol Chem 287:8782–8791. https://doi.org/10.1074/jbc.M111.308411
Mensenkamp AR, Hoenderop JGJ, Bindels RJM (2007) TRPV5, the gateway to Ca2+ homeostasis. Handb Exp Pharmacol 179:207–220. https://doi.org/10.1007/978-3-540-34891-7_12
de Sousa AR, Penalva LO, Marcotte EM, Vogel C (2009) Global signatures of protein and mRNA expression levels. Mol BioSyst 5:1512–1526. https://doi.org/10.1039/b908315d
Acknowledgements
This study was supported by grants from the National Natural Science Foundation of China (30872716), the Natural Science Foundation of Hubei Province (2015CFB288), and a Health and Family Planning Project in Hubei Province (WJ2015MB171).
Author information
Authors and Affiliations
Contributions
X.L.J. and T.Y. carried out the experiments and drafted the manuscript. C.X. and D.Q.S. was involved in data analysis. M.C.Y., Q.Y.C., J.W., and T.L. contributed to conducting the experiments. Y.Z. and S.Z.Z. reviewed the data, and revised the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study did not require ethical approval.
Informed consent
All author have seen the manuscript and approved its submission.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jia, X., Yu, T., Xiao, C. et al. Expression of transient receptor potential vanilloid genes and proteins in diabetic rat heart. Mol Biol Rep 48, 1217–1223 (2021). https://doi.org/10.1007/s11033-021-06182-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-021-06182-7