Abstract
Background
Postoperative anxiety is a common surgical complication in older patients. Research has recently linked excessive autophagy to several neurological disorders, including anxiety. This study aimed to determine whether 3-Methyladenine (3-MA) administration reduced anxiety-like behaviors in a mouse model following abdominal exploratory laparotomy.
Methods
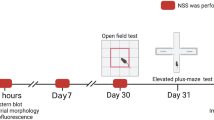
An abdominal exploratory laparotomy model of postoperative anxiety was established using male C57BL/6 mice aged 20 months. 3-MA (6, 30, and 150 mg/ml) was administered via intracerebroventricular immediately following surgery. The mice were assessed 14 days after surgery using the marble burying, elevated plus maze tests, and local field potential recording in the amygdala. The levels of expression of phosphorylated-Akt, Beclin-1, LC3B, nuclear factor erythroid 2-related factor 2 (Nrf2)-occupied regions in NeuN-positive cells, superoxide dismutase (SOD) activity, malondialdehyde (MDA), and glutathione (GSH) were measured at 24 h after surgery.
Results
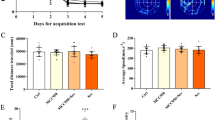
The injection of 3-MA reversed the increased number of marbles buried, decreased time spent in the open arm, and enhanced θ oscillation power after 14 days of abdominal exploratory laparotomy. In addition, administration of 3-MA reduced the ratio of phosphorylated- to total-Akt, decreased expression in Beclin-1 and LC3B, attenuated MDA levels, and increased the ratio of Nrf2-occupied areas in NeuN-positive cells, SOD activity, and GSH levels under abdominal exploratory laparotomy conditions.
Conclusions
3-MA improved anxiety-like behaviors in aged mice undergoing abdominal exploratory laparotomy by inhibiting excessive autophagy-induced oxidative stress. These results suggest that 3-MA could be an effective treatment for postoperative anxiety.




Similar content being viewed by others
Data Availability
All data generated and analyzed during this study are included in this published article and its supplementary information file.
References
Adhikari A, Lerner TN, Finkelstein J, Pak S, Jennings JH, Davidson TJ, Ferenczi E, Gunaydin LA, Mirzabekov JJ, Ye L, Kim SY, Lei A, Deisseroth K (2015) Basomedial amygdala mediates top-down control of anxiety and fear. Nature 527(7577):179–185. https://doi.org/10.1038/nature15698
Al-Amin MM, Akhter S, Hasan AT, Alam T, Nageeb Hasan SM, Saifullah AR, Shohel M (2015) The antioxidant effect of astaxanthin is higher in young mice than aged: a region specific study on brain. Metab Brain Dis 30(5):1237–1246. https://doi.org/10.1007/s11011-015-9699-4
Alegre-Zurano L, López-Arnau R, Luján M, Camarasa J, Valverde O (2021) Cannabidiol modulates the motivational and anxiety-like Effects of 3,4-Methylenedioxypyrovalerone (MDPV) in mice. Int J Mol Sci 22(15). https://doi.org/10.3390/ijms22158304
Babaev O, Piletti Chatain C, Krueger-Burg D (2018) Inhibition in the amygdala anxiety circuitry. Exp Mol Med 50(4):1–16. https://doi.org/10.1038/s12276-018-0063-8
Boone MD, Sites B, von Recklinghausen FM, Mueller A, Taenzer AH, Shaefi S (2020) Economic Burden of Postoperative Neurocognitive Disorders Among US Medicare Patients. JAMA network open3(7):e208931. https://doi.org/10.1001/jamanetworkopen.2020.8931
Bostancıklıoğlu M (2019) An update on the interactions between Alzheimer’s disease, autophagy and inflammation. https://doi.org/10.1016/j.gene.2019.04.040. Gene705157-166
Cao Y, Li Q, Liu L, Wu H, Huang F, Wang C, Lan Y, Zheng F, Xing F, Zhou Q, Li Q, Shi H, Zhang B, Wang Z, Wu X (2019) Modafinil protects hippocampal neurons by suppressing excessive autophagy and apoptosis in mice with sleep deprivation. Br J Pharmacol 176(9):1282–1297. https://doi.org/10.1111/bph.14626
Cruces-Solis H, Babaev O, Ali H, Piletti Chatain C, Mykytiuk V, Balekoglu N, Wenger S, Krueger-Burg D (2021) Altered theta and beta oscillatory synchrony in a genetic mouse model of pathological anxiety. Faseb j 35(6):e21585. https://doi.org/10.1096/fj.202002028RR
Cuenod M, Steullet P, Cabungcal JH, Dwir D, Khadimallah I, Klauser P, Conus P, Do KQ (2022) Caught in vicious circles: a perspective on dynamic feed-forward loops driving oxidative stress in schizophrenia. Mol Psychiatry 27(4):1886–1897. https://doi.org/10.1038/s41380-021-01374-w
De R, Mazumder S, Sarkar S, Debsharma S, Siddiqui AA, Saha SJ, Banerjee C, Nag S, Saha D, Bandyopadhyay U (2017) Acute mental stress induces mitochondrial bioenergetic crisis and hyper-fission along with aberrant mitophagy in the gut mucosa in rodent model of stress-related mucosal disease. Free radical biology. https://doi.org/10.1016/j.freeradbiomed.2017.10.009. & medicine113424-438
de Brouwer G, Fick A, Harvey BH, Wolmarans W (2019) A critical inquiry into marble-burying as a preclinical screening paradigm of relevance for anxiety and obsessive-compulsive disorder: mapping the way forward. Cogn Affect Behav Neurosci 19(1):1–39. https://doi.org/10.3758/s13415-018-00653-4
Duan K, Gu Q, Petralia RS, Wang YX, Panja D, Liu X, Lehmann ML, Zhu H, Zhu J, Li Z (2021) Mitophagy in the basolateral amygdala mediates increased anxiety induced by aversive social experience. Neuron109(23):3793–3809.e3798. https://doi.org/10.1016/j.neuron.2021.09.008
Glick D, Barth S, Macleod KF (2010) Autophagy: cellular and molecular mechanisms. J Pathol 221(1):3–12. https://doi.org/10.1002/path.2697
Gründemann J, Bitterman Y, Lu T, Krabbe S, Grewe BF, Schnitzer MJ, Lüthi A (2019) Amygdala ensembles encode behavioral states. Science. (New York, N.Y.)364(6437https://doi.org/10.1126/science.aav8736
Guglielmotto M, Monteleone D, Piras A, Valsecchi V, Tropiano M, Ariano S, Fornaro M, Vercelli A, Puyal J, Arancio O, Tabaton M, Tamagno E (2014) Aβ1–42 monomers or oligomers have different effects on autophagy and apoptosis. Autophagy10(10):1827–1843. https://doi.org/10.4161/auto.30001
Gür F, Gür B, Erkayman B, Halıcı Z, Karakoç A (2020) Investigation of serum and brain superoxide dismutase levels depending on atomoxetine used in attention-deficit/hyperactivity disorder treatment: a combination of in vivo and molecular docking studies. https://doi.org/10.1016/j.bioorg.2020.104435. Bioorganic chemistry105104435
Jacinto LR, Cerqueira JJ, Sousa N (2016) Patterns of Theta activity in limbic anxiety circuit preceding exploratory Behavior in Approach-Avoidance conflict. Frontiers in behavioral neuroscience10171 https://doi.org/10.3389/fnbeh.2016.00171
Jimenez JC, Su K, Goldberg AR, Luna VM, Biane JS, Ordek G, Zhou P, Ong SK, Wright MA, Zweifel L, Paninski L, Hen R, Kheirbek MA (2018) Anxiety Cells in a Hippocampal-Hypothalamic Circuit. Neuron97(3):670–683.e676. https://doi.org/10.1016/j.neuron.2018.01.016
Jimenez-Gomez C, Osentoski A, Woods JH (2011) Pharmacological evaluation of the adequacy of marble burying as an animal model of compulsion and/or anxiety. Behav Pharmacol 22(7):711–713. https://doi.org/10.1097/FBP.0b013e32834afebe
Jung S, Choe S, Woo H, Jeong H, An HK, Moon H, Ryu HY, Yeo BK, Lee YW, Choi H, Mun JY, Sun W, Choe HK, Kim EK, Yu SW (2020) Autophagic death of neural stem cells mediates chronic stress-induced decline of adult hippocampal neurogenesis and cognitive deficits. Autophagy16(3):512–530. https://doi.org/10.1080/15548627.2019.1630222
Kagerbauer SM, Martin J, Ulm B, Jungwirth B, Podtschaske AH (2019) Influence of perioperative stress on central and peripheral oxytocin and arginine-vasopressin concentrations. J Neuroendocrinol 31(10):e12797. https://doi.org/10.1111/jne.12797
Knight P, Chellian R, Wilson R, Behnood-Rod A, Panunzio S, Bruijnzeel AW (2021) Sex differences in the elevated plus-maze test and large open field test in adult Wistar rats. https://doi.org/10.1016/j.pbb.2021.173168. Pharmacol Biochem Behav204173168
Li X, He S, Ma B (2020) Autophagy and autophagy-related proteins in cancer. Mol Cancer 19(1):12. https://doi.org/10.1186/s12943-020-1138-4
Liu T, Han S, Dai Q, Zheng J, Liu C, Li S, Li J (2019) IL-17A-Mediated excessive autophagy aggravated neuronal ischemic injuries via Src-PP2B-mTOR pathway. https://doi.org/10.3389/fimmu.2019.02952. Front Immunol102952
Liu T, Li Z, He J, Yang N, Han D, Li Y, Tian X, Liu H, Manyande A, Xiang H, Xu F, Wang J, Guo X (2020) Regional metabolic patterns of abnormal postoperative behavioral performance in aged mice assessed by (1)H-NMR dynamic mapping method. Neurosci Bull 36(1):25–38. https://doi.org/10.1007/s12264-019-00414-4
Liu Y, Chen Z, Li B, Yao H, Zarka M, Welch J, Sachdev P, Bridge W, Braidy N (2021) Supplementation with γ-glutamylcysteine (γ-GC) lessens oxidative stress, brain inflammation and amyloid pathology and improves spatial memory in a murine model of AD. https://doi.org/10.1016/j.neuint.2020.104931. Neurochem Int144104931
Lu J, Liu Z, Shu M, Zhang L, Xia W, Tang L, Li J, Huang B, Li H (2021) Human placental mesenchymal stem cells ameliorate chemotherapy-induced damage in the testis by reducing apoptosis/oxidative stress and promoting autophagy. Stem Cell Res Ther 12(1):199. https://doi.org/10.1186/s13287-021-02275-z
Luo J, Yan D, Li S, Liu S, Zeng F, Cheung CW, Liu H, Irwin MG, Huang H, Xia Z (2020) Allopurinol reduces oxidative stress and activates Nrf2/p62 to attenuate diabetic cardiomyopathy in rats. J Cell Mol Med 24(2):1760–1773. https://doi.org/10.1111/jcmm.14870
Manou-Stathopoulou V, Korbonits M, Ackland GL (2019) Redefining the perioperative stress response: a narrative review. Br J Anaesth 123(5):570–583. https://doi.org/10.1016/j.bja.2019.08.011
McCoy CR, Sabbagh MN, Huaman JP, Pickrell AM, Clinton SM (2019) Oxidative metabolism alterations in the emotional brain of anxiety-prone rats. Prog Neuropsychopharmacol Biol Psychiatry95109706. https://doi.org/10.1016/j.pnpbp.2019.109706
McKenzie LH, Simpson J, Stewart M (2010) A systematic review of pre-operative predictors of post-operative depression and anxiety in individuals who have undergone coronary artery bypass graft surgery. Psychol Health Med 15(1):74–93. https://doi.org/10.1080/13548500903483486
Muscat SM, Deems NP, D’Angelo H, Kitt MM, Grace PM, Andersen ND, Silverman SN, Rice KC, Watkins LR, Maier SF, Barrientos RM (2021) Postoperative cognitive dysfunction is made persistent with morphine treatment in aged rats. Neurobiol Aging 98214–98224. https://doi.org/10.1016/j.neurobiolaging.2020.11.008
Netto MB, de Oliveira Junior AN, Goldim M, Mathias K, Fileti ME, da Rosa N, Laurentino AO, de Farias BX, Costa AB, Rezin GT, Fortunato JJ, Giustina AD, Barichello T, Dal-Pizzol F, Petronilho F (2018) Oxidative stress and mitochondrial dysfunction contributes to postoperative cognitive dysfunction in elderly rats. Brain Behav Immun 73661–73669. https://doi.org/10.1016/j.bbi.2018.07.016
Parzych KR, Klionsky DJ (2014) An overview of autophagy: morphology, mechanism, and regulation. Antioxid Redox Signal 20(3):460–473. https://doi.org/10.1089/ars.2013.5371
Paxinos G, Franklin K (2001) Mouse brain in stereotaxic coordinates. Psychoneuroendocrinology
Qiu LL, Ji MH, Zhang H, Yang JJ, Sun XR, Tang H, Wang J, Liu WX, Yang JJ (2016) NADPH oxidase 2-derived reactive oxygen species in the hippocampus might contribute to microglial activation in postoperative cognitive dysfunction in aged mice. Brain Behav Immun 51109–51118. https://doi.org/10.1016/j.bbi.2015.08.002
Salimando GJ, Hyun M, Boyt KM, Winder DG (2020) BNST GluN2D-Containing NMDA receptors influence anxiety- and depressive-like behaviors and ModulateCell-Specific Excitatory/Inhibitory synaptic balance. J neuroscience: official J Soc Neurosci 40(20):3949–3968. https://doi.org/10.1523/jneurosci.0270-20.2020
Salvi A, Liu H, Salim S (2020) Involvement of oxidative stress and mitochondrial mechanisms in air pollution-related neurobiological impairments. https://doi.org/10.1016/j.ynstr.2019.100205. Neurobiology of stress12100205
Stamenkovic DM, Rancic NK, Latas MB, Neskovic V, Rondovic GM, Wu JD, Cattano D (2018) Preoperative anxiety and implications on postoperative recovery: what can we do to change our history. Minerva Anestesiol 84(11):1307–1317. https://doi.org/10.23736/s0375-9393.18.12520-x
Su LJ, Zhang JH, Gomez H, Murugan R, Hong X, Xu D, Jiang F, Peng ZY (2019) Reactive Oxygen Species-Induced lipid peroxidation in apoptosis, Autophagy, and ferroptosis. Oxid Med Cell Longev 20195080843. https://doi.org/10.1155/2019/5080843
Tang Q, Guo Q, Li K, Fei F (2022) VRT-043198 ameliorates Surgery-Induced Neurocognitive Disorders by restoring the NGF and BNDF expression in aged mice. Neuropsychiatric disease and treatment181027-1037. https://doi.org/10.2147/ndt.S364250
Tovote P, Fadok JP, Lüthi A (2015) Neuronal circuits for fear and anxiety. Nat Rev Neurosci 16(6):317–331. https://doi.org/10.1038/nrn3945
Ułamek-Kozioł M, Furmaga-Jabłońska W, Januszewski S, Brzozowska J, Sciślewska M, Jabłoński M, Pluta R (2013) Neuronal autophagy: self-eating or self-cannibalism in Alzheimer’s disease. Neurochem Res 38(9):1769–1773. https://doi.org/10.1007/s11064-013-1082-4
Varga ZV, Giricz Z, Liaudet L, Haskó G, Ferdinandy P, Pacher P (2015) Interplay of oxidative, nitrosative/nitrative stress, inflammation, cell death and autophagy in diabetic cardiomyopathy. Biochim Biophys Acta 1852(2):232–242. https://doi.org/10.1016/j.bbadis.2014.06.030
Vollert C, Zagaar M, Hovatta I, Taneja M, Vu A, Dao A, Levine A, Alkadhi K, Salim S (2011) Exercise prevents sleep deprivation-associated anxiety-like behavior in rats: potential role of oxidative stress mechanisms. Behav Brain Res 224(2):233–240. https://doi.org/10.1016/j.bbr.2011.05.010
Wang X, Wang Z, Cao J, Dong Y, Chen Y (2021) Melatonin ameliorates anxiety-like behaviors induced by sleep deprivation in mice: role of oxidative stress, neuroinflammation, autophagy and apoptosis. https://doi.org/10.1016/j.brainresbull.2021.06.010. Brain research bulletin174161-172
Wei N, Yu SP, Gu XH, Chen DD, Whalin MK, Xu GL, Liu XF, Wei L (2013) The involvement of autophagy pathway in exaggerated ischemic brain damage in diabetic mice. CNS Neurosci Ther 19(10):753–763. https://doi.org/10.1111/cns.12123
Wu YT, Tan HL, Shui G, Bauvy C, Huang Q, Wenk MR, Ong CN, Codogno P, Shen HM (2010) Dual role of 3-methyladenine in modulation of autophagy via different temporal patterns of inhibition on class I and III phosphoinositide 3-kinase. J Biol Chem 285(14):10850–10861. https://doi.org/10.1074/jbc.M109.080796
Wu Y, Duan X, Gao Z, Yang N, Xue F (2022) AICAR attenuates postoperative abdominal adhesion formation by inhibiting oxidative stress and promoting mesothelial cell repair. PLoS ONE 17(9):e0272928. https://doi.org/10.1371/journal.pone.0272928
Zhang XY, Peng SY, Shen LP, Zhuang QX, Li B, Xie ST, Li QX, Shi MR, Ma TY, Zhang Q, Wang JJ, Zhu JN (2020) Targeting presynaptic H3 heteroreceptor in nucleus accumbens to improve anxiety and obsessive-compulsive-like behaviors. Proc Natl Acad Sci USA 117(50):32155–32164. https://doi.org/10.1073/pnas.2008456117
Zhao L, Zhang B, Cui Y, Hou C, Zeng Q, Gao T, Zhang Z, Yu J, Wang Y, Wang A, Liu H (2019) 3-Methyladenine alleviates excessive iodine-induced cognitive impairment via suppression of autophagy in rat hippocampus. Environ Toxicol 34(8):912–920. https://doi.org/10.1002/tox.22762
Zheng ZH, Tu JL, Li XH, Hua Q, Liu WZ, Liu Y, Pan BX, Hu P, Zhang WH (2021) Neuroinflammation induces anxiety- and depressive-like behavior by modulating neuronal plasticity in the basolateral amygdala. Brain, behavior, and immunity. 91505–91518. https://doi.org/10.1016/j.bbi.2020.11.007
Funding
This study was supported by the key research and development program from the Bureau of Science and Technology of Qinhuangdao (202101A186).
Author information
Authors and Affiliations
Contributions
Song-Tao Wu: Methodology, Investigation, Formal Analysis, Writing - Original Draft. Shan-Shan Han: Methodology, Formal Analysis, Data Curation. Xi-Ming Xu: Methodology, Formal Analysis, Software. Hai-Jun Sun: Methodology, Formal Analysis. Hua Zhou: Investigation, Formal Analysis. Kun Shang: Methodology, Software. Zi-Hao Liu: Methodology, Formal Analysis. Shu-Juan Liang: Conceptualization, Writing - Review & Editing, Supervision, Project Administration.
All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Statement of ethics
All animal studies were carried out under the National Institute of Health’s Guideline for the Care and Use of Laboratory Animals. The Animal Review Board also approved the protocols involving animals of the First Hospital of Qinhuangdao (2021Q085).
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, ST., Han, SS., Xu, XM. et al. 3-Methyladenine ameliorates surgery-induced anxiety-like behaviors in aged mice by inhibiting autophagy-induced excessive oxidative stress. Metab Brain Dis 38, 1913–1923 (2023). https://doi.org/10.1007/s11011-023-01217-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-023-01217-3