Abstract
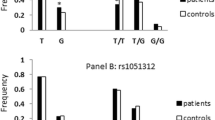
Imbalance in dopamine (DA) signaling is proposed to play a potential role in the etiology of Autism spectrum disorder (ASD) since, as a neuromodulator, DA regulates executive function, motor activity, social peering, attention as well as perception and subjects with ASD often exhibit deficit in these traits. Level of DA in the synaptic cleft is maintained by dopamine transporter (DAT) and hence, to identify the role of DAT in ASD, we have analyzed four functional genetic variants, rs28363170, rs3836790, rs2652511, rs27072, in nuclear families with ASD probands. Subjects were diagnosed based on Diagnostic and Statistical Manual for Mental Disorders and trait severity was assessed by Childhood Autism Rating Scale 2-Standard test. Informed written consent was obtained from the parents/care givers before recruitment followed by collection of peripheral blood for genomic DNA isolation. Target sites were investigated by PCR-based methods and data obtained was analyzed by population- as well as family-based statistical methods. Case-control analysis revealed significant higher frequencies of 9 repeat (9R) and 5 repeat (5R) alleles of rs28363170 and rs3836790 respectively in the ASD probands. Family-based analysis showed statistically significant higher paternal transmission of rs28363170 9R and rs2652511 T alleles. In the presence of rs28363170 9R, rs27072 C, rs3836790 6R6R, and rs2652511 CC variants, trait scores were higher. Studied variants showed independent as well as interactive effects, which varied based on gender of the probands. We infer that altered DA availability mediated through DAT may affect autistic traits warranting further in depth investigation in the field.



Similar content being viewed by others
Data availability
Data generated from the study are presented in tabular format. Further details are presented in Additional files. The sequences of SNP were obtained from NCBI SNP database.
Abbreviations
- ADHD :
-
Attention Deficit Hyperactive Disorder
- ASD :
-
Autism Spectrum Disorder
- CARS2-ST :
-
Childhood Autism Rating Scale 2-Standard Test
- DA :
-
Dopamine
- DAT :
-
Dopamine Transporter
- DSM :
-
Diagnostic and Statistical Manual of Mental Disorders
- FDA :
-
Food and Drug Administration
- HWE :
-
Hardy-Weinberg Equilibrium
- IG :
-
Information Gain
- LD :
-
Linkage Disequilibrium
- MDR :
-
Multifactor Dimensionality Reduction
- OR :
-
Odd Ratio
- PCR :
-
Polymerase Chain Reaction
- QTA :
-
Quantitative Trait Analysis
- RFLP :
-
Restriction Fragment Length Polymorphism
- SNP :
-
Single Nucleotide Polymorphisms
- TDT :
-
Transmission Disequilibrium Test
- UTR :
-
Untranslated Region
- VNTR :
-
Variable Number Tandem Repeat
References
Alsayouf HA, Talo H, Biddappa ML, Qasaymeh M, Qasem S, De Los RE (2020) Pharmacological intervention in children with autism Spectrum disorder with standard supportive therapies significantly improves core signs and symptoms: a single-center, retrospective case series. Neuropsychiatr Dis Treat 16:2779–2794. https://doi.org/10.2147/NDT.S277294
American Psychiatric Association (2000) Diagnostic and statistical manual of mental disorders, 4th edn. American Psychiatric Association, Washington, DC
American Psychiatric Association (2013) Diagnostic and statistical manual of mental disorders, 5th edn. American Psychiatric Association, Arlington
Barrett JC, Fry B, Maller J, Daly MJ (2005) Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 21(2):263–265. https://doi.org/10.1093/bioinformatics/bth457
Barrie ES, Pinsonneault JK, Sadee W, Hollway JA, Handen BL, Smith T, Arnold LE, Butter E, Hansen-Kiss E, Herman GE, Aman MG (2018) Testing genetic modifiers of behavior and response to atomoxetine in autism Spectrum disorder with ADHD. J Dev Phys Disabil 30:355–371. https://doi.org/10.1007/s10882-018-9590-4
Bedard AC, Schulz KP, Cook EH Jr, Fan J, Clerkin SM, Ivanov I, Halperin JM, Newcorn JH (2010) Dopamine transporter gene variation modulates activation of striatum in youth with ADHD. NeuroImage 53:935–942. https://doi.org/10.1016/j.neuroimage.2009.12.041
Bhaskar LV, Thangaraj K, Wasnik S, Singh L, Raghavendra Rao V (2012) Dopamine transporter (DAT1) VNTR polymorphism and alcoholism in two culturally different populations of South India. Am J Addict 21:343–347. https://doi.org/10.1111/j.1521-0391.2012.00244.x
Bowton E, Saunders C, Reddy IA, Campbell NG, Hamilton PJ, Henry LK, Coon H, Sakrikar D, Veenstra-VanderWeele JM, Blakely RD, Sutcliffe J, Matthies HJ, Erreger K, Galli A (2014) SLC6A3 coding variant Ala559Val found in two autism probands alters dopamine transporter function and trafficking. Transl Psychiatry 4:e464. https://doi.org/10.1038/tp.2014.90
Brookes KJ, Mill J, Guindalini C, Curran S, Xu X, Knight J, Chen CK, Huang YS, Sethna V, Taylor E, Chen W, Breen G, Asherson P (2006) A common haplotype of the dopamine transporter gene associated with attention-deficit/hyperactivity disorder and interacting with maternal use of alcohol during pregnancy. Arch Gen Psychiatry 63:74–81. https://doi.org/10.1001/archpsyc.63.1.74
CDC (2020) Centers for Disease control and Prevention. Prevalence of Autism Spectrum Disorder among children aged 8 years — Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2016. https://doi.org/10.15585/mmwr.ss6904a1
Chaste P, Leboyer M (2012) Autism risk factors: genes, environment, and gene-environment interactions. Dialogues Clin Neurosci 14:281–292. https://doi.org/10.31887/DCNS.2012.14.3/pchaste
de Azeredo LA, Rovaris DL, Mota NR, Polina ER, Marques FZ, Contini V, Vitola ES, Belmonte-de-Abreu P, Rohde LA, Grevet EH, Bau CH (2014) Further evidence for the association between a polymorphism in the promoter region of SLC6A3/DAT1 and ADHD: findings from a sample of adults. Eur Arch Psychiatry Clin Neurosci 264:401–408. https://doi.org/10.1007/s00406-014-0486-8
DiCarlo GE, Aguilar JI, Matthies HJ, Harrison FE, Bundschuh KE, West A, Hashemi P, Herborg F, Rickhag M, Chen H, Gether U, Wallace MT, Galli A (2019) Autism-linked dopamine transporter mutation alters striatal dopamine neurotransmission and dopamine-dependent behaviors. J Clin Invest 129:3407–3419. https://doi.org/10.1172/JCI127411
Dichter GS, Damiano CA, Allen JA (2012) Reward circuitry dysfunction in psychiatric and neurodevelopmental disorders and genetic syndromes: animal models and clinical findings. J Neurodev Disord 4:4–19. https://doi.org/10.1186/1866-1955-4-19
Drgon T, Lin Z, Wang GJ, Fowler J, Pablo J, Mash DC, Volkow N, Uhl GR (2006) Common human 5′ dopamine transporter (SLC6A3) haplotypes yield varying expression levels in vivo. Cell Mol Neurobiol 26:873–887. https://doi.org/10.1007/s10571-006-9014-3
Dudbridge F (2008) Likelihood-based association analysis for nuclear families and unrelated subjects with missing genotype data. Hum Hered 66:87–98. https://doi.org/10.1159/00011910
Ernst M, Zametkin AJ, Matochik JA, Pascualvaca D, Cohen RM (1997) Low medial prefrontal dopaminergic activity in autistic children. Lancet 350:638. https://doi.org/10.1016/S0140-6736(05)63326-0
Fanous AH, Neale MC, Straub RE, Webb BT, O'Neill AF, Walsh D, Kendler KS (2004) Clinical features of psychotic disorders and polymorphisms in HT2A, DRD2, DRD4, SLC6A3 (DAT1), and BDNF: a family based association study. Am J Med Genet B Neuropsychiatr Genet 125B:69–78
Faraone SV, Spencer TJ, Madras BK, Zhang-James Y, Biederman J (2014) Functional effects of dopamine transporter gene genotypes on in vivo dopamine transporter functioning: a meta-analysis. Mol Psychiatry 19:880–889. https://doi.org/10.1038/mp.2013.126
Fehér Á, Juhász A, Pákáski M, Kálmán J, Janka Z (2014) Association between the 9 repeat allele of the dopamine transporter 40bp variable tandem repeat polymorphism and Alzheimer’s disease. Psychiatry Res 220(1–2):730–731. https://doi.org/10.1016/j.psychres.2014.07.060
Feng Y, Wigg KG, Makkar R, Ickowicz A, Pathare T, Tannock R, Roberts W, Malone M, Kennedy JL, Schachar R, Barr CL (2005) Sequence variation in the 3′-untranslated region of the dopamine transporter gene and attention-deficit hyperactivity disorder (ADHD). Am J Med Genet B Neuropsychiatr Genet1 39B:1–6. https://doi.org/10.1002/ajmg.b.30190
Gadow KD, Roohi J, DeVincent CJ, Hatchwell E (2008) Association of ADHD, tics, and anxiety with dopamine transporter (DAT1) genotype in autism spectrum disorder. J Child Psychol Psychiatry 49:1331–1338. https://doi.org/10.1111/j.1469-7610.2008.01952.x
Gadow KD, Pinsonneault JK, Perlman G, Sadee W (2014) Association of dopamine gene variants, emotion dysregulation and ADHD in autism spectrum disorder. Res Dev Disabil 35:1658–1665. https://doi.org/10.1016/j.ridd.2014.04.007
Genro JP, Zeni C, Polanczyk GV, Roman T, Rohde LA, Hutz MH (2007) A promoter polymorphism (−839 C > T) at the dopamine transporter gene is associated with attention deficit/hyperactivity disorder in Brazilian children. Am J Med Genet B Neuropsychiatr Genet 144:215–219. https://doi.org/10.1002/ajmg.b.30428
Ghosh P, Maitra S, Saha T, Sinha S, Mukhopadhyay K (2017) Functional genetic polymorphisms in dopaminergic transporters: association with ADHD traits in the Indian probands. Metagene 11:117–122. https://doi.org/10.1016/j.mgene.2016.12.009
Giros B, Jaber M, Jones SR, Wightman RM, Caron MG (1996) Hyperlocomotion and indifference to cocaine and amphetamine in mice lacking the dopamine transporter. Nature 379:606–612. https://doi.org/10.1038/379606a0
Greenwood TA, Kelsoe JR (2003) Promoter and intronic variants affect the transcriptional regulation of the human dopamine transporter gene. Genomics 82:511–520. https://doi.org/10.1016/s0888-7543(03)00142-3
Guindalini C, Howard M, Haddley K, Laranjeira R, Collier D, Ammar N, Craig I, O'Gara C, Bubb VJ, Greenwood T, Kelsoe J, Asherson P, Murray RM, Castelo A, Quinn JP, Vallada H, Breen G (2006) A dopamine transporter gene functional variant associated with cocaine abuse in a Brazilian sample. Proc Natl Acad Sci U S A 103:4552–4557. https://doi.org/10.1073/pnas.0504789103
Hahn LW, Ritchie MD, Moore JH (2003) Multifactor dimensionality reduction software for detecting gene-gene and gene-environment interactions. Bioinformatics 19:376–382. https://doi.org/10.1093/bioinformatics/btf869
Hellings JA, Arnold LE, Han JC (2017) Dopamine antagonists for treatment resistance in autism spectrum disorders: review and focus on BDNF stimulators loxapine and amitriptyline. Expert Opin Pharmacother 18:581–588. https://doi.org/10.1080/14656566.2017.1308483
Hesapcioglu S, Ceylan MF, Kasak M, Sen CP (2020) Olanzapine, risperidone, and aripiprazole use in children and adolescents with autism spectrum disorders. Res Autism Spectr Disord 72:101520. https://doi.org/10.1016/j.rasd.2020.101520
Hill M, Anney RJ, Gill M, Hawi Z (2010) Functional analysis of intron 8 and 3' UTR variable number of tandem repeats of SLC6A3: differential activity of intron 8 variants. Pharmacogenomics J 10:442–447. https://doi.org/10.1038/tpj.2009.66
Hosenbocus S, Chahal R (2012) A review of executive function deficits and pharmacological management in children and adolescents. J Can Acad Child Adolesc Psychiatry 21:223–229
Huang SY, Chen HK, Ma KH, Shy MJ, Chen JH, Lin W, Lu RB (2010) Association of promoter variants of human dopamine transporter gene with schizophrenia in Han Chinese. Schizophr Res 116:68–74. https://doi.org/10.1016/j.schres.2009.10.004
Kaluzna-Czaplinska J, Socha E, Rynkowski J (2010) Determination of homovanillic acid and vanillylmandelic acid in urine of autistic children by gas chromatography/mass spectrometry. Med Sci Monit 16:CR445–CR450
King BH, Lord C (2011) Is schizophrenia on the autism spectrum? Brain Res 1380:34–41. https://doi.org/10.1016/j.brainres.2010.11.031
Kogan MD, Blumberg SJ, Schieve LA, Boyle CA, Perrin JM, Ghandour RM, Singh GK, Strickland BB, Trevathan E, van Dyck PC (2009) Prevalence of parent-reported diagnosis of autism spectrum disorder among children in the US, 2007. Pediatrics 124:1395–1403. https://doi.org/10.1542/peds.2009-1522
Langen M, Leemans A, Johnston P, Ecker C, Daly E, Murphy CM, Dell'acqua F, Durston S, AIMS Consortium, Murphy DG (2012) Frontostriatal circuitry and inhibitory control in autism: findings from diffusion tensor imaging tractography. Cortex 48:183–193. https://doi.org/10.1016/j.cortex.2011.05.018
Lewis MH, Tanimura Y, Lee LW, Bodfish JW (2007) Animal models of restricted repetitive behaviour in autism. Behav Brain Res. 176(1):66–74. https://doi.org/10.1016/j.bbr.2006.08.023
Maitra S, Sarkar K, Ghosh P, Karmakar A, Bhattacharjee A, Sinha S, Mukhopadhyay K (2014) Potential contribution of dopaminergic gene variants in ADHD core traits and co-morbidity: a study on eastern Indian probands. Cell Mol Neurobiol 34:549–564. https://doi.org/10.1007/s10571-014-0038-9
Mannion A, Leader G (2016) An investigation of comorbid psychological disorders, sleep problems, gastrointestinal symptoms and epilepsy in children and adolescents with autism spectrum disorder: a two year follow-up. Res Autism Spectr Disord 22:20–33. https://doi.org/10.1016/j.rasd.2015.11.002
Mill J, Asherson P, Browes C, D'Souza U, Craig I (2002) Expression of the dopamine transporter gene is regulated by the 3' UTR VNTR: evidence from brain and lymphocytes using quantitative RT-PCR. Am J Med Genet 114:975–979. https://doi.org/10.1002/ajmg.b.10948
Miller SA, Dykes DD, Polesky HF (1988) A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res 16:1215. https://doi.org/10.1093/nar/16.3.1215
Naaijen J, Bralten J, Poelmans G, IMAGE consortium, Glennon JC, Franke B, Buitelaar JK (2017) Glutamatergic and GABAergic gene sets in attention-deficit/hyperactivity disorder: association to overlapping traits in ADHD and autism. Transl Psychiatry 7:e999. https://doi.org/10.1038/tp.2016.273
Nakamura K, Sekine Y, Ouchi Y, Tsujii M, Yoshikawa E, Futatsubashi M, Tsuchiya KJ, Sugihara G, Iwata Y, Suzuki K, Matsuzaki H, Suda S, Sugiyama T, Takei N, Mori N (2010) Brain serotonin and dopamine transporter bindings in adults with high-functioning autism. Arch Gen Psychiatry 67:59–68. https://doi.org/10.1001/archgenpsychiatry.2009.137
Ouellet-Morin I, Wigg KG, Feng Y, Dionne G, Robaey P, Brendgen M, Vitaro F, Simard L, Schachar R, Tremblay RE, Pérusse D, Boivin M, Barr CL (2008) Association of the dopamine transporter gene and ADHD symptoms in a Canadian population-based sample of same-age twins. Am J Med Genet B Neuropsychiatr Genet 147B:1442–1449. https://doi.org/10.1002/ajmg.b.30677
Paval DA (2017) Dopamine hypothesis of autism spectrum disorder. DevNeurosci 39:355–360. https://doi.org/10.1159/000478725
Pinsonneault JK, Han DD, Burdick KE, Kataki M, Bertolino A, Malhotra AK, Gu HH, Sadee W (2011) Dopamine transporter gene variant affecting expression in human brain is associated with bipolar disorder. Neuropsychopharmacology 36:1644–1655. https://doi.org/10.1038/npp.2011.45
Poovathinal SA, Anitha A, Thomas R, Kaniamattam M, Melempatt N, Anilkumar A, Meena M (2016) Prevalence of autism spectrum disorders in a semiurban community in South India. Ann Epidemiol 26:663–665. https://doi.org/10.1016/j.annepidem.2016.07.003
Ray A, Maitra S, Chatterjee M, Ghosh P, Karmakar A, Sinha S, Mukhopadhyay K (2017) Dimorphic association of dopaminergic transporter gene variants with treatment outcome: pilot study in Indian ADHD probands. Meta Gene 11:64–69. https://doi.org/10.1016/j.mgene.2016.11.007
Rudra A, Belmonte MK, Soni PK, Banerjee S, Mukerji S, Chakrabarti B (2017) Prevalence of autism spectrum disorder and autistic symptoms in a school-based cohort of children in Kolkata, India. Autism Res 10:1597–1605. https://doi.org/10.1002/aur.1812
Simonoff E, Pickles A, Charman T, Chandler S, Loucas T, Baird G (2008) Psychiatric disorders in children with autism spectrum disorders: prevalence, comorbidity, and associated factors in a population-derived sample. J Am Acad Child Adolesc Psychiatry. 47(8):921–929. https://doi.org/10.1097/CHI.0b013e318179964f
Schopler E, Reichler RJ, Renner BR (1988) The Childhood Autism Rating Scale (CARS). Los Angeles, CA: Western Psychological Services
Schopler E, Van Bourgondien ME, Wellman GJ, Love SR (2010) Childhood Autism Rating Scale. 2nd ed. Los Angeles, CA: Western Psychological Services
van de Giessen E, de Win MM, Tanck MW, van den Brink W, Baas F, Booij J (2009) Striatal dopamine transporter availability associated with polymorphisms in the dopamine transporter gene SLC6A3. Journal of Nuclear Medicine 50(1):45–52. https://doi.org/10.2967/jnumed.108.053652
Xu X, Mill J, Sun B, Chen CK, Huang YS, Wu YY, et al. (2009) Association study of promoter polymorphisms at the dopamine transporter gene in attention deficit hyperactivity disorder. BMC Psychiatry, 9:3. https://doi.org/10.1186/1471-244x-9-3
Acknowledgements
We are thankful to the study participants for their voluntary involvement in the work.
Funding
This study was funded by the host Institute. Authors received no financial support for authorship, and/or publication of this article.
Author information
Authors and Affiliations
Contributions
All authors contributed distinctly in the presented work. Study designing, genotyping, data analysis, and manuscript draft preparation was performed by Sharmistha Saha. Mahasweta Chatterjee assisted in genotyping and data interpretation. Sayanti Shom helped in genotyping. Swagata Sinha assessed and recruited ASD probands. Kanchan Mukhopadhyay conceptualized and supervised study designing, its execution, and edited the final manuscript. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
All procedures performed in studies involving human participants were approved by the institutional Human Ethical Committee (No. PR-005-19) following the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Consent to participate
Written informed consent was obtained from the parents for participation in the study.
Consent for publication
Written informed consent was obtained from the parents for anonymous publication.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(PDF 282 kb)
Rights and permissions
About this article
Cite this article
Saha, S., Chatterjee, M., Shom, S. et al. Functional SLC6A3 polymorphisms differentially affect autism spectrum disorder severity: a study on Indian subjects. Metab Brain Dis 37, 397–410 (2022). https://doi.org/10.1007/s11011-021-00876-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-021-00876-4