Abstract
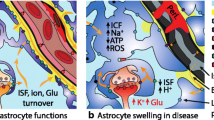
Hepatic encephalopathy (HE) may occur in patients with liver failure. The most critical pathophysiologic mechanism of HE is cerebral edema following systemic hyperammonemia. The dysfunctional liver cannot eliminate circulatory ammonia, so its plasma and brain levels rise sharply. Astrocytes, the only cells that are responsible for ammonia detoxification in the brain, are dynamic cells with unique phenotypic properties that enable them to respond to small changes in their environment. Any pathological changes in astrocytes may cause neurological disturbances such as HE. Astrocyte swelling is the leading cause of cerebral edema, which may cause brain herniation and death by increasing intracranial pressure. Various factors may have a role in astrocyte swelling. However, the exact molecular mechanism of astrocyte swelling is not fully understood. This article discusses the possible mechanisms of astrocyte swelling which related to hyperammonia, including the possible roles of molecules like glutamine, lactate, aquaporin-4 water channel, 18 KDa translocator protein, glial fibrillary acidic protein, alanine, glutathione, toll-like receptor 4, epidermal growth factor receptor, glutamate, and manganese, as well as inflammation, oxidative stress, mitochondrial permeability transition, ATP depletion, and astrocyte senescence. All these agents and factors may be targeted in therapeutic approaches to HE.




Similar content being viewed by others
Data availability
No datasets were generated or analyzed during the current study.
Abbreviations
- ALF:
-
Acute liver failure
- AQP4:
-
Aquaporin-4 water channel
- ATP:
-
Adenosine triphosphate
- BBB:
-
Blood-brain barrier
- cGMP:
-
Cyclic guanosin monophosphate
- CLF:
-
Chronic liver failure
- CNS:
-
Central nervous system
- EAAT:
-
Excitatory amino acid transporters
- EGFR:
-
Epidermal growth factor receptor
- GFAP:
-
Glial fibrillary acidic protein
- Gln:
-
Glutamine
- Glu:
-
Glutamate
- GS:
-
Glutamine synthetase
- HE:
-
Hepatic encephalopathy
- HMGB-1:
-
High-mobility group box protein-1
- ICH:
-
Intracranial hypertension
- ICP:
-
Intracranial pressure
- (INFϒ):
-
interferon-gamma
- IL-1:
-
Interleukin-1
- IL-1β:
-
interleukin 1 beta
- IL-6:
-
Interleukin-6
- MAPKs:
-
Mitogen-activated protein kinases
- MPT:
-
Mitochondrial permeability transition
- MSO:
-
Methionine sulfoximine
- NF-kB:
-
Nuclear factor-kappaB
- NMDA:
-
N-methyl-D-aspartate
- NO:
-
Nitric oxide
- PAG:
-
Phosphate activated glutaminase
- PCA/S:
-
Portocaval anastomosis/shunt
- PTBRs:
-
Peripheral type benzodiazepine receptors
- RNS:
-
Reactive nitrogen species
- ROS:
-
Reactive oxygen species
- Sur1-TRPM4:
-
Sulfonyl urea receptor 1- transient receptor potential melastatin 4
- TLR4:
-
Toll-like receptor 4
- TNFα:
-
Tumor necrosis factor-alpha
- TSPO:
-
18 KDa Translocator protein
References
Abdel-Khalek EE, El-Fakhry A, Helaly M, Hamed M, Elbaz O (2011) Systemic inflammatory response syndrome in patients with liver cirrhosis. Arab J Gastroenterol: the official publication of the Pan-Arab Association of Gastroenterology 12:173–177. https://doi.org/10.1016/j.ajg.2011.11.006
Agre P, Nielsen S, Ottersen OP (2004) Towards a molecular understanding of water homeostasis in the brain. Neuroscience 129:849–850. https://doi.org/10.1016/j.neuroscience.2004.10.001
Agusti A, Cauli O, Rodrigo R, Llansola M, Hernandez-Rabaza V, Felipo V (2011) p38 MAP kinase is a therapeutic target for hepatic encephalopathy in rats with portacaval shunts. Gut 60:1572–1579. https://doi.org/10.1136/gut.2010.236083
Ahboucha S, Desjardins P, Chatauret N, Pomier-Layrargues G, Butterworth RF (2003) Normal coupling of brain benzodiazepine and neurosteroid modulatory sites on the GABA-A receptor complex in human hepatic encephalopathy. Neurochem Int 43:551–556
Ahboucha S, Layrargues GP, Mamer O, Butterworth RF (2005) Increased brain concentrations of a neuroinhibitory steroid in human hepatic encephalopathy. Ann Neurol 58:169–170. https://doi.org/10.1002/ana.20534
Alavian KN et al (2014) An uncoupling channel within the c-subunit ring of the F1FO ATP synthase is the mitochondrial permeability transition pore. Proc Natl Acad Sci U S A 111:10580–10585. https://doi.org/10.1073/pnas.1401591111
Albrecht J, Norenberg MD (2006) Glutamine: a Trojan horse in ammonia neurotoxicity. Hepatology (Baltimore, Md) 44:788–794. https://doi.org/10.1002/hep.21357
Allaman I, Belanger M, Magistretti PJ (2011) Astrocyte-neuron metabolic relationships: for better and for worse. Trends Neurosci 34:76–87. https://doi.org/10.1016/j.tins.2010.12.001
Andre P, Villain F (2017) Free radical scavenging properties of mannitol and its role as a constituent of hyaluronic acid fillers: a literature review. Int J Cosmet Sci 39:355–360. https://doi.org/10.1111/ics.12386
Arima H, Yamamoto N, Sobue K, Umenishi F, Tada T, Katsuya H, Asai K (2003) Hyperosmolar mannitol simulates expression of aquaporins 4 and 9 through a p38 mitogen-activated protein kinase-dependent pathway in rat astrocytes. J Biol Chem 278:44525–44534. https://doi.org/10.1074/jbc.M304368200
Back A, Tupper KY, Bai T, Chiranand P, Goldenberg FD, Frank JI, Brorson JR (2011) Ammonia-induced brain swelling and neurotoxicity in an organotypic slice model. Neurol Res 33:1100–1108. https://doi.org/10.1179/1743132811y.0000000046
Bai G, Rama Rao KV, Murthy CR, Panickar KS, Jayakumar AR, Norenberg MD (2001) Ammonia induces the mitochondrial permeability transition in primary cultures of rat astrocytes. J Neurosci Res 66:981–991. https://doi.org/10.1002/jnr.10056
Bak LK, Schousboe A, Waagepetersen HS (2006) The glutamate/GABA-glutamine cycle: aspects of transport, neurotransmitter homeostasis and ammonia transfer. J Neurochem 98:641–653. https://doi.org/10.1111/j.1471-4159.2006.03913.x
Barnard EA, Bilbe G, Houamed K, Moss SJ, Van Renterghem C, Smart TG (1987) Functional expression in the Xenopus oocyte of messenger ribonucleic acids encoding brain neurotransmitter receptors: further characterisation of the implanted GABA receptor. Neuropharmacology 26:837–844
Belanger M, Magistretti PJ (2009) The role of astroglia in neuroprotection. Dialogues Clin Neurosci 11:281–295
Belanger M, Desjardins P, Chatauret N, Butterworth RF (2002) Loss of expression of glial fibrillary acidic protein in acute hyperammonemia. Neurochem Int 41:155–160
Bemeur C, Qu H, Desjardins P, Butterworth RF (2010a) IL-1 or TNF receptor gene deletion delays onset of encephalopathy and attenuates brain edema in experimental acute liver failure. Neurochem Int 56:213–215. https://doi.org/10.1016/j.neuint.2009.11.010
Bemeur C, Vaquero J, Desjardins P, Butterworth RF (2010b) N-acetylcysteine attenuates cerebral complications of non-acetaminophen-induced acute liver failure in mice: antioxidant and anti-inflammatory mechanisms. Metab Brain Dis 25:241–249. https://doi.org/10.1007/s11011-010-9201-2
Bemeur C, Cudalbu C, Dam G, Thrane AS, Cooper AJ, Rose CF (2016) Brain edema: a valid endpoint for measuring hepatic encephalopathy? Metab Brain Dis 31:1249–1258. https://doi.org/10.1007/s11011-016-9843-9
Bender AS, Norenberg MD (1998) Effect of benzodiazepines and neurosteroids on ammonia-induced swelling in cultured astrocytes. J Neurosci Res 54:673–680. https://doi.org/10.1002/(sici)1097-4547(19981201)54:5<673::aid-jnr12>3.0.co;2-p
Bernal W, Donaldson N, Wyncoll D, Wendon J (2002) Blood lactate as an early predictor of outcome in paracetamol-induced acute liver failure: a cohort study. Lancet (London, England) 359:558–563. https://doi.org/10.1016/s0140-6736(02)07743-7
Bernal W, Auzinger G, Sizer E, Wendon J (2008) Intensive care management of acute liver failure. Semin Liver Dis 28:188–200. https://doi.org/10.1055/s-2008-1073118
Blei AT (1996) Brain edema and intracranial hypertension in acute liver failure. In: Williams R, Lee WM (eds) Acute liver failure. Cambridge University Press, Cambridge, pp 144–157. https://doi.org/10.1017/CBO9780511575044.014
Brahma B, Forman RE, Stewart EE, Nicholson C, Rice ME (2000) Ascorbate inhibits edema in brain slices. J Neurochem 74:1263–1270
Cagnin A, Taylor-Robinson SD, Forton DM, Banati RB (2006) In vivo imaging of cerebral “peripheral benzodiazepine binding sites” in patients with hepatic encephalopathy. Gut 55:547–553. https://doi.org/10.1136/gut.2005.075051
Cauli O, Rodrigo R, Piedrafita B, Boix J, Felipo V (2007) Inflammation and hepatic encephalopathy: ibuprofen restores learning ability in rats with portacaval shunts. Hepatology (Baltimore, md) 46:514–519. https://doi.org/10.1002/hep.21734
Cauli O, Rodrigo R, Piedrafita B, Llansola M, Mansouri MT, Felipo V (2009) Neuroinflammation contributes to hypokinesia in rats with hepatic encephalopathy: ibuprofen restores its motor activity. J Neurosci Res 87:1369–1374. https://doi.org/10.1002/jnr.21947
Chan H, Hazell AS, Desjardins P, Butterworth RF (2000) Effects of ammonia on glutamate transporter (GLAST) protein and mRNA in cultured rat cortical astrocytes. Neurochem Int 37:243–248
Chavarria L, Romero-Giménez J, Monteagudo E, Lope-Piedrafita S, Cordoba J (2015) Real-time assessment of 13C metabolism reveals an early lactate increase in the brain of rats with acute liver failure. NMR Biomed 28:17–23. https://doi.org/10.1002/nbm.3226
Chen M, Dong Y, Simard JM (2003) Functional coupling between sulfonylurea receptor type 1 and a nonselective cation channel in reactive astrocytes from adult rat brain. J Neurosci 23:8568–8577
Chipuk JE, Bouchier-Hayes L, Green DR (2006) Mitochondrial outer membrane permeabilization during apoptosis: the innocent bystander scenario. Cell Death Differ 13:1396–1402. https://doi.org/10.1038/sj.cdd.4401963
Cooper AJ, Mora SN, Cruz NF, Gelbard AS (1985) Cerebral ammonia metabolism in hyperammonemic rats. J Neurochem 44:1716–1723
Crompton M (1999) The mitochondrial permeability transition pore and its role in cell death. Biochem J 341(Pt 2):233–249
Dai H, Song D, Xu J, Li B, Hertz L, Peng L (2013) Ammonia-induced Na,K-ATPase/ouabain-mediated EGF receptor transactivation, MAPK/ERK and PI3K/AKT signaling and ROS formation cause astrocyte swelling. Neurochem Int 63:610–625. https://doi.org/10.1016/j.neuint.2013.09.005
Dai H, Jia G, Wang W, Liang C, Han S, Chu M, Mei X (2017) Genistein inhibited ammonia induced astrocyte swelling by inhibiting NF-kappaB activation-mediated nitric oxide formation. Metab Brain Dis 32:841–848. https://doi.org/10.1007/s11011-017-9975-6
de Knegt RJ, Schalm SW, van der Rijt CC, Fekkes D, Dalm E, Hekking-Weyma I (1994) Extracellular brain glutamate during acute liver failure and during acute hyperammonemia simulating acute liver failure: an experimental study based on in vivo brain dialysis. World J Hepatol 20:19–26
de Vries HE, Blom-Roosemalen MCM, Mv O, de Boer AG, van TJC B, Breimer DD, Kuiper J (1996) The influence of cytokines on the integrity of the blood-brain barrier in vitro. J Neuroimmunol 64:37–43. https://doi.org/10.1016/0165-5728(95)00148-4
Dejong CH, Kampman MT, Deutz NE, Soeters PB (1992) Cerebral cortex ammonia and glutamine metabolism during liver insufficiency-induced hyperammonemia in the rat. J Neurochem 59:1071–1079
Ding M, Eliasson C, Betsholtz C, Hamberger A, Pekny M (1998) Altered taurine release following hypotonic stress in astrocytes from mice deficient for GFAP and vimentin. Brain Res Mol Brain Res 62:77–81
Du H, Yan SS (2010) Mitochondrial permeability transition pore in Alzheimer's disease: cyclophilin D and amyloid beta. Biochim Biophys Acta 1802:198–204. https://doi.org/10.1016/j.bbadis.2009.07.005
Durward QJ, Del Maestro RF, Amacher AL, Farrar JK (1983) The influence of systemic arterial pressure and intracranial pressure on the development of cerebral vasogenic edema. J Neurosurg 59:803–809. https://doi.org/10.3171/jns.1983.59.5.0803
Eefsen M, Jelnes P, Schmidt LE, Vainer B, Bisgaard HC, Larsen FS (2010) Brain expression of the water channels aquaporin-1 and -4 in mice with acute liver injury, hyperammonemia and brain edema. Metab Brain Dis 25:315–323. https://doi.org/10.1007/s11011-010-9213-y
Forbes A, Alexander GJ, O'Grady JG, Keays R, Gullan R, Dawling S, Williams R (1989) Thiopental infusion in the treatment of intracranial hypertension complicating fulminant hepatic failure. Hepatology (Baltimore, Md) 10:306–310. https://doi.org/10.1002/hep.1840100309
Francavilla A et al (1989) A dog model for acetaminophen-induced fulminant hepatic failure. Gastroenterology 96:470–478. https://doi.org/10.1016/0016-5085(89)91573-4
Gorg B, Foster N, Reinehr R, Bidmon HJ, Hongen A, Haussinger D, Schliess F (2003) Benzodiazepine-induced protein tyrosine nitration in rat astrocytes. Hepatology (Baltimore, Md) 37:334–342. https://doi.org/10.1053/jhep.2003.50061
Gorg B, Bidmon HJ, Keitel V, Foster N, Goerlich R, Schliess F, Haussinger D (2006) Inflammatory cytokines induce protein tyrosine nitration in rat astrocytes. Arch Biochem Biophys 449:104–114. https://doi.org/10.1016/j.abb.2006.02.012
Gorg B, Karababa A, Shafigullina A, Bidmon HJ, Haussinger D (2015) Ammonia-induced senescence in cultured rat astrocytes and in human cerebral cortex in hepatic encephalopathy. Glia 63:37–50. https://doi.org/10.1002/glia.22731
Görg B et al (2019) O-GlcNAcylation-dependent upregulation of HO1 triggers ammonia-induced oxidative stress and senescence in hepatic encephalopathy. J Hepatol. https://doi.org/10.1016/j.jhep.2019.06.020
Gow AG, Marques AI, Yool DA, Duncan A, Mellanby RJ (2010) Whole blood manganese concentrations in dogs with congenital portosystemic shunts. J Vet Intern Med 24:90–96. https://doi.org/10.1111/j.1939-1676.2009.0408.x
Grande RG, Pérez MJ (2017) Liver transplantation in acute liver failure: indications and outcome. In: Liver Research and Clinical Management. IntechOpen,
Grant S et al (2018) Direct comparison of the Thioacetamide and Azoxymethane models of type a hepatic encephalopathy in mice. Gene Expr 18:171–185. https://doi.org/10.3727/105221618x15287315176503
Guo Q, Sayeed I, Baronne LM, Hoffman SW, Guennoun R, Stein DG (2006) Progesterone administration modulates AQP4 expression and edema after traumatic brain injury in male rats. Exp Neurol 198:469–478. https://doi.org/10.1016/j.expneurol.2005.12.013
Guttner J, Heinecke H, Kuchler W, Werner W (1973) Immunosuppressive effects of barbituric acid derivatives. II. Effect of 1.3-bis(piperidinomethyl)-5-ethyl-5-phenyl-barbituric acid on humoral and cell-mediated immunity in mice. Chemotherapy 19:305–313. https://doi.org/10.1159/000221469
Hadjihambi A, Khetan V, Jalan R (2014) Pharmacotherapy for hyperammonemia. Expert Opin Pharmacother 15:1685–1695. https://doi.org/10.1517/14656566.2014.931372
Haghighat N, McCandless DW (1997) Effect of ammonium chloride on energy metabolism of astrocytes and C6-glioma cells in vitro. Metab Brain Dis 12:287–298
Halestrap AP, Woodfield KY, Connern CP (1997) Oxidative stress, thiol reagents, and membrane potential modulate the mitochondrial permeability transition by affecting nucleotide binding to the adenine nucleotide translocase. J Biol Chem 272:3346–3354
Harrison PM, Keays R, Bray GP, Alexander GJ, Williams R (1990) Improved outcome of paracetamol-induced fulminant hepatic failure by late administration of acetylcysteine. Lancet (London, England) 335:1572–1573. https://doi.org/10.1016/0140-6736(90)91388-q
Haussinger D, Sies H, Gerok W (1985) Functional hepatocyte heterogeneity in ammonia metabolism. J Hepatol 1:3–14
Haworth RA, Hunter DR (1979) The Ca2+−induced membrane transition in mitochondria. II. Nature Ca2+ trigger site. Arch Biochem Biophys 195:460–467
Hazell AS, Desjardins P, Butterworth RF (1999) Chronic exposure of rat primary astrocyte cultures to manganese results in increased binding sites for the 'peripheral-type' benzodiazepine receptor ligand 3H-PK 11195. Neurosci Lett 271:5–8
Henry CJ et al (2008) Minocycline attenuates lipopolysaccharide (LPS)-induced neuroinflammation, sickness behavior, and anhedonia. J Neuroinflammation 5:15. https://doi.org/10.1186/1742-2094-5-15
Hilgier W et al. (1999) Changes in the extracellular profiles of neuroactive amino acids in the rat striatum at the symptomatic stage of hepatic failure 56. https://doi.org/10.1002/(SICI)1097-4547(19990401)56:1<76::AID-JNR10>3.0.CO;2-Y
Hunter DR, Haworth RA, Southard JH (1976) Relationship between configuration, function, and permeability in calcium-treated mitochondria. J Biol Chem 251:5069–5077
Illarionova NB, Gunnarson E, Li Y, Brismar H, Bondar A, Zelenin S, Aperia A (2010) Functional and molecular interactions between aquaporins and Na,K-ATPase. Neuroscience 168:915–925. https://doi.org/10.1016/j.neuroscience.2009.11.062
Itzhak Y, Norenberg MD (1994) Ammonia-induced upregulation of peripheral-type benzodiazepine receptors in cultured astrocytes labeled with [3H]PK 11195. Neurosci Lett 177:35–38
Itzhak Y, Baker L, Norenberg MD (1993) Characterization of the peripheral-type benzodiazepine receptors in cultured astrocytes: evidence for multiplicity. Glia 9:211–218. https://doi.org/10.1002/glia.440090306
Itzhak Y, Roig-Cantisano A, Dombro RS, Norenberg MD (1995) Acute liver failure and hyperammonemia increase peripheral-type benzodiazepine receptor binding and pregnenolone synthesis in mouse brain. Brain Res 705:345–348
Jacob S, Khan A, Jacobs ER, Kandiah P, Nanchal R (2009) Prolonged hypothermia as a bridge to recovery for cerebral edema and intracranial hypertension associated with fulminant hepatic failure. Neurocrit Care 11:242–246. https://doi.org/10.1007/s12028-009-9266-0
Jalan R, Kapoor D (2003) Enhanced renal ammonia excretion following volume expansion in patients with well compensated cirrhosis of the liver. Gut 52:1041–1045. https://doi.org/10.1136/gut.52.7.1041
Jalan R et al (2003) Moderate hypothermia prevents cerebral hyperemia and increase in intracranial pressure in patients undergoing liver transplantation for acute liver failure. Transplantation 75:2034–2039. https://doi.org/10.1097/01.tp.0000066240.42113.ff
Jayakumar AR, Panickar KS, Norenberg MD (2002) Effects on free radical generation by ligands of the peripheral benzodiazepine receptor in cultured neural cells. J Neurosci 83:1226–1234. https://doi.org/10.1046/j.1471-4159.2002.01261.x
Jayakumar AR, Rama Rao KV, Kalaiselvi P, Norenberg MD (2004a) Combined effects of ammonia and manganese on astrocytes in culture. Neurochem Res 29:2051–2056
Jayakumar AR, Rama Rao KV, Schousboe A, Norenberg MD (2004b) Glutamine-induced free radical production in cultured astrocytes. Glia 46:296–301. https://doi.org/10.1002/glia.20003
Jayakumar AR, Panickar KS, Murthy Ch R, Norenberg MD (2006a) Oxidative stress and mitogen-activated protein kinase phosphorylation mediate ammonia-induced cell swelling and glutamate uptake inhibition in cultured astrocytes. J Neurosci 26:4774–4784. https://doi.org/10.1523/jneurosci.0120-06.2006
Jayakumar AR, Rao KV, Murthy Ch R, Norenberg MD (2006b) Glutamine in the mechanism of ammonia-induced astrocyte swelling. Neurochem Int 48:623–628. https://doi.org/10.1016/j.neuint.2005.11.017
Jayakumar AR, Liu M, Moriyama M, Ramakrishnan R, Forbush B 3rd, Reddy PV, Norenberg MD (2008) Na-K-cl Cotransporter-1 in the mechanism of ammonia-induced astrocyte swelling. J Biol Chem 283:33874–33882. https://doi.org/10.1074/jbc.M804016200
Jayakumar AR, Tong XY, Ospel J, Norenberg MD (2012) Role of cerebral endothelial cells in the astrocyte swelling and brain edema associated with acute hepatic encephalopathy. Neuroscience 218:305–316. https://doi.org/10.1016/j.neuroscience.2012.05.006
Jayakumar AR, Tong XY, Curtis KM, Ruiz-Cordero R, Abreu MT, Norenberg MD (2014) Increased toll-like receptor 4 in cerebral endothelial cells contributes to the astrocyte swelling and brain edema in acute hepatic encephalopathy. J Neurochem 128:890–903. https://doi.org/10.1111/jnc.12516
Jha SK (2003) Cerebral edema and its management. Med J Armed Forces India 59:326–331. https://doi.org/10.1016/s0377-1237(03)80147-8
Jiang W, Desjardins P, Butterworth RF (2009a) Cerebral inflammation contributes to encephalopathy and brain edema in acute liver failure: protective effect of minocycline. J Neurochem 109:485–493. https://doi.org/10.1111/j.1471-4159.2009.05981.x
Jiang W, Desjardins P, Butterworth RF (2009b) Direct evidence for central proinflammatory mechanisms in rats with experimental acute liver failure: protective effect of hypothermia. J Cereb Blood Flow Metab 29:944–952. https://doi.org/10.1038/jcbfm.2009.18
Kakisaka K, Kataoka K, Suzuki Y, Kuroda H, Takikawa Y (2017) Appropriate timing to start and optimal response evaluation of high-dose corticosteroid therapy for patients with acute liver failure. J Gastroenterol 52:977–985. https://doi.org/10.1007/s00535-017-1306-5
Kala G, Kumarathasan R, Peng L, Leenen FH, Hertz L (2000) Stimulation of Na+,K+-ATPase activity, increase in potassium uptake, and enhanced production of ouabain-like compounds in ammonia-treated mouse astrocytes. Neurochem Int 36:203–211
Karababa A, Görg B, Schütz E, Bidmon HJ, Häussinger D (2017) Ammonia affects iron homeostasis in cultured rat astrocytes and in human cerebral cortex in hepatic encephalopathy. J Clin Exp Hepatol 7:S3–S4. https://doi.org/10.1016/j.jceh.2017.01.006
Kato M, Hughes RD, Keays RT, Williams R (1992) Electron microscopic study of brain capillaries in cerebral edema from fulminant hepatic failure. Hepatology (Baltimore, Md) 15:1060–1066
Khakh BS, Sofroniew MV (2015) Diversity of astrocyte functions and phenotypes in neural circuits. Nat Neurosci 18:942–952. https://doi.org/10.1038/nn.4043
Kintner DB et al (2004) Increased tolerance to oxygen and glucose deprivation in astrocytes from Na(+)/H(+) exchanger isoform 1 null mice. Am J Phys Cell Physiol 287:C12–C21. https://doi.org/10.1152/ajpcell.00560.2003
Klatzo I (1967) Neuropathological aspects of brain edema. J Neuropathol Exp Neurol 26:1–14. https://doi.org/10.1097/00005072-196701000-00001
Knecht K, Michalak A, Rose C, Rothstein JD, Butterworth RF (1997) Decreased glutamate transporter (GLT-1) expression in frontal cortex of rats with acute liver failure. Neurosci Lett 229:201–203
Kosenko E, Kaminsky Y, Grau E, Minana MD, Marcaida G, Grisolia S, Felipo V (1994) Brain ATP depletion induced by acute ammonia intoxication in rats is mediated by activation of the NMDA receptor and Na+,K(+)-ATPase. J Neurochem 63:2172–2178
Kosenko E, Kaminsky Y, Grau E, Minana MD, Grisolia S, Felipo V (1995) Nitroarginine, an inhibitor of nitric oxide synthetase, attenuates ammonia toxicity and ammonia-induced alterations in brain metabolism. Neurochem Res 20:451–456
Kosenko E, Felipo V, Montoliu C, Grisolia S, Kaminsky Y (1997) Effects of acute hyperammonemia in vivo on oxidative metabolism in nonsynaptic rat brain mitochondria. Metab Brain Dis 12:69–82
Kosenko E, Kaminsky Y, Stavroskaya IG, Felipo V (2000) Alteration of mitochondrial calcium homeostasis by ammonia-induced activation of NMDA receptors in rat brain in vivo. Brain Res 880:139–146
Lachmann V, Gorg B, Bidmon HJ, Keitel V, Haussinger D (2013) Precipitants of hepatic encephalopathy induce rapid astrocyte swelling in an oxidative stress dependent manner. Arch Biochem Biophys 536:143–151. https://doi.org/10.1016/j.abb.2013.05.004
Lavoie J, Layrargues GP, Butterworth RF (1990) Increased densities of peripheral-type benzodiazepine receptors in brain autopsy samples from cirrhotic patients with hepatic encephalopathy. Hepatology (Baltimore, Md) 11:874–878
Lee WM et al (2009) Intravenous N-acetylcysteine improves transplant-free survival in early stage non-acetaminophen acute liver failure. Gastroenterology 137:856–864, 864.e851. https://doi.org/10.1053/j.gastro.2009.06.006
Lemasters JJ, Theruvath TP, Zhong Z, Nieminen AL (2009) Mitochondrial calcium and the permeability transition in cell death. Biochim Biophys Acta 1787:1395–1401. https://doi.org/10.1016/j.bbabio.2009.06.009
Leong DK, Therrien G, Swain MS, Butterworth RF (1994) Densities of binding sites for the “peripheral-type” benzodiazepine receptor ligand3H-PK11195 are increased in brain 24 hours following portacaval anastomosis. Metab Brain Dis 9:267–273. https://doi.org/10.1007/bf01991200
Licinio J, Wong ML (1997) Pathways and mechanisms for cytokine signaling of the central nervous system. J Clin Investig 100:2941–2947. https://doi.org/10.1172/jci119846
Liu L, Ivanov AV, Gable ME, Jolivel F, Morrill GA, Askari A (2011) Comparative properties of caveolar and noncaveolar preparations of kidney Na+/K+-ATPase. Biochemistry 50:8664–8673. https://doi.org/10.1021/bi2009008
Lockwood AH, Yap EWH, Wong W-H (1991) Cerebral Ammonia Metabolism in Patients with Severe Liver Disease and Minimal Hepatic Encephalopathy. J Cereb Blood Flow Metab 11:337–341. https://doi.org/10.1038/jcbfm.1991.67
Manley GT et al (2000) Aquaporin-4 deletion in mice reduces brain edema after acute water intoxication and ischemic stroke. Nat Med 6:159–163. https://doi.org/10.1038/72256
Marcaida G, Felipo V, Hermenegildo C, Minana MD, Grisolia S (1992) Acute ammonia toxicity is mediated by the NMDA type of glutamate receptors. FEBS Lett 296:67–68. https://doi.org/10.1016/0014-5793(92)80404-5
Masago K, Kihara Y, Yanagida K, Hamano F, Nakagawa S, Niwa M, Shimizu T (2018) Lysophosphatidic acid receptor, LPA6, regulates endothelial blood-brain barrier function: implication for hepatic encephalopathy. Biochem Biophys Res Commun 501:1048–1054. https://doi.org/10.1016/j.bbrc.2018.05.106
Master S, Gottstein J, Blei AT (1999) Cerebral blood flow and the development of ammonia-induced brain edema in rats after portacaval anastomosis. Hepatology (Baltimore, Md) 30:876–880. https://doi.org/10.1002/hep.510300428
McPheeters CM, VanArsdale VM, Weant KA (2016) N-Acetylcysteine use in non-acetaminophen-induced acute liver failure. Adv Emerg Nurs J 38:183–189. https://doi.org/10.1097/tme.0000000000000116
Michalak A, Butterworth RF (1997) Selective loss of binding sites for the glutamate receptor ligands [3H]kainate and (S)-[3H]5-fluorowillardiine in the brains of rats with acute liver failure. Hepatology (Baltimore, Md) 25:631–635. https://doi.org/10.1002/hep.510250323
Michalak A, Rose C, Butterworth J, Butterworth RF (1996) Neuroactive amino acids and glutamate (NMDA) receptors in frontal cortex of rats with experimental acute liver failure. Hepatology (Baltimore, Md) 24:908–913. https://doi.org/10.1002/hep.510240425
Mladenovic D et al (2014) Finasteride improves motor, EEG, and cellular changes in rat brain in thioacetamide-induced hepatic encephalopathy. Am J Physiol Gastrointest Liver Physiol 307:G931–G940. https://doi.org/10.1152/ajpgi.00463.2013
Mladenović D, Petronijević N, Stojković T, Velimirović M, Jevtić G, Hrnčić D, Radosavljević T, Rašić-Marković A, Maksić N, Djuric D, Stanojlović O (2015) Finasteride has regionally different effects on brain oxidative stress and acetylcholinesterase activity in acute thioacetamide-induced hepatic encephalopathy in rats. PLoS One 10:e0134434. https://doi.org/10.1371/journal.pone.0134434
Mohsenin V (2013) Assessment and management of cerebral edema and intracranial hypertension in acute liver failure. J Crit Care 28:783–791. https://doi.org/10.1016/j.jcrc.2013.04.002
Molofsky AV, Deneen B (2015) Astrocyte development: A guide for the perplexed. Glia 63:1320–1329. https://doi.org/10.1002/glia.22836
Morishima T et al (2008) Lactic acid increases aquaporin 4 expression on the cell membrane of cultured rat astrocytes. Neurosci Res 61:18–26. https://doi.org/10.1016/j.neures.2008.01.005
Murphy N, Auzinger G, Bernel W, Wendon J (2004) The effect of hypertonic sodium chloride on intracranial pressure in patients with acute liver failure. Hepatology (Baltimore, Md) 39:464–470. https://doi.org/10.1002/hep.20056
Murthy CR, Bender AS, Dombro RS, Bai G, Norenberg MD (2000) Elevation of glutathione levels by ammonium ions in primary cultures of rat astrocytes. Neurochem Int 37:255–268
Murthy CR, Rama Rao KV, Bai G, Norenberg MD (2001) Ammonia-induced production of free radicals in primary cultures of rat astrocytes. J Neurosci Res 66:282–288. https://doi.org/10.1002/jnr.1222
Nicholls DG (2004) Mitochondrial dysfunction and glutamate excitotoxicity studied in primary neuronal cultures. Curr Mol Med 4:149–177
Norenberg MD (1994) Astrocyte responses to CNS injury. J Neuropathol Exp Neurol 53:213–220
Norenberg MD, Neary JT, Norenberg LO, McCarthy M (1990) Ammonia induced decrease in glial fibrillary acidic protein in cultured astrocytes. J Neuropathol Exp Neurol 49:399–405
Norenberg MD, Huo Z, Neary JT, Roig-Cantesano A (1997) The glial glutamate transporter in hyperammonemia and hepatic encephalopathy: relation to energy metabolism and glutamatergic neurotransmission. Glia 21:124–133
Norenberg MD, Jayakumar AR, Rama Rao KV, Panickar KS (2007) New concepts in the mechanism of ammonia-induced astrocyte swelling. Metab Brain Dis 22:219–234. https://doi.org/10.1007/s11011-007-9062-5
Odeh M, Sabo E, Srugo I, Oliven A (2005) Relationship between tumor necrosis factor-alpha and ammonia in patients with hepatic encephalopathy due to chronic liver failure. Ann Med 37:603–612. https://doi.org/10.1080/07853890500317414
Oenarto J, Karababa A, Castoldi M, Bidmon HJ, Gorg B, Haussinger D (2016) Ammonia-induced miRNA expression changes in cultured rat astrocytes. Sci Rep 6:18493. https://doi.org/10.1038/srep18493
Oh YJ, Francis JW, Markelonis GJ, Oh TH (1992) Interleukin‐1‐β and tumor necrosis factor‐α increase peripheral‐type benzodiazepine binding sites in cultured polygonal astrocytes. J Neurochem 58:2131–2138. https://doi.org/10.1111/j.1471-4159.1992.tb10955.x
Ohnishi M et al (2014) High-mobility group box 1 up-regulates aquaporin 4 expression via microglia-astrocyte interaction. Neurochem Int 75:32–38. https://doi.org/10.1016/j.neuint.2014.05.007
Olivo R, Guarrera JV, Pyrsopoulos NT (2018) Liver Transplantation for Acute Liver Failure. Clin Liver Dis 22:409–417. https://doi.org/10.1016/j.cld.2018.01.014
Ordaz B, Tuz K, Ochoa LD, Lezama R, Pena-Segura C, Franco R (2004) Osmolytes and mechanisms involved in regulatory volume decrease under conditions of sudden or gradual osmolarity decrease. Neurochem Res 29:65–72
Ozawa K, Kamiyama Y, Kimura K, Yamamoto M, Aoyama H, Yasuda K, Tobe T (1983) Contribution of the arterial blood ketone body ratio to elevate plasma amino acids in hepatic encephalopathy of surgical patients. Am J Surg 146:299–305. https://doi.org/10.1016/0002-9610(83)90401-4
Pannasch U, Rouach N (2013) Emerging role for astroglial networks in information processing: from synapse to behavior. Trends Neurosci 36:405–417. https://doi.org/10.1016/j.tins.2013.04.004
Papadopoulos MC, Verkman AS (2013) Aquaporin water channels in the nervous system. Nat Rev Neurosci 14:265–277. https://doi.org/10.1038/nrn3468
Park CH, Carboni E, Wood PL, Gee KW (1996) Characterization of peripheral benzodiazepine type sites in a cultured murine BV-2 microglial cell line. Glia 16:65–70. https://doi.org/10.1002/(SICI)1098-1136(199601)16:1<65::AID-GLIA7>3.0.CO;2-A
Peeling J, Shoemaker L, Gauthier T, Benarroch A, Sutherland GR, Minuk GY (1993) Cerebral metabolic and histological effects of thioacetamide-induced liver failure. Am J Phys 265:G572–G578. https://doi.org/10.1152/ajpgi.1993.265.3.G572
Pekny M, Wilhelmsson U (2006) GFAP and astrocyte intermediate filaments. In: Lajtha A, Lim R (eds) Handbook of neurochemistry and molecular neurobiology: Neuroactive proteins and peptides. Springer US, Boston, pp 289–314. https://doi.org/10.1007/978-0-387-30381-9_14
Rabinstein AA (2010) Treatment of brain edema in acute liver failure. Curr Treat Options Neurol 12:129–141. https://doi.org/10.1007/s11940-010-0062-0
Rai R, Saraswat VA, Dhiman RK (2015) Gut microbiota: its role in hepatic encephalopathy. J Clin Exp Hepatol 5:S29–S36. https://doi.org/10.1016/j.jceh.2014.12.003
Rama Rao KV, Chen M, Simard JM, Norenberg MD (2003a) Increased aquaporin-4 expression in ammonia-treated cultured astrocytes. Neuroreport 14:2379–2382. https://doi.org/10.1097/01.wnr.0000099610.19426.a6
Rama Rao KV, Chen M, Simard JM, Norenberg MD (2003b) Suppression of ammonia-induced astrocyte swelling by cyclosporin A. J Neurosci Res 74:891–897. https://doi.org/10.1002/jnr.10755
Rama Rao KV, Jayakumar AR, Norenberg MD (2003c) Induction of the mitochondrial permeability transition in cultured astrocytes by glutamine. Neurochem Int 43:517–523
Rama Rao KV, Jayakumar AR, Norenberg MD (2005a) Differential response of glutamine in cultured neurons and astrocytes. J Neurosci Res 79:193–199. https://doi.org/10.1002/jnr.20295
Rama Rao KV, Jayakumar AR, Norenberg MD (2005b) Role of oxidative stress in the ammonia-induced mitochondrial permeability transition in cultured astrocytes. Neurochem Int 47:31–38. https://doi.org/10.1016/j.neuint.2005.04.004
Rama Rao KV, Jayakumar AR, Tong X, Alvarez VM, Norenberg MD (2010a) Marked potentiation of cell swelling by cytokines in ammonia-sensitized cultured astrocytes. J Neuroinflammation 7:66. https://doi.org/10.1186/1742-2094-7-66
Rama Rao KV, Jayakumar AR, Tong X, Curtis KM, Norenberg MD (2010b) Brain aquaporin-4 in experimental acute liver failure. J Neuropathol Exp Neurol 69:869–879. https://doi.org/10.1097/NEN.0b013e3181ebe581
Rama Rao KV, Reddy PV, Tong X, Norenberg MD (2010c) Brain edema in acute liver failure: inhibition by L-histidine. Am J Pathol 176:1400–1408. https://doi.org/10.2353/ajpath.2010.090756
Rama Rao KV, Verkman AS, Curtis KM, Norenberg MD (2014) Aquaporin-4 deletion in mice reduces encephalopathy and brain edema in experimental acute liver failure. Neurobiol Dis 63:222–228. https://doi.org/10.1016/j.nbd.2013.11.018
Rao KV, Norenberg MD (2001) Cerebral energy metabolism in hepatic encephalopathy and hyperammonemia. Metab Brain Dis 16:67–78
Rao VL, Audet RM, Butterworth RF (1997) Increased neuronal nitric oxide synthase expression in brain following portacaval anastomosis. Brain Res 765:169–172
Rao KV, Brahmbhatt M, Norenberg MD (2013) Microglia contribute to ammonia-induced astrocyte swelling in culture. Metab Brain Dis 28:139–143. https://doi.org/10.1007/s11011-012-9339-1
Rao VK, Carlson EA, Yan SS (2014) Mitochondrial permeability transition pore is a potential drug target for neurodegeneration. Biochim Biophys Acta 1842:1267–1272. https://doi.org/10.1016/j.bbadis.2013.09.003
Raschke RA et al (2008) Results of a protocol for the management of patients with fulminant liver failure. Crit Care Med 36:2244–2248. https://doi.org/10.1097/CCM.0b013e31818029a3
Record CO, Buxton B, Chase RA, Curzon G, Murray-Lyon IM, Williams R (1976) Plasma and brain amino acids in fulminant hepatic failure and their relationship to hepatic encephalopathy. Eur J Clin Investig 6:387–394
Reinehr R et al (2007) Hypoosmotic swelling and ammonia increase oxidative stress by NADPH oxidase in cultured astrocytes and vital brain slices. Glia 55:758–771. https://doi.org/10.1002/glia.20504
Ren C, Li X-h WY, Dong N, Y-l T, Y-m Y (2019) Inhibition of cerebral high-mobility group Box 1 protein attenuates multiple organ damage and improves T Cell-Mediated immunity in septic rats. Mediat Inflamm 2019:10. https://doi.org/10.1155/2019/6197084
Report from the European Association for the Study of the Liver (EASL) (1979) Randomised trial of steroid therapy in acute liver failure. Gut 20:620–623. https://doi.org/10.1136/gut.20.7.620
Rivera-Mancia S, Rios C, Montes S (2012) Manganese and ammonia interactions in the brain of cirrhotic rats: effects on brain ammonia metabolism. Neurochem Res 37:1074–1084. https://doi.org/10.1007/s11064-012-0710-8
Rodrigo R, Cauli O, Gomez-Pinedo U, Agusti A, Hernandez-Rabaza V, Garcia-Verdugo JM, Felipo V (2010) Hyperammonemia induces neuroinflammation that contributes to cognitive impairment in rats with hepatic encephalopathy. Gastroenterology 139:675–684. https://doi.org/10.1053/j.gastro.2010.03.040
Rolando N, Clapperton M, Wade J, Panetsos G, Mufti G, Williams R (2000a) Granulocyte colony-stimulating factor improves function of neutrophils from patients with acute liver failure. Eur J Gastroenterol Hepatol 12:1135–1140. https://doi.org/10.1097/00042737-200012100-00011
Rolando N, Wade J, Davalos M, Wendon J, Philpott-Howard J, Williams R (2000b) The systemic inflammatory response syndrome in acute liver failure. Hepatology (Baltimore, Md) 32:734–739. https://doi.org/10.1053/jhep.2000.17687
Rose CF (2012) Ammonia-lowering strategies for the treatment of hepatic encephalopathy. Clin Pharmacol Ther 92:321–331. https://doi.org/10.1038/clpt.2012.112
Rose C, Butterworth R.F., Zayed J., Normandin L., Todd K., Michalak A., Spahr L., Huet P.M., Pomier-Layrargues G. (1999) Manganese deposition in basal ganglia structures results from both portal-systemic shunting and liver dysfunction. Gastroenterology 117:640–644
Sanchez-Prieto J, Gonzalez P (1988) Occurrence of a large Ca2+−independent release of glutamate during anoxia in isolated nerve terminals (synaptosomes). J Neurochem 50:1322–1324
San-Miguel B, Alvarez M, Culebras JM, Gonzalez-Gallego J, Tunon MJ (2006) N-acetyl-cysteine protects liver from apoptotic death in an animal model of fulminant hepatic failure. Apoptosis: an international journal on programmed cell death 11:1945–1957. https://doi.org/10.1007/s10495-006-0090-0
Schliess F et al (2002) Ammonia induces MK-801-sensitive nitration and phosphorylation of protein tyrosine residues in rat astrocytes. FASEB J 16:739–741. https://doi.org/10.1096/fj.01-0862fje
Schliess F, Foster N, Gorg B, Reinehr R, Haussinger D (2004) Hypoosmotic swelling increases protein tyrosine nitration in cultured rat astrocytes. Glia 47:21–29. https://doi.org/10.1002/glia.20019
Schneider GH, Baethmann A, Kempski O (1992) Mechanisms of glial swelling induced by glutamate. Can J Physiol Pharmacol 70:S334–S343
Shah N et al (2013) Role of toll-like receptor 4 in mediating multiorgan dysfunction in mice with acetaminophen induced acute liver failure liver transplantation: official publication of the American Association for the Study of Liver Diseases and the international. Liver Transpl Soc 19:751–761. https://doi.org/10.1002/lt.23655
Shawcross DL, Wendon JA (2012) The neurological manifestations of acute liver failure. Neurochem Int 60:662–671. https://doi.org/10.1016/j.neuint.2011.10.006
Shawcross DL, Davies NA, Williams R, Jalan R (2004) Systemic inflammatory response exacerbates the neuropsychological effects of induced hyperammonemia in cirrhosis. J Hepatol 40:247–254
Shawcross DL, Shabbir SS, Taylor NJ, Hughes RD (2010) Ammonia and the neutrophil in the pathogenesis of hepatic encephalopathy in cirrhosis. Hepatology (Baltimore, Md) 51:1062–1069. https://doi.org/10.1002/hep.23367
Simard JM, Yurovsky V, Tsymbalyuk N, Melnichenko L, Ivanova S, Gerzanich V (2009) Protective effect of delayed treatment with low-dose glibenclamide in three models of ischemic stroke. Stroke 40:604–609. https://doi.org/10.1161/strokeaha.108.522409
Sinke A, Jayakumar A, Panickar K, Moriyama M, Reddy P, Norenberg M (2008) NFκB in the Mechanism of Ammonia-Induced Astrocyte Swelling in Culture. Neurochem 106:2302–2311. https://doi.org/10.1111/j.1471-4159.2008.05549.x
Sobel RA, DeArmond SJ, Forno LS, Eng LF (1981) Glial fibrillary acidic protein in hepatic encephalopathy. An immunohistochemical study. J Neuropathol Exp Neurol 40:625–632
Sofroniew MV, Vinters HV (2010) Astrocytes: biology and pathology. Acta Neuropathol 119:7–35. https://doi.org/10.1007/s00401-009-0619-8
Sohn Y et al (2011) Na+/HCO3- cotransporter immunoreactivity changes in neurons and expresses in astrocytes in the gerbil hippocampal CA1 region after ischemia/reperfusion. Neurochem Res 36:2459–2469. https://doi.org/10.1007/s11064-011-0572-5
Song G, Dhodda VK, Blei AT, Dempsey RJ, Rao VL (2002) GeneChip analysis shows altered mRNA expression of transcripts of neurotransmitter and signal transduction pathways in the cerebral cortex of portacaval shunted rats. J Neurosci Res 68:730–737. https://doi.org/10.1002/jnr.10268
Spahr L et al (1996) Increased blood manganese in cirrhotic patients: relationship to pallidal magnetic resonance signal hyperintensity and neurological symptoms. Hepatology (Baltimore, Md) 24:1116–1120. https://doi.org/10.1002/hep.510240523
Sriuttha P, Sirichanchuen B, Permsuwan U (2018, 2018) Hepatotoxicity of nonsteroidal anti-inflammatory drugs: a systematic review of randomized controlled trials. Int J Hepatol:5253623. https://doi.org/10.1155/2018/5253623
Stadlbauer V, Wright GA, Jalan R (2009) Role of artificial liver support in hepatic encephalopathy. Metab Brain Dis 24:15–26. https://doi.org/10.1007/s11011-008-9117-2
Staub F, Baethmann A, Peters J, Weigt H, Kempski O (1990) Effects of lactacidosis on glial cell volume and viability. J Cereb Blood Flow Metab: official journal of the International Society of Cerebral Blood Flow and Metabolism 10:866–876. https://doi.org/10.1038/jcbfm.1990.143
Staub F, Peters J, Kempski O, Schneider G-H, Schürer L, Baethmann A (1993) Swelling of glial cells in lactacidosis and by glutamate: significance of Cl—-transport. Brain Res 610:69–74. https://doi.org/10.1016/0006-8993(93)91218-H
Staub F, Winkler A, Peters J, Kempski O, Kachel V, Baethmann A (1994) Swelling, acidosis, and irreversible damage of glial cells from exposure to arachidonic acid in vitro. J Cereb Blood Flow Metab 14:1030–1039. https://doi.org/10.1038/jcbfm.1994.135
Steinman MQ, Gao V, Alberini CM (2016) The role of lactate-mediated metabolic coupling between astrocytes and neurons in long-term memory formation frontiers in integrative. Neuroscience 10. https://doi.org/10.3389/fnint.2016.00010
Stravitz RT, Larsen FS (2009) Therapeutic hypothermia for acute liver failure. Crit Care Med 37:S258–S264. https://doi.org/10.1097/CCM.0b013e3181aa5fb8
Su G, Kintner DB, Sun D (2002) Contribution of Na(+)-K(+)-cl(−) cotransporter to high-[K(+)](o)- induced swelling and EAA release in astrocytes. Am J Phys Cell Physiol 282:C1136–C1146. https://doi.org/10.1152/ajpcell.00478.2001
Suarez I, Bodega G, Fernandez B (2000) Modulation of glutamate transporters (GLAST, GLT-1 and EAAC1) in the rat cerebellum following portocaval anastomosis. Brain Res 859:293–302
Swain M, Butterworth RF, Blei AT (1992) Ammonia and related amino acids in the pathogenesis of brain edema in acute ischemic liver failure in rats. Hepatology (Baltimore, md) 15:449–453
Szabo I, De Pinto V, Zoratti M (1993) The mitochondrial permeability transition pore may comprise VDAC molecules. II The electrophysiological properties of VDAC are compatible with those of the mitochondrial megachannel. FEBS Lett 330:206–210
Tabata H (2015) Diverse subtypes of astrocytes and their development during corticogenesis. Front Neurosci 9:114. https://doi.org/10.3389/fnins.2015.00114
Tait MJ, Saadoun S, Bell BA, Verkman AS, Papadopoulos MC (2010) Increased brain edema in aqp4-null mice in an experimental model of subarachnoid hemorrhage. Neuroscience 167:60–67. https://doi.org/10.1016/j.neuroscience.2010.01.053
Tajiri K, Shimizu Y (2013) Branched-chain amino acids in liver diseases. World J Gastroenterol 19:7620–7629. https://doi.org/10.3748/wjg.v19.i43.7620
Theobaldo MC, Barbeiro HV, Barbeiro DF, Petroni R, Soriano FG (2012) Hypertonic saline solution reduces the inflammatory response in endotoxemic rats. Clinics (Sao Paulo, Brazil) 67:1463–1468. https://doi.org/10.6061/clinics/2012(12)18
Therrien G, Butterworth RF (1991) Cerebrospinal fluid amino acids in relation to neurological status in experimental portal-systemic encephalopathy. Metab Brain Dis 6:65–74
Therrien G, Giguere JF, Butterworth RF (1991) Increased cerebrospinal fluid lactate reflects deterioration of neurological status in experimental portal-systemic encephalopathy. Metab Brain Dis 6:225–231
Thumburu KK et al (2014) Expression of astrocytic genes coding for proteins implicated in neural excitation and brain edema is altered after acute liver failure. J Neurochem 128:617–627. https://doi.org/10.1111/jnc.12511
Tofteng F, Larsen FS (2004) The effect of indomethacin on intracranial pressure, cerebral perfusion and extracellular lactate and glutamate concentrations in patients with fulminant hepatic failure. J Cereb Blood Flow Metab 24:798–804. https://doi.org/10.1097/01.wcb.0000125648.03213.1d
Tofteng F, Jorgensen L, Hansen BA, Ott P, Kondrup J, Larsen FS (2002) Cerebral microdialysis in patients with fulminant hepatic failure. Hepatology (Baltimore, Md) 36:1333–1340. https://doi.org/10.1053/jhep.2002.36944
Tofteng F, Hauerberg J, Hansen BA, Pedersen CB, Jorgensen L, Larsen FS (2006) Persistent arterial hyperammonemia increases the concentration of glutamine and alanine in the brain and correlates with intracranial pressure in patients with fulminant hepatic failure. J Cereb Blood Flow Metab: official journal of the International Society of Cerebral Blood Flow and Metabolism 26:21–27. https://doi.org/10.1038/sj.jcbfm.9600168
Traber PG, Dal Canto M, Ganger DR, Blei AT (1987) Electron microscopic evaluation of brain edema in rabbits with galactosamine-induced fulminant hepatic failure: ultrastructure and integrity of the blood-brain barrier. Hepatology (Baltimore, Md) 7:1272–1277
Van Harreveld A, Fifkova E (1971) Light- and electron-microscopic changes in central nervous tissue after electrophoretic injection of glutamate. Exp Mol Pathol 15:61–81
Vergara M, Castro-Gutierrez V, Rada G (2016) Do branched chain amino acids improve hepatic encephalopathy in cirrhosis? Medwave 16:e6795. https://doi.org/10.5867/medwave.2016.6795
Vogels BA, Maas MA, Daalhuisen J, Quack G, Chamuleau RA (1997a) Memantine, a noncompetitive NMDA receptor antagonist improves hyperammonemia-induced encephalopathy and acute hepatic encephalopathy in rats. Hepatology (Baltimore, Md) 25:820–827. https://doi.org/10.1002/hep.510250406
Vogels BA, van Steynen B, Maas MA, Jorning GG, Chamuleau RA (1997b) The effects of ammonia and portal-systemic shunting on brain metabolism, neurotransmission and intracranial hypertension in hyperammonaemia-induced encephalopathy. J Hepatol 26:387–395
Wang T, Suzuki K, Kakisaka K, Onodera M, Sawara K, Takikawa Y (2019) L-carnitine prevents ammonia-induced cytotoxicity and disturbances in intracellular amino acid levels in human astrocytes. J Gastroenterol Hepatol 34:1249–1255. https://doi.org/10.1111/jgh.14497
Ware AJ, D'Agostino AN, Combes B (1971) Cerebral edema: a major complication of massive hepatic necrosis. Gastroenterology 61:877–884
Warskulat U, Gorg B, Bidmon HJ, Muller HW, Schliess F, Haussinger D (2002) Ammonia-induced heme oxygenase-1 expression in cultured rat astrocytes and rat brain in vivo. Glia 40:324–336. https://doi.org/10.1002/glia.10128
Watanabe A, Takei N, Higashi T, Shiota T, Nakatsukasa H, Fujiwara M, Sakata T, Nagashima H (1984) Glutamic acid and glutamine levels in serum and cerebrospinal fluid in hepatic encephalopathy. Biochem Med 32:225–231
Waterhouse NJ, Goldstein JC, von Ahsen O, Schuler M, Newmeyer DD, Green DR (2001) Cytochrome c maintains mitochondrial transmembrane potential and ATP generation after outer mitochondrial membrane permeabilization during the apoptotic process. J Cell Biol 153:319–328
Willard-Mack CL, Koehler RC, Hirata T, Cork LC, Takahashi H, Traystman RJ, Brusilow SW (1996) Inhibition of glutamine synthetase reduces ammonia-induced astrocyte swelling in rat. Neuroscience 71:589–599
Williams R (2007) Review article: bacterial flora and pathogenesis in hepatic encephalopathy. Aliment Pharmacol Ther 25(Suppl 1):17–22. https://doi.org/10.1111/j.1746-6342.2006.03217.x
Wright G, Shawcross D, Olde Damink SW, Jalan R (2007) Brain cytokine flux in acute liver failure and its relationship with intracranial hypertension. Metab Brain Dis 22:375–388. https://doi.org/10.1007/s11011-007-9071-4
Wright G et al (2010) Role of aquaporin-4 in the development of brain oedema in liver failure. J Hepatol 53:91–97. https://doi.org/10.1016/j.jhep.2010.02.020
Xue Z, Li B, Gu L, Hu X, Li M, Butterworth RF, Peng L (2010) Increased Na, K-ATPase alpha2 isoform gene expression by ammonia in astrocytes and in brain in vivo. Neurochem Int 57:395–403. https://doi.org/10.1016/j.neuint.2010.04.014
Yang Z, Wang KK (2015) Glial fibrillary acidic protein: from intermediate filament assembly and gliosis to neurobiomarker. Trends Neurosci 38:364–374. https://doi.org/10.1016/j.tins.2015.04.003
Ytrebo LM, Korvald C, Nedredal GI, Elvenes OP, Nielsen Grymyr OJ, Revhaug A (2001) N-acetylcysteine increases cerebral perfusion pressure in pigs with fulminant hepatic failure. Crit Care Med 29:1989–1995. https://doi.org/10.1097/00003246-200110000-00023
Ytrebo LM et al (2009) L-ornithine phenylacetate attenuates increased arterial and extracellular brain ammonia and prevents intracranial hypertension in pigs with acute liver failure. Hepatology (Baltimore, Md) 50:165–174. https://doi.org/10.1002/hep.22917
Zamzami N, Hirsch T, Dallaporta B, Petit PX, Kroemer G (1997) Mitochondrial implication in accidental and programmed cell death: apoptosis and necrosis. J Bioenerg Biomembr 29:185–193
Zemtsova I, Gorg B, Keitel V, Bidmon HJ, Schror K, Haussinger D (2011) Microglia activation in hepatic encephalopathy in rats and humans. Hepatology (Baltimore, Md) 54:204–215. https://doi.org/10.1002/hep.24326
Zoratti M, Szabo I, De Marchi U (2005) Mitochondrial permeability transitions: how many doors to the house? Biochim Biophys Acta 1706:40–52. https://doi.org/10.1016/j.bbabio.2004.10.006
Zwingmann C, Flogel U, Pfeuffer J, Leibfritz D (2000) Effects of ammonia exposition on glioma cells: changes in cell volume and organic osmolytes studied by diffusion-weighted and high-resolution NMR spectroscopy. Dev Neurosci 22:463–471. https://doi.org/10.1159/000017476
Zwingmann C, Chatauret N, Rose C, Leibfritz D, Butterworth RF (2004) Selective alterations of brain osmolytes in acute liver failure: protective effect of mild hypothermia. Brain Res 999:118–123
Author information
Authors and Affiliations
Contributions
Ali Sepehrinezhad, Roger Williams and Ali Shahbazi designed the study, carried out the literature review and drafted the manuscript. Gholamreza Namvar carried out the literature review and participated in drafting the manuscript. Also, Roger Williams and Asadollah Zarifkar critically edited the manuscript and corrected grammatical errors in the revised manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sepehrinezhad, A., Zarifkar, A., Namvar, G. et al. Astrocyte swelling in hepatic encephalopathy: molecular perspective of cytotoxic edema. Metab Brain Dis 35, 559–578 (2020). https://doi.org/10.1007/s11011-020-00549-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-020-00549-8