Abstract
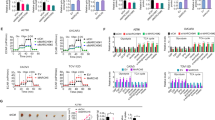
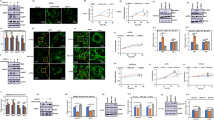
As a critical catalytic subunit of N6-methyladenosine (m6A) modification in messenger RNA, ALKBH5 has been reported to affect the progression of numerous tumors. However, the functions and mechanisms of ALKBH5 in thyroid cancer remain largely unknown. Relative mRNA and protein levels in thyroid cancer tissues and cells were detected by qRT-PCR and western blot, respectively. The proliferation and viability were evaluated using colony formation and CCK-8 assays. Intracellular iron level was measured by an iron colorimetric assay kit. ROS level was determined using CellRox Green reagent. TIAM1 mRNA m6A level was detected by MeRIP. Xenograft tumor growth was performed to examine the role of ALKBH5 in thyroid tumor growth in vivo. ALKBH5 was decreased in thyroid cancer tissues and cells. ALKBH5 overexpression inhibited thyroid cancer cell proliferation and increased the levels of Fe2+ and ROS and reduced the proteins expression of GPX4 and SLC7A11. Furthermore, overexpression of ALKBH5 inhibited TIAM1 expression by m6A modification, and overexpression of TIAM1 reversed the regulatory of oe-ALKBH5 on cell proliferation and ferroptosis in thyroid cancer. In addition, TIAM1 was elevated in thyroid cancer, and TIAM1 knockdown repressed thyroid cancer cell proliferation and promoted ferroptosis through regulating Nrf2/HO-1 axis. In addition, in vivo evidences also showed that ALKBH5 suppressed thyroid cancer progression by decreasing the m6A level of TIAM1. Our findings suggested that ALKBH5 inhibited thyroid cancer progression by inducing ferroptosis through m6A-TIAM1–Nrf2/HO-1 axis, suggesting ALKBH5 might be a potential target molecule for the treatment and diagnosis of thyroid cancer.






Similar content being viewed by others
Data availability
Enquiries about data availability should be directed to the authors.
References
Santiago K, Chen Wongworawat Y, Khan S (2020) Differential microRNA-signatures in thyroid cancer subtypes. J Oncol 2020:2052396. https://doi.org/10.1155/2020/2052396
Siegel RL, Miller KD, Fuchs HE, Jemal A (2021) Cancer statistics, 2021. CA Cancer J Clin 71:7–33. https://doi.org/10.3322/caac.21654
Pelizzo MR, Merante Boschin I, Toniato A, Pagetta C, Casal Ide E, Mian C, Rubello D (2008) Diagnosis, treatment, prognostic factors and long-term outcome in papillary thyroid carcinoma. Minerva Endocrinol 33:359–379
Adams JM, Cory S (1975) Modified nucleosides and bizarre 5’-termini in mouse myeloma mRNA. Nature 255:28–33. https://doi.org/10.1038/255028a0
Beemon K, Keith J (1977) Localization of N6-methyladenosine in the Rous sarcoma virus genome. J Mol Biol 113:165–179. https://doi.org/10.1016/0022-2836(77)90047-x
Zhu H, Gan X, Jiang X, Diao S, Wu H, Hu J (2019) ALKBH5 inhibited autophagy of epithelial ovarian cancer through miR-7 and BCL-2. J Exp Clin Cancer Res 38:163. https://doi.org/10.1186/s13046-019-1159-2
Zhang J, Guo S, Piao HY, Wang Y, Wu Y, Meng XY, Yang D, Zheng ZC, Zhao Y (2019) ALKBH5 promotes invasion and metastasis of gastric cancer by decreasing methylation of the lncRNA NEAT1. J Physiol Biochem 75:379–389. https://doi.org/10.1007/s13105-019-00690-8
Tang B, Yang Y, Kang M, Wang Y, Wang Y, Bi Y, He S, Shimamoto F (2020) m(6)A demethylase ALKBH5 inhibits pancreatic cancer tumorigenesis by decreasing WIF-1 RNA methylation and mediating Wnt signaling. Mol Cancer 19:3. https://doi.org/10.1186/s12943-019-1128-6
Hou J, Shan H, Zhang Y, Fan Y, Wu B (2020) m(6)A RNA methylation regulators have prognostic value in papillary thyroid carcinoma. Am J Otolaryngol 41:102547. https://doi.org/10.1016/j.amjoto.2020.102547
Minard ME, Kim LS, Price JE, Gallick GE (2004) The role of the guanine nucleotide exchange factor Tiam1 in cellular migration, invasion, adhesion and tumor progression. Breast Cancer Res Treat 84:21–32. https://doi.org/10.1023/B:BREA.0000018421.31632.e6
Izumi D, Toden S, Ureta E, Ishimoto T, Baba H, Goel A (2019) TIAM1 promotes chemoresistance and tumor invasiveness in colorectal cancer. Cell Death Dis 10:267. https://doi.org/10.1038/s41419-019-1493-5
Walch A, Seidl S, Hermannstadter C, Rauser S, Deplazes J, Langer R, von Weyhern CH, Sarbia M, Busch R, Feith M, Gillen S, Hofler H, Luber B (2008) Combined analysis of Rac 1, IQGAP1, Tiam1 and E-cadherin expression in gastric cancer. Mod Pathol 21:544–552. https://doi.org/10.1038/modpathol.2008.3
Yang Y, Wu Q, Li N, Che S, Jin T, Nan Y, Lin Z, Chen L (2018) Upregulation of Tiam1 contributes to cervical cancer disease progression and indicates poor survival outcome. Hum Pathol 75:179–188. https://doi.org/10.1016/j.humpath.2018.02.006
Hsueh C, Lin JD, Yang CF, Chang YS, Chao TC, Sun JH, Wu IC, Tseng NM, Ueng SH (2011) Prognostic significance of Tiam1 expression in papillary thyroid carcinoma. Virchows Arch 459:587–593. https://doi.org/10.1007/s00428-011-1164-0
Liu L, Wu B, Cai H, Li D, Ma Y, Zhu X, Lv Z, Fan Y, Zhang X (2018) Tiam1 promotes thyroid carcinoma metastasis by modulating EMT via Wnt/beta-catenin signaling. Exp Cell Res 362:532–540. https://doi.org/10.1016/j.yexcr.2017.12.019
Kobayashi EH, Suzuki T, Funayama R, Nagashima T, Hayashi M, Sekine H, Tanaka N, Moriguchi T, Motohashi H, Nakayama K, Yamamoto M (2016) Nrf2 suppresses macrophage inflammatory response by blocking proinflammatory cytokine transcription. Nat Commun 7:11624. https://doi.org/10.1038/ncomms11624
Ziros PG, Manolakou SD, Habeos IG, Lilis I, Chartoumpekis DV, Koika V, Soares P, Kyriazopoulou VE, Scopa CD, Papachristou DJ, Sykiotis GP (2013) Nrf2 is commonly activated in papillary thyroid carcinoma, and it controls antioxidant transcriptional responses and viability of cancer cells. J Clin Endocrinol Metab 98:E1422–E1427. https://doi.org/10.1210/jc.2013-1510
Dong H, Qiang Z, Chai D, Peng J, Xia Y, Hu R, Jiang H (2020) Nrf2 inhibits ferroptosis and protects against acute lung injury due to intestinal ischemia reperfusion via regulating SLC7A11 and HO-1. Aging (Albany NY) 12:12943–12959. https://doi.org/10.18632/aging.103378
Mou Y, Wang J, Wu J, He D, Zhang C, Duan C, Li B (2019) Ferroptosis, a new form of cell death: opportunities and challenges in cancer. J Hematol Oncol 12:34. https://doi.org/10.1186/s13045-019-0720-y
Xu T, Ding W, Ji X, Ao X, Liu Y, Yu W, Wang J (2019) Molecular mechanisms of ferroptosis and its role in cancer therapy. J Cell Mol Med 23:4900–4912. https://doi.org/10.1111/jcmm.14511
Chen W, Fu J, Chen Y, Li Y, Ning L, Huang D, Yan S, Zhang Q (2021) Circular RNA circKIF4A facilitates the malignant progression and suppresses ferroptosis by sponging miR-1231 and upregulating GPX4 in papillary thyroid cancer. Aging (Albany NY) 13:16500–16512. https://doi.org/10.18632/aging.203172
Wang HH, Ma JN, Zhan XR (2021) Circular RNA Circ_0067934 attenuates ferroptosis of thyroid cancer cells by miR-545-3p/SLC7A11 signaling. Front Endocrinol (Lausanne) 12:670031. https://doi.org/10.3389/fendo.2021.670031
Wang L, Zhang Y, Yang J, Liu L, Yao B, Tian Z, He J (2021) The knockdown of ETV4 inhibits the papillary thyroid cancer development by promoting ferroptosis upon SLC7A11 downregulation. DNA Cell Biol. https://doi.org/10.1089/dna.2021.0216
Shu T, Yang L, Sun L, Lu J, Zhan X (2020) CircHIPK3 promotes thyroid cancer tumorigenesis and invasion through the mirna-338-3p/RAB23 axis. Med Princ Pract. https://doi.org/10.1159/000512548
Shi D, Wang H, Ding M, Yang M, Li C, Yang W, Chen L (2019) MicroRNA-26a-5p inhibits proliferation, invasion and metastasis by repressing the expression of Wnt5a in papillary thyroid carcinoma. Onco Targets Ther 12:6605–6616. https://doi.org/10.2147/OTT.S205994
Guo T, Liu DF, Peng SH, Xu AM (2020) ALKBH5 promotes colon cancer progression by decreasing methylation of the lncRNA NEAT1. Am J Transl Res 12:4542–4549
Zhu Z, Qian Q, Zhao X, Ma L, Chen P (2020) N(6)-methyladenosine ALKBH5 promotes non-small cell lung cancer progress by regulating TIMP3 stability. Gene 731:144348. https://doi.org/10.1016/j.gene.2020.144348
Yu H, Yang X, Tang J, Si S, Zhou Z, Lu J, Han J, Yuan B, Wu Q, Lu Q, Yang H (2021) ALKBH5 inhibited cell proliferation and sensitized bladder cancer cells to cisplatin by m6A-CK2alpha-mediated glycolysis. Mol Ther Nucleic Acids 23:27–41. https://doi.org/10.1016/j.omtn.2020.10.031
Chen Y, Zhao Y, Chen J, Peng C, Zhang Y, Tong R, Cheng Q, Yang B, Feng X, Lu Y, Xie H, Zhou L, Wu J, Zheng S (2020) ALKBH5 suppresses malignancy of hepatocellular carcinoma via m(6)A-guided epigenetic inhibition of LYPD1. Mol Cancer 19:123. https://doi.org/10.1186/s12943-020-01239-w
Ding J, Yang F, Wu W (2019) Tiam1 high expression is associated with poor prognosis in solid cancers: a meta-analysis. Medicine (Baltimore) 98:e17529. https://doi.org/10.1097/MD.0000000000017529
Han FJ, Li J, Shen Y, Guo Y, Liu YC, Yu Y, Xu JY, Liu SX, Wang YH (2020) microRNA-1271-5p/TIAM1 suppresses the progression of ovarian cancer through inactivating Notch signaling pathway. J Ovarian Res 13:110. https://doi.org/10.1186/s13048-020-00720-w
Zhang X, Liu L, Deng X, Li D, Cai H, Ma Y, Jia C, Wu B, Fan Y, Lv Z (2019) MicroRNA 483–3p targets Pard3 to potentiate TGF-beta1-induced cell migration, invasion, and epithelial-mesenchymal transition in anaplastic thyroid cancer cells. Oncogene 38:699–715. https://doi.org/10.1038/s41388-018-0447-1
Abdalkader M, Lampinen R, Kanninen KM, Malm TM, Liddell JR (2018) Targeting Nrf2 to suppress ferroptosis and mitochondrial dysfunction in neurodegeneration. Front Neurosci 12:466. https://doi.org/10.3389/fnins.2018.00466
Song X, Long D (2020) Nrf2 and ferroptosis: a new research direction for neurodegenerative diseases. Front Neurosci 14:267. https://doi.org/10.3389/fnins.2020.00267
Roh JL, Kim EH, Jang H, Shin D (2017) Nrf2 inhibition reverses the resistance of cisplatin-resistant head and neck cancer cells to artesunate-induced ferroptosis. Redox Biol 11:254–262. https://doi.org/10.1016/j.redox.2016.12.010
Lou JS, Zhao LP, Huang ZH, Chen XY, Xu JT, Tai WC, Tsim KWK, Chen YT, Xie T (2021) Ginkgetin derived from Ginkgo biloba leaves enhances the therapeutic effect of cisplatin via ferroptosis-mediated disruption of the Nrf2/HO-1 axis in EGFR wild-type non-small-cell lung cancer. Phytomedicine 80:153370. https://doi.org/10.1016/j.phymed.2020.153370
Wei R, Zhao Y, Wang J, Yang X, Li S, Wang Y, Yang X, Fei J, Hao X, Zhao Y, Gui L, Ding X (2021) Tagitinin C induces ferroptosis through PERK-Nrf2-HO-1 signaling pathway in colorectal cancer cells. Int J Biol Sci 17:2703–2717. https://doi.org/10.7150/ijbs.59404
Gong Z, Xue L, Wei M, Liu Z, Vlantis AC, van Hasselt CA, Chan JYK, Li D, Zeng X, Tong MCF, Chen GG (2021) The knockdown of Nrf2 suppressed tumor growth and increased the sensitivity to lenvatinib in anaplastic thyroid cancer. Oxid Med Cell Longev 2021:3900330. https://doi.org/10.1155/2021/3900330
Li S, Zhang Y, Zhang J, Yu B, Wang W, Jia B, Chang J, Liu J (2022) Neferine exerts ferroptosis-inducing effect and antitumor effect on thyroid cancer through Nrf2/HO-1/NQO1 inhibition. J Oncol 2022:7933775. https://doi.org/10.1155/2022/7933775
Acknowledgements
We would like to give our sincere gratitude to the reviewers for their constructive comments.
Funding
This work was supported by the National Natural Science Foundation of China (81873730), the Guiding Project of Clinical Medical Technology Innovation in Hunan Province (2020SK51706), Clinical Research Center for Breast & Thyroid Disease Prevention in Hunan Province (2018SK4001), and General Guidance Project of Hunan Provincial Health Commission (202204013843).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors have no commercial or other associations that might pose a conflict of interest.
Ethical approval
Human thyroid cancer tissue specimens were obtained from the Second Affiliated Hospital of Soochow University. Our study was approved by the Ethics Committee of the Second Affiliated Hospital of Soochow University. Animal procedures complied with the guidelines of the Committee on the Ethics of Animal Experiments of the Second Affiliated Hospital of Soochow University.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (TIF 5411 kb)
Fig. S1 qRT-PCR analysis of the expression of different m6A modifying enzymes in thyroid cancer tissues. (A) qRT-PCR detected the expression of METTL14. (B) METTL3 expression was tested by qRT-PCR. (C) RBM15 expression was measured by qRT-PCR. (D) FTO expression was tested by qRT-PCR. (E) WTAP level was detected by qRT-PCR.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, W., Huang, G., Wei, J. et al. ALKBH5 inhibits thyroid cancer progression by promoting ferroptosis through TIAM1–Nrf2/HO-1 axis. Mol Cell Biochem 478, 729–741 (2023). https://doi.org/10.1007/s11010-022-04541-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-022-04541-x