Abstract
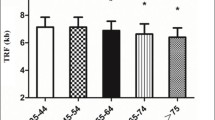
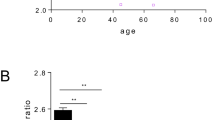
Somatic telomere DNA length is known to shorten with certain disease states and senescence. Furthermore, we have reported that the telomere length of a sub-healthy population also correlates with the blood data of laboratory tests. These facts suggest that patients with shorter telomere length tend to be hospitalized more easily than patients with longer telomere length. And such hospitalization tendencies can also be reflected in differences in clinical laboratory data. To address this issue, we evaluated and compared the telomere length and clinical laboratory data of outpatients and inpatients. In this study, 35 inpatients with chronic illness and 38 outpatients with one or more weeks without hospitalization experience were enrolled. Telomere length was shorter in hospitalized patients than outpatients. Inpatients and outpatients showed significant differences in some laboratory test results. Male outpatients showed higher values of fast blood sugar, HbA1c, blood urea nitrogen, creatinine, C-reactive protein, red blood cell count, and hemoglobin. Among female outpatients, the values of aspartate aminotransferase, alanine aminotransferase, albumin, creatine kinase, red blood cell count and hemoglobin were high. Of these, only albumin levels showed a positive correlation with telomere length in both sexes. Unexpectedly, all the other clinical data distinguishing outpatients and inpatients showed no significant association with telomere length. These items appeared to be related to hospital risk independently of TL. Having a shorter somatic telomere length appeared to be at a higher risk of hospitalization. This risk can be augmented by further complications such as deterioration of nutritional status and anemia. Maintaining sufficiently high nutritional status and erythropoietic potential may lead to avoidance of clinical events that require hospitalization.



Similar content being viewed by others
References
Blackburn EH (1991) Structure and function of telomeres. Nature 350(6319):569–573. https://doi.org/10.1038/350569a0
Zakian VA (1995) Telomeres: beginning to understand the end. Science 270:1601–1607
Aubert G, Lansdorp PM (2008) Telomeres and aging. Physiol Rev 88:557–579. https://doi.org/10.1152/physrev.00026.2007
Iwama H, Ohyashiki K, Ohyashiki JH, Hayashi S, Yahata N, Ando K, Toyama K, Hoshika A, Takasaki M, Mori M, Shay JW (1998) Telomeric length and telomerase activity vary with age in peripheral blood cells obtained from normal individuals. Hum Genet 102:397–402
Guan JZ, Maeda T, Sugano M, Oyama J, Higuchi Y, Makino N (2007) Change in the telomere length distribution with age in the Japanese population. Mol Cell Biochem 304:353–360. https://doi.org/10.1007/s11010-007-9518-2
Uziel O, Singer JA, Danicek V, Sahar G, Berkov E, Luchansky M, Fraser A, Ram R, Lahav M (2007) Telomere dynamics in arteries and mononuclear cells of diabetic patients: effect of diabetes and of glycemic control. Exp Gerontol 42:971–978. https://doi.org/10.1016/j.exger.2007.07.005
Brouilette S, Singh RK, Thompson JR, Goodall AH, Samani NJ (2003) White cell telomere length and risk of premature myocardial infarction. Arterioscler Thromb Vasc Biol 23:842–846. https://doi.org/10.1161/01.ATV.0000067426.96344.32
Ogami M, Ikura Y, Ohsawa M, Matsuo T, Kayo S, Yoshimi N, Hai E, Shirai N, Ehara S, Komatsu R, Naruko T, Ueda M (2004) Telomere shortening in human coronary artery diseases. Arterioscler Thromb Vasc Biol 24:546–550. https://doi.org/10.1161/01.ATV.0000117200.46938.e7
Chen W, Gardner JP, Kimura M, Brimacombe M, Cao X, Srinivasan SR, Berenson GS, Aviv A (2009) Leukocyte telomere length is associated with HDL cholesterol levels: The Bogalusa heart study. Atherosclerosis 205: 620–625. https://doi.org/10.1016/j.atherosclerosis.2009.01.021
Valdes AM, Andrew T, Gardner JP, Kimura M, Oelsner E, Cherkas LF, Aviv A, Spector TD (2005) Obesity, cigarette smoking, and telomere length in women. Lancet 366:662–664. https://doi.org/10.1016/S0140-6736(05)66630-5
Epel ES, Blackburn EH, Lin J, Dhabhar FS, Adler NE, Morrow JD, Cawthon RM (2004) Accelerated telomere shortening in response to life stress. Proc Natl Acad Sci USA 101:17312–17315. https://doi.org/10.1073/pnas.0407162101
Maeda T, Oyama JI, Higuchi Y, Arima T, Mimori K, Makino N (2010) The correlation between the telomeric parameters and the clinical laboratory data in the patients with brain infarct and metabolic disorders. J Nutr Health Aging 14:793–797
Maeda T, Oyama J, Higuchi Y, Koyanagi M, Sasaki M, Arima T, Mimori K, Makino N (2011) The correlation between clinical laboratory data and telomeric status of male patients with metabolic disorders and no clinical history of vascular events. Aging Male 14:21–26. https://doi.org/10.3109/13685538.2010.502270
Maeda T, Oyama J, Higuchi Y, Nishiyama Y, Kudo Y, Yamori T, Nakazono T, Arima T, Mimori K, Makino N (2011) The physical ability of Japanese female elderly with cerebrovascular disease correlates with the telomere length and subtelomeric methylation status in their peripheral blood leukocytes. Gerontology 57:137–143. https://doi.org/10.1159/000314633
Maeda T, Oyama JI, Sasaki M, Arima T, Makino N (2011) The correlation between the clinical laboratory data and the telomere length in peripheral blood leukocytes of Japanese female patients with hypertension. J Nutr Health Aging 15:240–244
Acknowledgements
This work was supported by a Grant-in-Aid from the Ministry of Education, Science, and Culture of Japan (#15K08919).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No potential conflicts of interest were disclosed.
Rights and permissions
About this article
Cite this article
Maeda, T., Horiuchi, T. & Makino, N. Shorter somatic telomere can be an increased risk for hospitalization. Mol Cell Biochem 455, 1–5 (2019). https://doi.org/10.1007/s11010-018-3465-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-018-3465-y