Abstract
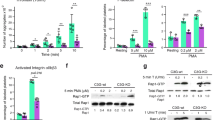
Rac1 and Cdc42 are members of the Rho family of small GTPases and have been shown to promote the formation of lamellipodia and filopodia at the leading edge of motile cells and affect cell migration. In this study the authors have investigated the activation of Rac1 and Cdc42 by thrombin or collagen using the megakaryocytic cell line, CHRF-288-11. Maximal activation of Rac1 by thrombin or collagen was observed at 3 and 1 min, respectively. Similar results were obtained for the activation of Cdc42 by thrombin or collagen. The activation of Rac1 by thrombin and collagen was inhibited by the calmodulin-specific inhibitor, W7. However, W7 had no effect on the activation of Cdc42 by thrombin or collagen. The less potent calmodulin inhibitor, W5, did not have any effect on Rac1 or Cdc42 activation by thrombin or collagen. Transient over-expression of calmodulin in CHRF cells increased the basal and thrombin-mediated activation of Rac1 when compared to control but had no effect on the basal and thrombin-mediated activation of Cdc42 when compared to control. The results suggest that calmodulin regulates the activation of Rac1 in CHRF-288-11 cells in a manner similar to that in the human platelets.





Similar content being viewed by others
References
Jaffe AB, Hall A (2005) Rho GTPases: biochemistry and biology. Annu Rev Cell Dev Biol 21:247–269
Ridley AJ (2006) Rho proteins and actin dynamics in membrane protrusions and vesicle trafficking. Trends Cell Biol 16:522–529
Bos JL, Rehmann H, Wittinghofer A (2007) A GEFs and GAPs: critical elements in the control of small G proteins. Cell 129:865–877
Soulet CS, Gendreau S, Missy K, Benard V, Plantavid M, Payrastre B (2001) Characterisation of Rac activation in thrombin- and collagen-stimulated human blood platelets. FEBS Lett 507:253–258
Pelletier S, Duhamel F, Coulombe P, Popoff MR, Meloche S (2003) Rho family GTPases are required for activation of Jak/STAT signaling by G protein-coupled receptors. Mol Cell Biol 23:1316–1333
Kurokawa K, Itoh RE, Yoshizaki H, Nakamura YO, Matsuda M (2004) Coactivation of Rac1 and Cdc42 at lamellipodia and membrane ruffles induced by epidermal growth factor. Mol Biol Cell 15:1003–1010
Itoh RE, Kiyokawa E, Aoki K, Nishioka T, Akiyama T, Matsuda M (2008) Phosphorylation and activation of the Rac1 and Cdc42 GEF Asef in A431 cells stimulated by EGF. J Cell Sci 121:2635–2642
Grill B, Schrader JW (2002) Activation of Rac1, Rac2, and Cdc42 by hemopoietic growth factors or cross-linking of the B-lymphocyte receptor for antigen. Blood 100:3183–3192
Hou Y, Ye RD, Browning DD (2004) Activation of the small GTPase Rac1 by cGMP-dependent protein kinase. Cell Signal 16:1061–1069
Barber SC, Mellor H, Gampel A, Scolding NJ (2004) S1P and LPA trigger Schwann cell actin changes and migration. Eur J Neurosci 19:3142–3150
Akbar H, Kim J, Funk K, Cancelas JA, Shang X, Chen L, Johnson JF, Williams DA, Zheng Y (2007) Genetic and pharmacologic evidence that Rac1 GTPase is involved in regulation of platelet secretion and aggregation. J Thromb Haemost 5:1747–1755
Elsaraj SM, Bhullar RP (2008) Regulation of platelet Rac1 and Cdc42 activation through interaction with calmodulin. Biochim Biophys Acta 1783:770–778
Vittet D, Mathieu MN, Launay JM, Chevillard C (1992) Platelet receptor expression on three human megakaryoblast like cell lines. Exp Hematol 20:1129–1134
Kaplan KL, Broekmam MJ, Chemoff A, Lesznik GR, Drillings M (1979) Platelet alpha-granule proteins: studies on release and subcellular localization. Blood 53:604–618
Holt JC, Niewiarowski S (1985) Biochemistry of alpha granule proteins. Semin Hematol 22:151–163
Fugman DA, Witte DP, Jones CL, Aronow BJ, Lieberman MA (1990) In vitro establishment and characterization of a human megakaryoblastic cell line. Blood 75:1252–1261
Dorn GW, Davis MG (1992) Differential megakaryocytic desensitization to platelet agonists. Am J Physiol 263:c864–c872
Dash D, Aepfelbacher M, Siess W (1995) Integrin αIIbβ3-mediated translocation of CDC42Hs to the cytoskeleton in stimulated human platelets. J Biol Chem 270:17321–17326
Lian P, Crossley L, Zhan Q, Huang RY, Coffer P, Toker A, Robinson D, Badwey JA (2001) Antagonists of calcium fluxes and calmodulin block activation of the p21-activated protein kinases in neutrophils. J Immunol 166:2643–2650
Manser E, Leung T, Salihuddin H, Zaho ZS, Lim L (1994) A brain serine/threonine protein kinase activated by Cdc42 and Rac1. Nature 367:40–46
Bagrodia S, Cerione RA (1999) PAK to the future. Trends Cell Biol 9:350–355
Price LS, Langeslag M, Klooster JPT, Hordijk PL, Jalink K, Collard JG (2003) Calcium signaling regulates translocation and activation of Rac. J Biol Chem 278:39413–39421
Yeh YC, Wang CZ, Tang MJ (2009) Discoidin receptor 1 activation suppresses α2β1 integrin-dependent cell spreading through inhibition of cdc42 activity. J Cell Physiol 218:146–156
Scita G, Tenca P, Frittoli EA, Tocchetti A, Innocenti M, Giardina G, Di Fiore PP (2000) Signaling from Ras to Rac and beyond: not just a matter of GEFs. EMBO J 19:2393–2398
Quatela SE, Philips MR (2006) Ras signaling on the Golgi. Curr Opin Cell Bio 18:162–167
Wang KL, Roufogalis BD (1999) Ca2+/calmodulin stimulates GTP binding to the ras-related protein ral-A. J Biol Chem 274:14525–14528
Acknowledgments
The authors wish to thank Dr. Mitsunobu Hoshino (National Center of Neurology and Psychiatry, Tokyo, Japan) for providing the GST-Pak1 plasmid. This study was supported by a grant from the Heart and Stroke Foundation of Manitoba to R.P.B.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xu, B., Bhullar, R.P. Regulation of Rac1 and Cdc42 activation in thrombin- and collagen-stimulated CHRF-288-11 cells. Mol Cell Biochem 353, 73–79 (2011). https://doi.org/10.1007/s11010-011-0776-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-011-0776-7