Abstract
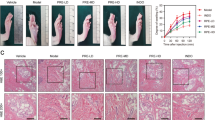
The present study was designed to investigate the anti-inflammatory activity of atrial natriuretic peptide (ANP) using λ-Carrageenan (λ-CAN)—induced acute inflammation in the mice model. A single subplantar injection of λ-CAN (1% in 0.9% saline) triggers acute paw edema. ANP (2 μg/kg body weight/day/i.p) was given 30 min prior or after administration of λ-CAN. The standard drug Aspirin (ASP) treated group was used as a positive control. A marked increase in paw thickness (32.56% p < 0.001) and thermal pain latency (0.82 ± 0.03 vs 3.6 ± 0.12; P < 0.001) were noticed in λ-CAN-induced mice as compared with control. The level of pro-inflammatory marker genes such as Nuclear factor-κB and Cyclooxygenase-2 were significantly increased (p < 0.001 respectively) in λ-CAN-induced mice as compared with control. A similar trend was noticed in pro-inflammatory cytokines such as Interleukin-6, Interleukin-1 beta, Tumour necrosis factor-alpha and Prostaglandin E2 (p < 0.001 respectively). On the contrary, ANP exhibits a marked reduction in paw edema thickness (pre-8.76% and post-9.12%, p < 0.001) and thermal pain latency in both pre-0.82 ± 0.03 vs 1.2 ± 0.06 and post-0.82 ± 0.03 vs 1.6 ± 0.02 (P < 0.001) were noticed in ANP pre-and post-treated group. Further myeloperoxidase activity, pro-inflammatory marker genes and pro-inflammatory cytokines were also reverted (p < 0.01 respectively) to near-normal levels. The present study results suggest that pre-and post-treatment of ANP positively neutralizes the λ-CAN induced acute edematic and inflammatory responses. The anti-edematic and anti-inflammatory action of ANP was found to be much more effective than that of the non-steroidal drug aspirin (ASP).







Similar content being viewed by others
References
Acheva A, Schettino G, Prise KM (2017) Pro-inflammatory signaling in a 3D organotypic skin model after low LET irradiation-NF-κB, COX-2 activation, and impact on cell differentiation. Front Immunol. https://doi.org/10.3389/fimmu.2017.00082
Birukova AA, Xing J, Fu P et al (2010) Atrial natriuretic peptide attenuates LPS-induced lung vascular leak: Role of PAK1. Am J Physiol -Lung Cell MolPhysiol 299:652–663. https://doi.org/10.1152/ajplung.00202.2009
Bensman A (2020) Non-steroidal Anti-inflammatory drugs (NSAIDs) systemic use: The risk of renal failure. Front Pediatr 7:517. https://doi.org/10.3389/fped.2019.00517
Chu S, Zhang X, Sun Y et al (2019) Atrial natriuretic peptide inhibits epithelial-mesenchymal transition (EMT) of bronchial epithelial cells through cGMP/PKG signaling by targeting Smad3 in a murine model of allergic asthma. Exp Lung Res 45:245–254. https://doi.org/10.1080/01902148.2019.1660734
Courreges AP, Najenson AC, Vatta MS, Bianciotti LG (2019) Atrial natriuretic peptide attenuates endoplasmic reticulum stress in experimental acute pancreatitis. Biochim Biophys Acta 1865:485–493. https://doi.org/10.1016/j.bbadis.2018.12.004
Curry FRE, Clark JF, Jiang Y et al (2016) The role of atrial natriuretic peptide to attenuate inflammation in a mouse skin wound and individually perfused rat mesenteric microvessels. Physiol Rep 4:1–11. https://doi.org/10.14814/phy2.12968
de Aquino PEA, Magalhães TR, Nicolau LAD et al (2017) The anti-inflammatory effects of N-methyl-(2S,4R)-trans-4-hydroxy-L-proline from Syderoxylonobtusifolium are related to its inhibition of TNF-alpha and inflammatory enzymes. Phytomedicine 24:14–23. https://doi.org/10.1016/j.phymed.2016.11.010
Elbaradey G, Elshmaa N, Hodeib H (2016) The effect of atrial natriuretic peptide infusion on intestinal injury in septic shock. J Anaesthesiol Clin Pharmacol 32:470–475. https://doi.org/10.4103/0970-9185.194778
Fontenelle TPC, Lima GC, Mesquita JX et al (2018) Lectin obtained from the red seaweed Bryothamniontriquetrum: secondary structure and anti-inflammatory activity in mice. Int J BiolMacromol 112:1122–1130. https://doi.org/10.1016/j.ijbiomac.2018.02.058
Gogulamudi VR, Mani I, Subramanian U, Pandey KN (2019) Genetic disruption of Npr1 depletes regulatory T cells and provokes high levels of pro-inflammatory cytokines and fibrosis in the kidneys of female mutant mice. Am J Physiol 316:F1254–F1272. https://doi.org/10.1152/ajprenal.00621.2018
Gøtzsche PC, Johansen HK (2005) Short-term low-dose corticosteroids vs placebo and nonsteroidalantiinflammatory drugs in rheumatoid arthritis. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.cd000189.pub2
Hisamuddin N, Shaik Mossadeq WM, Sulaiman MR et al (2019) Anti-edematogenic and anti-granuloma activity of a synthetic curcuminoid analog, 5-(3, 4-dihydroxyphenyl)-3-hydroxy-1-(2-hydroxyphenyl) penta-2, 4-dien-1-one, in mouse models of inflammation. Molecules 24(14):2614. https://doi.org/10.3390/molecules24142614
Kalaiarasu LP, Subramanian V, Sowndharrajan B, Vellaichamy E (2016) Insight into the anti-inflammatory mechanism of action of atrial natriuretic peptide, a heart derived peptide hormone: involvement of COX-2, MMPs, and NF-kB pathways. Int J Pept Res Ther 22:451–463. https://doi.org/10.1007/s10989-016-9525-9
Kiemer AK, Hartung T, Vollmar AM (2000) cGMP-mediated inhibition of TNF-α production by the atrial natriuretic peptide in murine macrophages. J Immunol 165:175–181. https://doi.org/10.4049/jimmunol.165.1.175
Kiemer AK, Lehner MD, Hartung T, Vollmar AM (2002) Inhibition of cyclooxygenase-2 by natriuretic peptides. Endocrinology 143:846–852. https://doi.org/10.1210/endo.143.3.8680
Koga H, Hagiwara S, Shingu C et al (2010) Human atrial natriuretic peptide ameliorates lps-induced acute lung injury in rats. Lung 188:241–246. https://doi.org/10.1007/s00408-010-9239-2
Kumar P, Periyasamy R, Das S et al (2014) All-trans retinoic acid and sodium butyrate enhance natriuretic peptide receptor a gene transcription: role of histone modification. Mol Pharmacol 85:946–957. https://doi.org/10.1124/mol.114.092221
Mahmoud YI, Abd El-Ghffar EA (2019) Spirulina ameliorates aspirin-induced gastric ulcer in albino mice by alleviating oxidative stress and inflammation. Biomed Pharmacother 109:314–321. https://doi.org/10.1016/j.biopha.2018.10.118
Medzhitov R (2008) Origin and physiological roles of inflammation. Nature 454:428–435. https://doi.org/10.1038/nature07201
Mezzasoma L, Antognelli C, Talesa VN (2016) Atrial natriuretic peptide down-regulates LPS/ATP-mediated IL-1β release by inhibiting NF-kB, NLRP3 inflammasome and caspase-1 activation in THP-1 cells. Immunol Res 64:303–312. https://doi.org/10.1007/s12026-015-8751-0
Mezzasoma L, Talesa VN, Romani R, Bellezza I (2020) ANP and BNP exert anti-inflammatory action via NPR-1/cGMP axis by interfering with canonical, non-canonical, and alternative routes of inflammasome activation in human THP1 cells. Int J Mol Sci 22(1):24
Morikis VA, Radecke C, Jiang Y et al (2015) Atrial natriuretic peptide down-regulates neutrophil recruitment on inflamed endothelium by reducing cell deformability and resistance to detachment force. Biorheology 52:447–463. https://doi.org/10.3233/BIR-15067
Moro C, Klimcakova E, Lolmède K et al (2007) Atrial natriuretic peptide inhibits the production of adipokines and cytokines linked to inflammation and insulin resistance in human subcutaneous adipose tissue. Diabetologia 50:1038–1047. https://doi.org/10.1007/s00125-007-0614-3
Morris CJ (2003) Carrageenan-induced paw edema in the rat and mouse. Inflammation Protocols 115–121. https://doi.org/10.1385/1-59259-374-7:115
Mpofu S, Mpofu CMA, Hutchinson D et al (2004) Steroids, non-steroidal anti-inflammatory drugs, and sigmoid diverticular abscess perforation in rheumatic conditions. Ann Rheum Dis 63:588–590. https://doi.org/10.1136/ard.2003.010355
Necas J, Bartosikova L (2013) Carrageenan: a review. Vet Med (praha) 58:187–205. https://doi.org/10.17221/6758-VETMED
Nojiri T, Hosoda H, Tokudome T et al (2014) Atrial natriuretic peptide inhibits lipopolysaccharide-induced acute lung injury. Pulm Pharmacol Ther 29:24–30. https://doi.org/10.1016/j.pupt.2014.01.003
Nojiri T, Hosoda H, Kimura T et al (2015) Atrial natriuretic peptide protects against cisplatin-induced acute kidney injury. Cancer Chemother Pharmacol 75:123–129. https://doi.org/10.1007/s00280-014-2624-4
Okamoto A, Nojiri T, Konishi K et al (2017) Atrial natriuretic peptide protects against bleomycin-induced pulmonary fibrosis via vascular endothelial cells in mice. Respir Res 18:1–4. https://doi.org/10.1186/s12931-016-0492-7
Pandey KN (2014) Guanylylcyclase/natriuretic peptide receptor-A signaling antagonizes phosphoinositide hydrolysis, Ca2+ release, and activation of protein kinase C. Front Mol Neurosci 7:1–14. https://doi.org/10.3389/fnmol.2014.00075
Pandey KN (2019) Genetic ablation and guanylylcyclase/natriuretic peptide receptor-a: impact on the pathophysiology of cardiovascular dysfunction. Int J Mol Sci. https://doi.org/10.3390/ijms20163946
Pandey KN (2021) Molecular signaling mechanisms and function of natriuretic peptide receptor-A in the pathophysiology of cardiovascular homeostasis. Front Physiol 12:693099
Periyasamy R, Das S, Pandey KN (2019) Genetic disruption of guanylylcyclase/natriuretic peptide receptor-Aupregulates renal (pro) renin receptor expression in Npr1 null mutant mice. Peptides 114:17–28. https://doi.org/10.1016/j.peptides.2019.03.001
Pober JS, Sessa WC (2007) Evolving functions of endothelial cells in inflammation. Nat Rev Immunol 7:803–815. https://doi.org/10.1038/nri2171
Potter LR, Abbey-Hosch S, Dickey DM (2006) Natriuretic peptides, their receptors, and cyclic guanosine monophosphate-dependent signaling functions. Endocr Rev 27:47–72. https://doi.org/10.1210/er.2005-0014
Potter LR, Yoder AR, Flora DR et al (2009) Natriuretic peptides: their structures, receptors, physiologic functions and therapeutic applications. Handb Exp Pharmacol 191:341–366. https://doi.org/10.1007/978-3-540-68964-5_15
Song Z, Cui Y, Ding MZ et al (2013) Protective effects of recombinant human brain natriuretic peptide against LPS-Induced acute lung injury in dogs. IntImmunopharmacol 17:508–512. https://doi.org/10.1016/j.intimp.2013.05.028
Song W, Wang H, Wu Q (2015) Atrial natriuretic peptide in cardiovascular biology and disease (NPPA). Gene 569:1–6. https://doi.org/10.1016/j.gene.2015.06.029
SowndharRajan B, Krishnan K, Vellaichamy E (2021) Diet-derived advanced glycation end products (dAGEs) induce proinflammatory cytokine expression in cardiac and renal tissues of experimental mice: protective effect of curcumin. Cardiovasc Toxicol. https://doi.org/10.1007/s12012-021-09697-4
Subramanian V, Vellaichamy E (2014) Atrial natriuretic peptide (ANP) inhibits DMBA/croton oil induced skin tumor growth by modulating NF-κB, MMPs, and infiltrating mast cells in swiss albino mice. Eur J Pharmacol 740:388–397. https://doi.org/10.1016/j.ejphar.2014.07.024
Taher YA, Samud AM, El-Taher FE et al (2015) Experimental evaluation of anti-inflammatory, antinociceptive and antipyretic activities of clove oil inmice. Libyan J Med 10:1–7. https://doi.org/10.3402/ljm.v10.28685
Tsujimoto S, Mokuda S, Matoba K et al (2018) The prevalence of endoscopic gastric mucosal damage in patients with rheumatoid arthritis. PLoS ONE 13:1–11. https://doi.org/10.1371/journal.pone.0200023
Tsukagoshi H, Shimizu Y, Kawata T et al (2001) Atrial natriuretic peptide inhibits tumor necrosis factor-alpha production by interferon-gamma-activated macrophages via suppression of p38 mitogen-activated protein kinase and nuclear factor-kappa B activation. RegulPept 99:21–29. https://doi.org/10.1016/S0167-0115(01)00218-X
Varela ML, Mogildea M, Moreno I, Lopes A (2018) Acute inflammation and metabolism. Inflammation 41:1115–1127. https://doi.org/10.1007/s10753-018-0739-1
Vellaichamy E, Zhao D, Somanna N, Pandey KN (2007) Genetic disruption of guanylylcyclase/natriuretic peptide receptor-A up-regulates ACE and AT1 receptor gene expression and signaling: role in cardiac hypertrophy. Physiol Genomics 31:193–202. https://doi.org/10.1152/physiolgenomics.00079.2007
Vellaichamy E, Das S, Subramanian U et al (2014) Genetically altered mutant mouse models of guanylylcyclase/natriuretic peptide receptor-A exhibit the cardiac expression of pro-inflammatory mediators in a gene-dose-dependent manner. Endocrinology 155:1045–1056. https://doi.org/10.1210/en.2013-1416
Volpe M, Carnovali M, Mastromarino V (2016) The natriuretic peptides system in the pathophysiology of heart failure: from molecular basis to treatment. ClinSci 130:57–77. https://doi.org/10.1042/CS20150469
Watanabe T, Fujiwara Y, Chan FKL (2020) Current knowledge on non-steroidal anti-inflammatory drug-induced small-bowel damage: a comprehensive review. J Gastroenterol 55:481–495. https://doi.org/10.1007/s00535-019-01657-8
Wienecke T, Gøtzsche PC (2004) Paracetamol versus non-steroidal anti-inflammatory drugs for rheumatoid arthritis. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.cd003789.pub2
Xing J, Birukova AA (2010) ANP attenuates inflammatory signaling and Rho pathway of lung endothelial permeability induced by LPS and TNFα. Microvasc Res 79:56–62. https://doi.org/10.1016/j.mvr.2009.11.006
Yilmazer N, Coskun C, Gurel-Gurevin E et al (2016) Antioxidant and anti-inflammatory activities of a commercial noni juice revealed by carrageenan-induced paw edema. Pol J Vet Sci 19:589–595. https://doi.org/10.1515/pjvs-2016-0074
Young JM, Spires DA, Bedord CJ et al (1984) The mouse ear inflammatory response to topical arachidonic acid. J Invest Dermatol 82:367–371. https://doi.org/10.1111/1523-1747.ep12260709
Zhang JM, An J (2007) Cytokines, inflammation and pain. Int Anesthesiol Clin 45(2):27. https://doi.org/10.1097/AIA.0b013e318034194e
Acknowledgements
Dr. EV greatly acknowledges UGC-SAP DRS-II (F.5/2015/DRS-II (SAP-II), India, for the financial support. LK wishes to thank the Indian Council of Medical Research (ICMR) (45/7/2019-IMM/BMS) for the Senior Research Fellowship (SRF). AB wishes to thank the Indian council of medical research (ICMR) (F.No.45/28/2020-BIO/BMS) for the Senior Research Fellowship (SRF). KK wishes to thank the Indian Council of Medical Research (ICMR) (45/29/2018/TM/BMS) for the Senior Research Fellowship (SRF). SD wish to thank the University Grant Commission-Basic Scientific Research (UGC-BSR) (GCCO/A-2/UGC/ MERITORIOUS/2015) fellowship in science.
Funding
Dr. EV greatly acknowledges UGC-SAP DRS-II (F.5/2015/DRS-II (SAP-II), India, for the financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical Approval
All the animal studies were approved by the Institutional Animal Ethics Committee (IAEC) of the University of Madras IAEC No: 01/02/2018 and were performed in accordance with CPCSEA guidelines.
Informed Consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kalaiarasu, L.P., Babu, A.A., Krishnan, K. et al. Atrial Natriuretic Peptide (ANP) Suppress λ-Carrageenan-Induced Acute Paw Edema by Modulating Pro-inflammatory Genes in Swiss Albino Mice. Int J Pept Res Ther 28, 138 (2022). https://doi.org/10.1007/s10989-022-10444-8
Accepted:
Published:
DOI: https://doi.org/10.1007/s10989-022-10444-8