Abstract
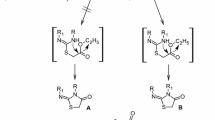
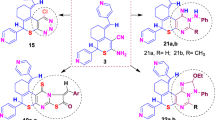
Herein, we report design and synthesis of series of adamantane derivatives containing modified peptides with thiazol moiety. New compounds were synthesized in solution using TBTU (2-(1H-Benzotriazole-1-yl)-1,1,3,3-tetramethylaminium tetrafluoroborate) as coupling agent. All derivatives were obtained with good yields. The antiviral activity of newly synthesized compounds against influenza virus H1N1 is studied. Cytotoxicity assay for determination of CC50 were done and IC50 values were calculated. The rimantadine analogue with thiazole ring Gly-Thz-rimantadine showed good activity against influenza virus A/Hongkong/68 with IC50 = 0.11 μg/mL and CC50 = 50 μg/mL. In addition, antimicrobial and antifungal activity against model strains of gram positive (Bacillus cereus), gram negative (Escherichia coli) microorganisms and fungi strain Yarrowia lipolytica 3344 were investigated. The compounds Gly-Thz-rimantadine with good antiviral activity also showed very good antifungal activity in two different concentrations.




Similar content being viewed by others
References
Angelova T, Rangelova N, Yuryev R, Georgieva N, Müller R (2012) Antibacterial activity of SiO2/hydroxypropyl cellulose hybrid materials containing silver nano particles. Mater Sci Eng C 32:1241–1246. https://doi.org/10.1016/j.msec.2012.03.015
Bender C, Hall H, Huang J, Klimov A, Cox N, Hay A, Gregory V, Cameron K, Lim W, Subbarao K (1999) Characterization of the surface proteins of influenza A (H5N1) viruses isolated from humans in 1997-1998. Virology 254:115–123. https://doi.org/10.1006/viro.1998.9529
Bennett JE, Dolin R, Martin Blaser M (2015) Principles and practice of infectious diseases, 8th edn. Elsevier, Philadelphia
Bozdaganyan ME, Orekhov PS, Bragazzi NL, Panatto D, Amicizia D, Pechkova E, Nicolini C, Gasparini R (2014) Docking and molecular dynamics (MD) simulations in potential drugs discovery: an application to influenza virus M2 protein. Am J Biochem Biotech 10:180–188. https://doi.org/10.3844/ajbbsp.2014.180.188
Brotchie J (2010) Antidyskinetic actions of amantadine in Parkinson’s disease: are benefits maintained in the long term? Expert Rev Neurother 10:871–873. https://doi.org/10.1586/ern.10.70
Charalampopoulos D, Rastall RA (2009) Prebiotics and probiotics science and technology. Springer, New York
Chen JJ, Swope DM (2007) Pharmacotherapy for Parkinson’s disease. Pharmacotherapy 27(12 Pt 2):161S–173S. https://doi.org/10.1592/phco.27.12part2.161s
Escherichia coli (2019) Centers for Disease Control and Prevention, National Center for Emerging and Zoonotic Infectious Diseases, Division of Foodborne, Waterborne, and Environmental Diseases
Gaiday A, Levandovskiy I, Byler K, Shubina T (2008) Mechanism of influenza a M2 ion-channel inhibition: a docking and QSAR study. In: Computational science—ICCS 2008. 8th international conference, Kraków, Poland, June 23–25, 2008. Proceedings, Part II, pp 360–368. https://doi.org/10.1007/978-3-540-69387-1_40
Karpenko I, Deev S, Kiselev O, Charushin V, Rusinov V, Ulomsky E, Deeva E, Yanvarev D, Ivanov A, Smirnova O, Kochetkov S, Chupakhin O, Kukhanova M (2010) Antiviral properties, metabolism, and pharmacokinetics of a novel azolo-1, 2, 4-triazine-derived inhibitor of influenza A and B virus replication. Antimicrob Agents Chemother 54:2017–2022. https://doi.org/10.1128/AAC.01186-09
Knorr R, Trzeciak A, Bannwarth W, Gillessen D (1989) New coupling reagents in peptide chemistry. Tetrahedron Lett 30:1927–1930. https://doi.org/10.1016/S0040-4039(00)99616-3
Kolocouris A, Johnson B, Tzitzoglaki C, Gay NC, Busath DD (2013) Amantadine analogs that inhibit Mdck cell infection by influenza a with M2 (S31N). Biophys J 104:277a. https://doi.org/10.1016/j.bpj.2012.11.1555
Long J, Wright E, Molesti E, Temperton N, Barclay W (2015) Antiviral therapies against Ebola and other emerging viral diseases using existing medicines that block virus entry. F1000Research. https://doi.org/10.12688/f1000research.6085.2
Madzak C (2015) Yarrowia lipolytica: recent achievements in heterologous protein expression and pathway engineering. Appl Microbiol Biotechnol 99:4559–4577. https://doi.org/10.1007/s00253-015-6624-z
Moorthy HN, Poongavanam NS, Pratheepa V (2014) Viral M2 ion channel protein: a promising target for anti-influenza drug discovery. Mini Rev Med Chem 14:819–830
Rey-Carrizo M, Torres E, Ma C, Barniol-Xicota M, Wang J, Wu Y, Naesens L, DeGrado WF, Lamb RA, Pinto LH, Vázquez S (2013) 3-Azatetracyclo [5.2. 1.15, 8.01, 5] undecane derivatives: from wild-type inhibitors of the M2 ion channel of influenza A virus to derivatives with potent activity against the V27A mutant. J Med Chem 56:9265–9274. https://doi.org/10.1021/jm401340p
Ryan KJ, Ray CG (2004) Sherris medical microbiology, 4th edn. McGraw Hill, New York
Shibnev VA, Garaev TM, Finogenova MP, Shevchenko ES, Burtseva EI (2012) Search for new drugs: some pathways to overcoming drug resistance of influenza a virus to adamantane derivatives. Pharm Chem J. https://doi.org/10.1007/s11094-012-0723-2
Singleton P (1999) Bacteria in biology, biotechnology and medicine, 5th edn. Wiley, Hoboken
Smirnova TD, Danilenko DM, Eropkin M, Deeva EG, Kiselev OI (2011) Influence of rimantadine, ribavirine and triazavirine on influenza A virus replication in human monolayer and lymphoblastoid cell lines. Antibiot Khimioter 56:11–16
Smith PW, Whittington AR, Sollis SL, Howes PD, Taylor NR (1997) Novel inhibitors of influenza sialidases related to zanamivir. Heterocyclic replacements of the glycerol sidechain. Bioorg Med Chem Lett 7:2239–2242. https://doi.org/10.1016/S0960-894X(97)00399-5
Spilovska K, Zemek F, Korabecny J, Nepovimova E, Soukupa O, Windisch M, Kuca K (2016) Adamantane—a lead structure for drugs in clinical practice. Curr Med Chem 23:3245–3266. https://doi.org/10.2174/0929867323666160525114026
Stankova IG, Simeonov MF, Maximova V, Galabov AS, Golovinsky EV (1999) Synthesis and anti-virus activity of some nucleosides analogues. Zeitschrift für Naturforschung C 54:75–83. https://doi.org/10.1515/znc-1999-1-214
Stouffer AL, Acharya R, Salom D, Levine AS, Di Costanzo L, Soto CS, Tereshko V, Nanda V, Stayrook S, DeGrado WF (2008) Structural basis for the function and inhibition of an influenza virus proton channel. Nature 451:596–599. https://doi.org/10.1038/nature06528
Tenaillon O, Skurnik D, Picard B, Denamur E (2010) The population genetics of commensal Escherichia coli. Nat Rev Microbiol 8:207–217. https://doi.org/10.1038/nrmicro2298
Vogt RL, Dippold L (2005) Escherichia coli O157:H7 outbreak associated with consumption of ground beef, June-July 2002. Public Health Rep 120:174–178. https://doi.org/10.1177/003335490512000211
Wang J, Wu Y, Ma C, Fiorin G, Wang J, Pinto LH, Lamb RA, Klein ML, Degrado WF (2013) Structure and inhibition of the drug-resistant S31 N mutant of the M2 ion channel of influenza A virus. Proc Natl Acad Sci USA 110:1315–1320. https://doi.org/10.1073/pnas.1216526110
Zarubaev VZ, Golod EL, Anfimov PM, Shtro AA, Saraev VV, Gavrilov AS, Logvinov AV, Kiselev OI (2010) Synthesis and anti-viral activity of azolo-adamantanes against influenza A virus. Bioorg Med Chem 18:839–848. https://doi.org/10.1016/j.bmc.2009.11.047
Zoidis G, Fytas C, Papanastasiou I, Foscolos GB, Fytas G, Padalko E, De Clercq E, Naesens L, Neyts J, Kolocouris N (2006) Heterocyclic rimantadine analogues with antiviral activity. Bioorg Med Chem 14:3341–3348. https://doi.org/10.1016/j.bmc.2005.12.056
Zoidis G, Kolocouris N, Kelly JM, Prathalingam SR, Naesens L, De Clercq E (2010) Design and synthesis of bioactive adamantanaminoalcohols and adamantanamines. Eur J Med Chem 45:5022–5030. https://doi.org/10.1016/j.ejmech.2010.08.009
Acknowledgements
All strains of microorganisms, included in the Bulgarian National Collection, was kindly provided by the National Bank for Industrial Microorganisms and Cell Cultures (NBIMCC), Sofia, Bulgaria. We are grateful to South-West University “Neofit Rilski”, Blagoevgrad for financial support by Project RPY-A3/19.
Author information
Authors and Affiliations
Contributions
IS and DD contributed to the study conception and design of aimed compounds as well as in the writing, review and editing of the manuscript. Synthesis, isolation and characterization of newly synthesized compounds is realized by IS, KC and RC. Material preparation, data collection and interpretation of antiviral analysis is done by LM and AG. Material preparation, data collection and interpretation of antimicrobial analysis is realized by DM.
Corresponding author
Ethics declarations
Conflict of interest
Authors Ivanka Stankova, Kiril Chuchkov, Radoslav Chayrov, Luchia Mukova, Angel Galabov, Desislava Marinkova and Dancho Danalev declare that they have no conflict of interest. This work is not related to any experiments with human participants or animals.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Stankova, I., Chuchkov, K., Chayrov, R. et al. Adamantane Derivatives Containing Thiazole Moiety: Synthesis, Antiviral and Antibacterial Activity. Int J Pept Res Ther 26, 1781–1787 (2020). https://doi.org/10.1007/s10989-019-09983-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10989-019-09983-4