Abstract
CoFe2O4 has been attracting attention for its ferrimagnetism applicable to spin transfer, resonance imaging, drug delivery etc. CoFe2O4 thin films were synthesized on Si(100) substrates by using a sol–gel deposition process. The CoFe2O4 specimen produced by post-annealing in air at 800 °C showed flat surface and polycrystalline grains with no secondary phase. The specimen exhibited magnetic hysteresis curve with magnetization up to 415 emu/cm3 and coercivity of 1.7 kOe. Such a large magnetization implies migration of a number of Co2+ ions from octahedral to tetrahedral sites of the spinel lattice. The distribution of Co2+ ions among tetrahedral and octahedral sites of CoFe2O4 was estimated by curve-fitting analysis on the Raman scattering spectrum of the specimen. The result suggests 30% of the Co2+ ions residing in the tetrahedral sites. The coexistence of Co2+ ions in both tetrahedral and octahedral sites of CoFe2O4 was also detectable by using Co 2p X-ray photoelectron spectroscopy.

The CoFe2O4 thin-film specimen deposited on Si(100) substrate through a sol–gel process exhibited surface flatness and phase-pure polycrystalline grains. The specimen showed magnetic hysteresis curve with magnetization up to 415 emu/cm3, coercivity of 1.7 kOe and remnant magnetization of 170 emu/cm3. The observed magnetization exceeded theoretical value expected for pure inverse spinel CoFe2O4 and can be explained in terms of migration of a fraction of Co2+ ions from octahedral to tetrahedral sites of the spinel lattice. The inversion parameter of the CoFe2O4 specimen was estimated to be 0.70 through a curve-fitting on its Raman scattering spectrum. The curve-fitting analysis on Co 2p XPS spectrum of the specimen turned out to be supportive of the Raman analysis.
Highlights
-
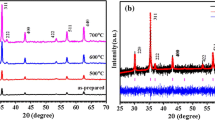
CoFe2O4 thin films with high crystalline quality have been fabricated on Si(100) substrates by using a sol–gel technique and post-annealing in air at 800 °C.
-
Thin-film CoFe2O4 specimen exhibits magnetization (415 emu/cm3) that is larger than theoretical value (380 emu/cm3) expected for pure inverse spinel CoFe2O4.
-
The enhanced magnetization of CoFe2O4 is ascribed to the migration of Co2+ ions to the tetrahedral sites of the spinel lattice.
-
The inversion parameter of CoFe2O4 is estimated to be 0.70 through a curve-fitting analysis on the Raman scattering spectrum of the specimen.





Similar content being viewed by others
References
Chandramohan P, Srinivasan MP, Velmurugan S, Narasimhan SV (2011) J Solid State Chem 184:89
Georgiadou V, Tangoulis V, Arvanitidis I, Kalogirou O, Dendrinou-Samara C (2015) J Phys Chem C 119:8336
Nappini S, Magnano E, Bondino F, Pis FI, Barla A, Fantechi E, Pineider F, Sangregorio C, Vaccari L, Venturella L, Baglioni P (2015) J Phys Chem C 119:25529
Dun C, Xi G, Zhang Y, Zhao T, Liu Y, Heng X, Yao L (2018) Ceram Int 44:20984
Thang PD, Rijnders G, Blank DHA (2007) J Magn Magn Mater 310:2621
Kumar P, Sharma SK, Knobel M, Singh M (2010) J Alloy Compd 508:115
Yadav SP, Shinde SS, Kadam AA, Rajpure KY (2013) J Alloy Compd 555:330
Soeya S, Hayakawa J, Takahashi H, Ito K, Yamamoto C, Kida A, Asano H, Matsui M (2002) Appl Phys Lett 80:823
Phase DM, Tiwari S, Prakash R, Dubey A, Sathe VG, Choudhary RJ (2006) J Appl Phys 100:123703
Indhrajothi R, Prakash I, Venkateswarlu M, Satyanarayana N (2015) New J Chem 39:4601
Tiwari S, Choudhary RJ, Prakash R, Phase DM (2007) J Phys: Condens Matter 19:176002
Wang Z, Downs RT, Pischedda V, Shetty R, Saxena SK, Zha CS, Zhao YS, Schiferl D, Waskowska A (2003) Phys Rev B 68:094101
Sharma D, Khare N (2014) Appl Phys Lett 105:032404
Carta D, Casula MF, Falqui A, Loche D, Mountjoy G, Sangregorie C, Corrias A (2009) J Phys Chem C 113:8606
Li R, Sun C, Liu J, Zhen Q (2017) RSC Adv 7:50546
Kim KJ, Lee JH, Koh TY, Kim MH (2016) Electro Acta 200:84
Kim KJ, Koh TY (2016) J Sol-Gel Sci Technol 77:528
Acknowledgements
This work was supported by Konkuk University in the program year of 2017.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kim, K.J., Park, J. Spectroscopic investigation on tetrahedral Co2+ in thin-film CoFe2O4. J Sol-Gel Sci Technol 92, 40–44 (2019). https://doi.org/10.1007/s10971-019-05099-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-019-05099-9