Abstract
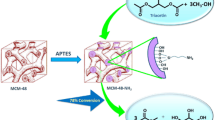
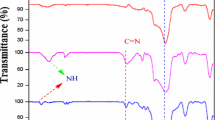
In this study, we synthesized new silica-MCM-41 modified with kryptofix 23 immobilized with MnCl2·2H2O, an effective and heterogeneous reusable nanocatalyst, and characterized using FT-IR, SEM, TEM, EDS, XRD and BET techniques. This nanocatalyst catalyzed the oxidation of primary and secondary alcohols in the presence of H2O2 as an oxidant to corresponding carbonylated compounds under reflux in high yields. The oxidation of some primary alcohols gave the mixture of corresponding aldehyde and carboxylic acid and secondary alcohols gave corresponding ketones. Some cyclic secondary alcohols such as cyclohexanol (1m) and 1,4-cyclohexanediol (1,4-CHD, 1g) underwent ring opening oxidation and formed adipic acid and succinic acid, respectively, under the same condition.











Similar content being viewed by others
References
E.P. Ng, S.N.M. Subari, O. Marie, R.R. Mukti, J.C. Juan, Appl. Catal. A 450, 34 (2012)
M. Manzano, V. Aina, C.O. Arean, F. Balas, V. Cauda, M. Colilla, M.R. Delgado, M. Vallet-Regi, Chem. Eng. J. 137, 30 (2008)
D. Rath, S. Rana, K.M. Parida, RSC Adv. 4, 57111 (2014)
M. Nikoorazm, A. Ghorbani-Choghamarani, M. Khanmoradi, Appl. Organomet. Chem. 30, 236 (2016)
X. Hu, G.K. Chuah, S. Jaenicke, Appl. Catal. A 217, 1 (2001)
G. Karthikeyan, A. Pandurangan, Mol. Catal. 311, 36 (2009)
M. Hajjami, L. Shiri, A. Jahanbakhshi, Appl. Organomet. Chem. 29, 668 (2015). https://doi.org/10.1002/aoc.3348
M. Mandal, V. Nagaraju, B. Sarma, G.V. Karunakar, K.K. Bania, ChemPlusChem 80, 749 (2015)
S. Jana, B. Dutta, R. Bera, S. Koner, Langmuir 23, 2492 (2007). https://doi.org/10.1021/la062409t
T. Kikhavani, P. Moradi, M. Mashari-Karir, J. Naji, Appl. Organomet. Chem. 36, e6895 (2022). https://doi.org/10.1002/aoc.6895
M. Nikoorazm, B. Tahmasbi, S. Gholami, P. Moradi, Appl. Organomet. Chem. 34, e5919 (2020). https://doi.org/10.1002/aoc.5919
P. Moradi, B. Zarei, Y.A. Tyula, M. Nikoorazm, Appl. Organomet. Chem. 37, e7020 (2023). https://doi.org/10.1002/aoc.7020
M. Nikoorazm, Z. Rezaei, B. Tahmasbi, J. Porous Mater. 27, 671 (2020). https://doi.org/10.1007/s10934-019-00835-6
A. Jabbari, M. Nikoorazm, P. Moradi, J. Porous Mater. 30, 1395 (2023). https://doi.org/10.1007/s10934-023-01427-1
A. Jabbari, M. Nikoorazm, P. Moradi, Res. Chem. Intermed. 49, 1485 (2023). https://doi.org/10.1007/s11164-023-04977-w
M. Nikoorazm, B. Tahmasbi, S. Gholami, M. Khanmoradi, Y.A. Tyula, M. Darabi, M. Koolivand, Polyhedron 244, 116587 (2023). https://doi.org/10.1016/j.poly.2023.116587
B. Tahmasbi, M. Nikoorazm, P. Moradi, Y.A. Tyula, RSC Adv. 12, 34303 (2022). https://doi.org/10.1039/D2RA05413B
L. Zhang, Y. Li, H. Zhou, Energy 149, 414 (2018). https://doi.org/10.1016/j.energy.2018.02.060
B.M. Abu-Zied, M.M. Alam, A.M. Asiri, W. Schwieger, M.M. Rahman, Colloid Surf. A 562, 161 (2019). https://doi.org/10.1016/j.colsurfa.2018.11.024
H. Batmani, N. Noroozi Pesyan, F. Havasi, Microporous Mesoporous Mater. 257, 27 (2018). https://doi.org/10.1016/j.micromeso.2017.08.024
F. Qi, W. Chu, B. Xu, Appl. Catal. B 134–135, 324 (2013). https://doi.org/10.1016/j.apcatb.2013.01.038
I.I. Slowing, J.L. Vivero-Escoto, C.-W. Wu, V.S.-Y. Lin, Adv. Drug Deliv. Rev. 60, 1278 (2008). https://doi.org/10.1016/j.addr.2008.03.012
K. Morishige, H. Fujii, M. Uga, D. Kinukawa, Langmuir 13, 3494 (1997)
M. Aalinejad, N. Noroozi Pesyan, N. Heidari, H. Batmani, A. Danandeh Asl, Appl. Organomet. Chem. 33, e4878 (2019). https://doi.org/10.1002/aoc.4878
A. Taghizadeh, N. Noroozi Pesyan, H. Alamgholiloo, G. Sheykhaghaei, Appl. Organomet. Chem. 36, e6787 (2022). https://doi.org/10.1002/aoc.6787
S. Mohammadlou, N. Noroozi Pesyan, J. Porous Mater. 30, 11 (2023). https://doi.org/10.1007/s10934-022-01315-0
M. Aalinejad, N. Noroozi Pesyan, H. Alamgholiloo, Colloids Surf. A 634, 127956 (2022). https://doi.org/10.1016/j.colsurfa.2021.127956
M. Aalinejad, N. Noroozi Pesyan, E. Doustkhah, Mol. Catal. 494, 111117 (2020). https://doi.org/10.1016/j.mcat.2020.111117
P.A. Wright, R.E. Morris, P.S. Wheatley, Dalton Trans. 2007, 3559 (2007). https://doi.org/10.1039/B710352B
A. Anzellotti, A. McFarland, K.F. Olson, Anal. Methods 5, 4317 (2013). https://doi.org/10.1039/C3AY40995C
R. Mozafari, M. Ghadermazi, RSC Adv. 10, 15052 (2020). https://doi.org/10.1039/d0ra01671c
R.M. Izatt, K. Pawlak, J.S. Bradshaw, R.L. Bruening, Chem. Rev. 91, 1721 (1991). https://doi.org/10.1021/cr00008a003
P. Chandra, T. Ghosh, N. Choudhary, A. Mohammad, S.M. Mobin, Coord. Chem. Rev. 411, 213241 (2020). https://doi.org/10.1016/j.ccr.2020.213241
Z. Zhang, Z. Zha, C. Gan, C. Pan, Y. Zhou, Z. Wang, M.-M. Zhou, J. Org. Chem. 71, 4339 (2006). https://doi.org/10.1021/jo060372b
A.R. Gholap, K. Venkatesan, R. Pasricha, T. Daniel, R.J. Lahoti, K.V. Srinivasan, J. Org. Chem. 70, 4869 (2005). https://doi.org/10.1021/jo0503815
Sh. Hamedimehr, K. Ojaghi Aghbash, N. Noroozi Pesyan, ACS Omega 8, 8227 (2023)
H. Chen, S. Fu, L. Fu, H. Yang, D. Chen, Minerals 9(5), 264 (2019). https://doi.org/10.3390/min9050264
T.T.M. Le, N.X.D. Mai, H.K.T. Ta, T.L.H. Doan, L.H.T. Nguyen, New J. Chem. 39, 6474 (2015)
K. Li, Z. Zeng, J. Xiong, L. Yan, H. Guo, S. Liu, Y. Dai, T. Chen, Colloids Surf. A 465, 113 (2015)
K. Fujitani, T. Mizutani, T. Oida, T. Kawase, J. Oleo Sci. 58, 323 (2009)
R. Tayebee, M.M. Amini, M. Akbari, A. Aliakbari, Dalton Trans. 44, 9596 (2015). https://doi.org/10.1039/C5DT00368G
R. Tayebee, M.M. Amini, H. Rostamian, A. Aliakbari, Dalton Trans. 43, 1550 (2014). https://doi.org/10.1039/C3DT51594J
R. Tayebee, M.M. Amini, F. Nehzat, O. Sadeghi, M. Armaghan, J. Mol. Catal. A 366, 140 (2013). https://doi.org/10.1016/j.molcata.2012.09.016
N.M. Ghohe, R. Tayebee, M.M. Amini, A. Osatiashtiani, M.A. Isaacs, A.F. Lee, Tetrahedron 73, 5862 (2017). https://doi.org/10.1016/j.tet.2017.08.030
M. Moudjahed, L. Dermeche, Y. Idrissou, T. Mazari, C. Rabia, J. Chem. Sci. 134, 31 (2022). https://doi.org/10.1007/s12039-022-02028-2
M.E. Assal, M.R. Shaik, M. Kuniyil, M. Khan, A.Y. Alzahrani, A. Al-Warthan, M.R.H. Siddiqui, S.F. Adil, Catalysts 7, 391 (2017). https://doi.org/10.3390/catal7120391
J. Alcañiz-Monge, G. Trautwein, A. Garcia-Garcia, J. Mol. Catal. A 394, 211 (2014). https://doi.org/10.1016/j.molcata.2014.07.023
J.C.S. Soares, A.H.A. Gonçalves, F.M.Z. Zotin, L.R.R. de Araújo, A.B. Gaspar, Mol. Catal. 458, 223 (2018). https://doi.org/10.1016/j.mcat.2018.02.020
G. Liu, M. Chen, X. Jin, C. Song, F. He, Q. Huang, J. Environ. Chem. Eng. 9, 105422 (2021). https://doi.org/10.1016/j.jece.2021.105422
P. Jin, H. Wei, L. Zhou, D. Wei, Y. Wen, B. Zhao, X. Wang, B. Li, Mol. Catal. 510, 111705 (2021). https://doi.org/10.1016/j.mcat.2021.111705111705
J. Pisk, D. Agustin, R. Poli, Molecules 24, 783 (2019). https://doi.org/10.3390/MOLECULES24040783
J.C.S. Soares, A.H.A. Gonçalves, F.M.Z. Zotin, L.R.R. de Araújo, A.B. Gaspar, Catal. Today 381, 143 (2021). https://doi.org/10.1016/J.CATTOD.2020.07.052
H. Keypour, S.G. Saremi, H. Veisi, R. Azadbakht, RSC Adv. 6, 77020 (2016). https://doi.org/10.1039/C6RA15967B
Q. Zhou, Z. Wan, X. Yuan, J. Luo, Appl. Organomet. Chem. 30, 215 (2016). https://doi.org/10.1002/aoc.3419
M.F. Pinto, M. Olivares, A. Vivancos, G. Guisado-Barrios, M. Albrecht, B. Royo, Catal. Sci. Technol. 9, 2421 (2019). https://doi.org/10.1039/C9CY00685K
A. Shaabani, M.B. Boroujeni, M.S. Laeini, Appl. Organomet. Chem. 30, 154 (2016). https://doi.org/10.1002/aoc.3412
K. Dhanalaxmi, R. Singuru, S.K. Kundu, B.M. Reddy, A. Bhaumik, J. Mondal, RSC Adv. 6, 36728 (2016). https://doi.org/10.1039/C6RA07200C
M.T. Hafshejani, S. Saeednia, M.H. Ardakani, Z.P. Parizi, Transit. Met. Chem. 43, 579 (2018). https://doi.org/10.1007/s11243-018-0244-2
H.-K. Kwong, P.-K. Lo, K.-C. Lau, T.-C. Lau, Chem. Commun. 47, 4273 (2011). https://doi.org/10.1039/c0cc05487a
Acknowledgements
We express our gratitude to the Urmia University Research Council for supporting this research work.
Author information
Authors and Affiliations
Contributions
SM: Investigation, conceptualization, methodology. NNP: Funding acquisition and supervisor of project.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mahjabin, S., Noroozi Pesyan, N. Mesoporous MCM-41@SnPr-Kryf.23-Mn a versatile and reusable nanocatalyst for the oxidation of primary and secondary alcohols in the presence of hydrogen peroxide as oxidant. J Porous Mater 31, 557–567 (2024). https://doi.org/10.1007/s10934-023-01532-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10934-023-01532-1